HU 210
Tocris Bioscience | Catalog # 0966

Key Product Details
Description
Product Description
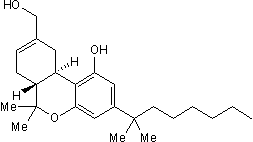
HU 210 is a highly potent cannabinoid receptor agonist (Ki values are 0.061 and 0.52 nM at cloned human CB1 and CB2 receptors respectively). Induces spatial memory deficits and suppresses hippocampal firing rates in rats. Also displays agonist activity at GPR55 (EC50 = 26 nM).
Product Specifications for HU 210
Molecular Weight
Formula
Storage
Purity
Chemical Name
CAS Number
PubChem ID
InChI Key
SMILES
The technical data provided above is for guidance only. For batch specific data refer to the Certificate of Analysis.
Solubility
| Solvent | Max Conc. mg/mL | Max Conc. mM | |
|---|---|---|---|
| Solubility | |||
| DMSO | 38.66 | 100 | |
| Ethanol | 38.66 | 100 |
Preparing Stock Solutions for HU 210
The following data is based on the product molecular weight 386.57.
Batch specific molecular weights may vary from batch to batch due to the degree of hydration, which all affect the solvent volumes required to prepare stock solutions.
| Concentration / Solvent Volume / Mass | 1 mg | 5 mg | 10 mg |
|---|---|---|---|
| 1 mM | 2.59 mL | 12.93 mL | 25.87 mL |
| 5 mM | 0.52 mL | 2.59 mL | 5.17 mL |
| 10 mM | 0.26 mL | 1.29 mL | 2.59 mL |
| 50 mM | 0.05 mL | 0.26 mL | 0.52 mL |
Calculators
Background References
References are publications that support the biological activity of the product. See our Citations tab to view 9 publications citing the usage of this product.
- Ryberg The orphan receptor GPR55 is a novel cannabinoid receptor. Br.J.Pharmacol. 2007 PMID: 17876302
- Mechoulam Enantiomeric cannabinoids: stereospecificity of psychotropic activity. Experientia 1988 PMID: 3416993
- Robinson The synthetic cannabinoid HU210 reduces spatial memory deficits and suppresses hippocampal firing rates in rats. Br.J.Pharmacol. 2007 PMID: 17502849
- Howlett Stereochemical effects of 11-OH-Δ8-tetrahydrocannabinol-dimethylheptyl to inhibit adenylate cyclase and bind to the cannabinoid receptor. Neuropharmacology 1990 PMID: 2158635
Product Documents for HU 210
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Product Specific Notices for HU 210
For research use only
Citations for HU 210
Customer Reviews for HU 210
There are currently no reviews for this product. Be the first to review HU 210 and earn rewards!
Have you used HU 210?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
