CD200, also known as OX-2, is a 45 kDa transmembrane immunoregulatory protein that belongs to the immunoglobulin superfamily (1, 2). The human CD200 cDNA encodes a 278 amino acid (aa) precursor that includes a 30 aa signal sequence, a 202 aa extracellular domain (ECD), a 27 aa transmembrane segment, and a 19 aa cytoplasmic domain. The ECD is composed of one Ig-like V-type domain and one Ig-like C2-type domain (3). A splice variant of CD200 has been described and has a truncated cytoplasmic tail. Within the ECD, human CD200 shares 76% aa sequence identity with mouse and rat CD200. CD200 is widely but not ubiquitously expressed (4). Its receptor (CD200R) is restricted primarily to mast cells, basophils, macrophages, and dendritic cells, which suggests myeloid cell regulation as the major function of CD200 (5‑7). CD200 knockout mice are characterized by increased macrophage number and activation and are predisposed to autoimmune disorders (8). CD200 and CD200R associate via their respective N-terminal Ig-like domains (9). In myeloid cells, CD200R initiates inhibitory signals following receptor‑ligand contact (6, 7, 10). In T cells, however, CD200 functions as a co‑stimulatory molecule independent of the CD28 pathway (11). Several additional CD200R-like molecules have been identified in human and mouse, but their capacity to interact with CD200 is controversial (12, 13). Several viruses encode CD200 homologs which are expressed on infected cells during the lytic phase (14, 15). Like CD200 itself, viral CD200 homologs also suppress myeloid cell activity, enabling increased viral propagation (5, 14‑16).
Human CD200 PE‑conjugated Antibody
R&D Systems | Catalog # FAB27241P

Key Product Details
Species Reactivity
Validated:
Cited:
Applications
Validated:
Cited:
Label
Antibody Source
Product Specifications
Immunogen
Gln31-Gly232
Accession # P41217.3
Specificity
Clonality
Host
Isotype
Scientific Data Images for Human CD200 PE‑conjugated Antibody
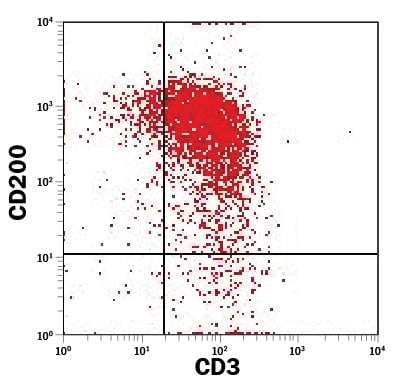
Detection of CD200 in Th17 Differentiated Human PBMCs by Flow Cytometry.
Th17 differentiated human peripheral blood mononuclear cells (PBMCs) were stained with Mouse Anti-Human CD200 PE-conjugated Monoclonal Antibody (Catalog # FAB27241P) and Mouse Anti-Human CD3e APC-conjugated Monoclonal Antibody (Catalog # FAB100A). Quadrant markers were set based on control antibody staining (Catalog # IC002P). View our protocol for Staining Membrane-associated Proteins.Detection of CD200 in Human Blood Lymphocytes by Flow Cytometry.
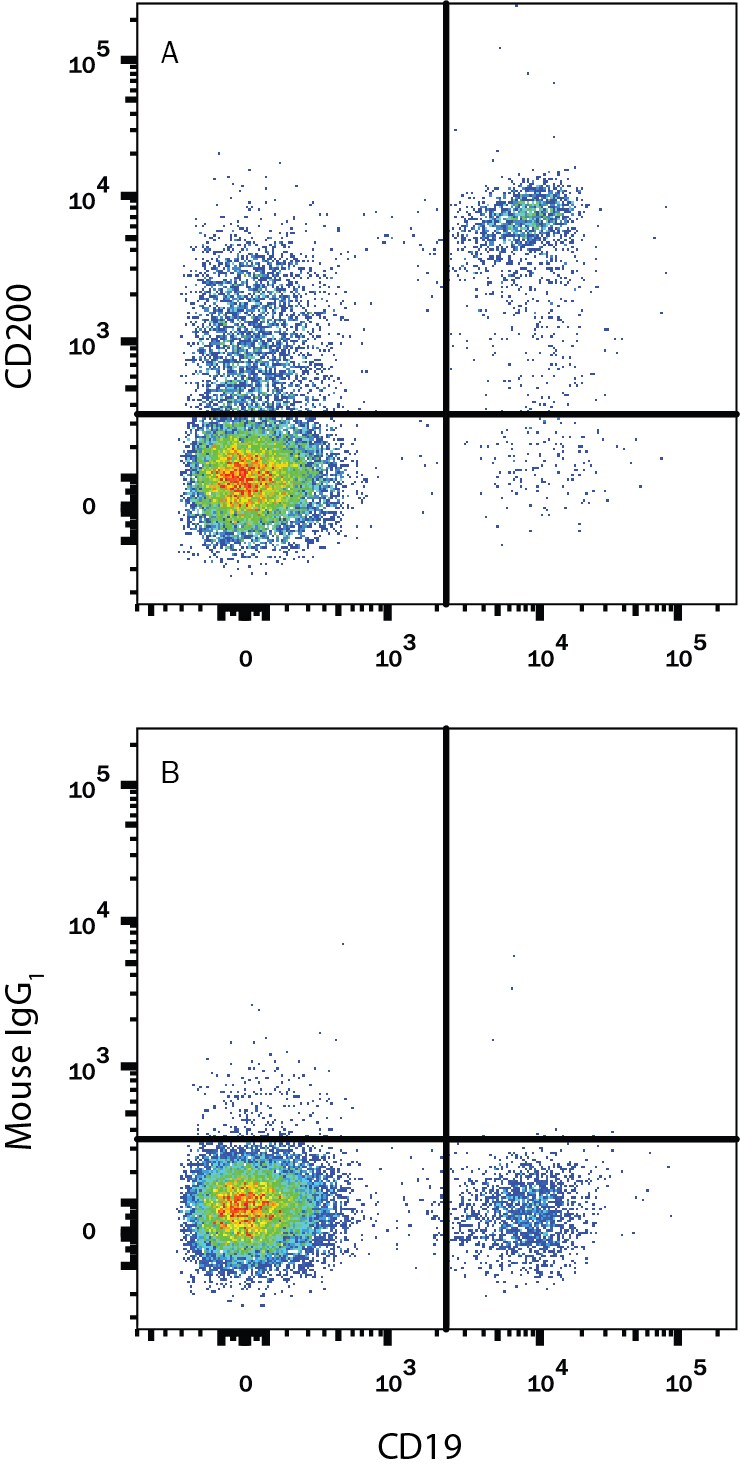
Human peripheral blood lymphocytes were stained with Mouse Anti-Human CD19 APC-conjugated Monoclonal Antibody (Catalog # FAB4867A) and either (A) Mouse Anti-Human CD200 PE-conjugated Monoclonal Antibody (Catalog # FAB27241P) or (B) Mouse IgG1Phycoerythrin Isotype Control (Catalog # IC002P). View our protocol for Staining Membrane-associated Proteins.Applications for Human CD200 PE‑conjugated Antibody
Flow Cytometry
Sample: Th17 differentiated human peripheral blood mononuclear cells (PBMCs) and human peripheral blood lymphocytes
Spectra Viewer
Plan Your Experiments
Use our spectra viewer to interactively plan your experiments, assessing multiplexing options. View the excitation and emission spectra for our fluorescent dye range and other commonly used dyes.
Spectra ViewerFlow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Formulation
Shipping
Stability & Storage
Background: CD200
References
- Gorczynski, R.M. (2005) Curr. Opin. Invest. Drugs 6:483.
- Barclay, A.N. et al. (2002) Trends Immunol. 23:285.
- McCaughan, G.W. et al. (1987) Immunogenetics 25:329.
- Wright, G.J. et al. (2001) Immunology 102:173.
- Shiratori, I. et al. (2005) J. Immunol. 175:4441.
- Cherwinski, H.M. et al. (2005) J. Immunol. 174:1348.
- Fallarino, F. et al. (2004) J. Immunol. 173:3748.
- Hoek, R.M. et al. (2000) Science 290:1768.
- Hatherley, D. and A.N. Barclay (2004) Eur. J. Immunol. 34:1688.
- Jenmalm, M.C. et al. (2006) J. Immunol. 176:191.
- Borriello, F. et al. (1997) J. Immunol. 158:4548.
- Gorczynski, R. et al. (2004) J. Immunol. 172:7744.
- Hatherley, D. et al. (2005) J. Immunol. 175:2469.
- Foster-Cuevas, M. et al. (2004) J. Virol. 78:7667.
- Cameron, C.M. et al. (2005) J. Virol. 79:6052.
- Langlais, C.L. et al. (2006) J. Virol. 80:3098.
Alternate Names
Gene Symbol
UniProt
Additional CD200 Products
Product Documents for Human CD200 PE‑conjugated Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human CD200 PE‑conjugated Antibody
For research use only
Related Research Areas
Citations for Human CD200 PE‑conjugated Antibody
Customer Reviews for Human CD200 PE‑conjugated Antibody
There are currently no reviews for this product. Be the first to review Human CD200 PE‑conjugated Antibody and earn rewards!
Have you used Human CD200 PE‑conjugated Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Liperfluo
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- View all Protocols, Troubleshooting, Illustrated assays and Webinars