EMR2 (EGF-like module-containing mucin-like receptor 2; designated CD312) is a glycoprotein belonging to the EGF-TM7 family of adhesion-type class B 7‑transmembrane (TM) receptors. EGF-like sequences within long extracellular N-termini, and a GPS (G-protein proteolytic site) domain are characteristic of this family, which is mainly expressed on cells of the immune system (1, 2). The human EMR2 cDNA encodes an 823 amino acid (aa) protein with five EGF-like domains within the first 250 aa, followed by a mucin-like stalk, a GPS domain (aa 479-530) and a 7-TM sequence (aa 531-785). The GPS domain is the site of autocatalytic cleavage, forming two cleaved portions that remain non-covalently attached as a heterodimer (1, 3). Of the first 290 aa of human EMR2, 284 aa (97%) are identical with family member CD97, likely due to gene duplication (2). The portion of human EMR2 N-terminal to the GPS domain (aa 1-478) shares 64%, 59%, 48% and 45% aa identity with corresponding regions of canine EMR2, equine EMR2, mouse CD97 and rat CD97, respectively. Alternate splicing of EMR2 creates isoforms that contain 2-5 EGF-like domains. Only the 5-EGF form contains EGF4, which is necessary for calcium-dependent binding of the EMR2/CD97 ligand, chondroitin sulfate (CS) (2, 4‑6). None of the isoforms engage the CD97 ligand, CD55 (DAF). EMR2 is restricted to myeloid cells (1, 2). EMR2 expression increases as monocytes differentiate into macrophages, and decreases with differentiation into dendritic cells (5). Activation increases neutrophil EMR2 expression (5). EMR2 localizes to the leading edge of migrating neutrophils and plays an important role in migration, adhesion and superoxide production (7). It is also thought to facilitate specific interaction of myeloid cells with peripheral B lymphocytes which express CS (6).

Key Product Details
Species Reactivity
Validated:
Human
Cited:
Human
Applications
Validated:
Western Blot, Neutralization
Cited:
Western Blot, Neutralization, Immunocytochemistry, Immunoprecipitation
Label
Unconjugated
Antibody Source
Polyclonal Sheep IgG
Loading...
Product Specifications
Immunogen
Chinese hamster ovary cell line CHO-derived recombinant human EMR2
Gln24-Gln478 (Gln24 predicted)
Accession # NP_038475
Gln24-Gln478 (Gln24 predicted)
Accession # NP_038475
Specificity
Detects human EMR2 in direct ELISAs and Western blots. In direct ELISAs, this antibody shows less than 1% cross-reactivity with recombinant mouse EMR1.
Clonality
Polyclonal
Host
Sheep
Isotype
IgG
Scientific Data Images for Human EMR2 Antibody
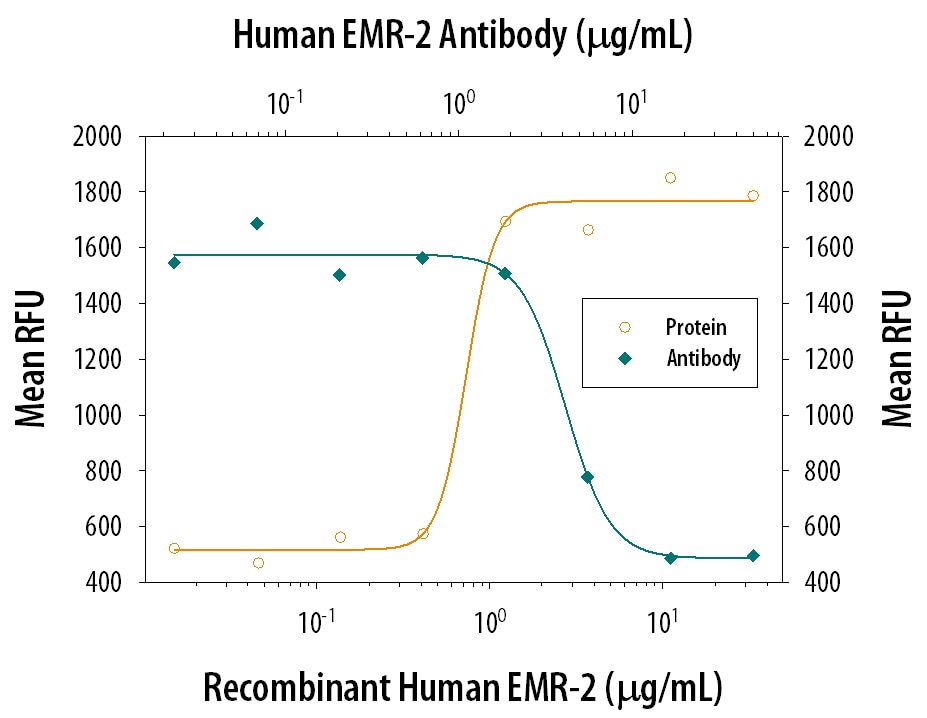
Cell Adhesion Mediated by EMR2 and Neutralization by Human EMR2 Antibody.
Recombinant Human EMR2 (Catalog # 4894-EM), immobilized onto a microplate, supports the adhesion of the NIH-3T3 mouse embryonic fibroblast cell line in a dose-dependent manner (orange line). Adhesion elicited by Recombinant Human EMR2 (1.5 µg/mL) is neutralized (green line) by increasing concentrations of Sheep Anti-Human EMR2 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF4894). The ND50 is typically 1.5-6 µg/mL.Detection of Human EMR2 by Western Blot.
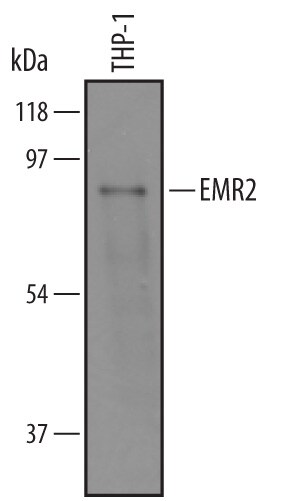
Western blot shows lysates of THP-1 human acute monocytic leukemia cell line. PVDF membrane was probed with 1 µg/mL of Sheep Anti-Human EMR2 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF4894) followed by HRP-conjugated Anti-Sheep IgG Secondary Antibody (Catalog # HAF016). A specific band was detected for EMR2 at approximately 90 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 8.Applications for Human EMR2 Antibody
Application
Recommended Usage
Western Blot
1 µg/mL
Sample: THP‑1 human acute monocytic leukemia cell line
Sample: THP‑1 human acute monocytic leukemia cell line
Neutralization
Measured by its ability to neutralize EMR2-mediated adhesion of the NIH‑3T3 mouse embryonic fibroblast cell line. Stacey, M. et al. (2003) Blood 102:29161. The Neutralization Dose (ND50) is typically 1.5-6 µg/mL in the presence of 1.5 µg/mL Recombinant Human EMR2.
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: EMR2
References
- Kwakkenbos, M.J. et al. (2004) Immunogenetics 55:655.
- Lin, H.-H. et al. (2000) Genomics 67:188.
- Lin, H.-H. et al. (2004) J. Biol. Chem. 279:31823.
- Stacey, M. et al. (2003) Blood 102:2916.
- Chang G.-W. et al. (2007) Biochem. Biophys. Res. Commun. 353:133.
- Kwakkenbos, M.J. et al. (2005) J. Leukoc. Biol. 77:112.
- Yona, S. et al. (2008) FASEB J. 22:741.
Long Name
EGF-like Module Containing, Mucin-like, Hormone Receptor-like 2
Alternate Names
CD312
Entrez Gene IDs
30817 (Human)
Gene Symbol
ADGRE2
UniProt
Additional EMR2 Products
Product Documents for Human EMR2 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human EMR2 Antibody
For research use only
Citations for Human EMR2 Antibody
Customer Reviews for Human EMR2 Antibody
There are currently no reviews for this product. Be the first to review Human EMR2 Antibody and earn rewards!
Have you used Human EMR2 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Cellular Response to Hypoxia Protocols
- R&D Systems Quality Control Western Blot Protocol
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...