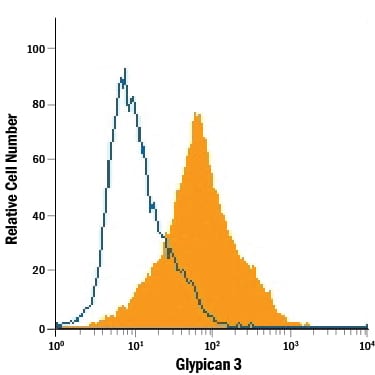
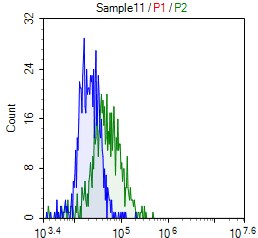
Glypicans (GPC) are a family of heparan sulfate proteoglycans that are attached to the cell surface by a glycosylphosphatidylinositol (GPI) anchor. Six members of this family have been identified in mammals (GPC1-GPC6). All glypican core proteins contain an N-terminal signal peptide, a large globular cysteine-rich domain (CRD) with 14 invariant cysteine residues, a stalk-like region containing the heparan sulfate attachment sites, and a C-terminal GPI attachment site. While glypican proteins do not share strong amino acid sequence identity (they range from 17-63%), the conserved cysteine residues in their CRDs suggests similarity in their three‑dimensional structure (1, 2).
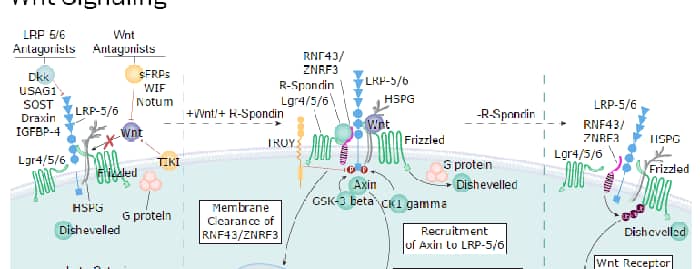
Mutations in GPC3 cause a rare disorder in humans, Simpson-Golabi-Behmel Syndrome, which is characterized by pre and postnatal overgrowth of multiple tissues and organs and an increased risk for developing embryonic tumors (3). These features are also present in the mouse knock-out of GPC3 indicating that GPC3 regulates cell survival and inhibits cell proliferation during development (4). Glypican 3 has been implicated in regulating many different signaling pathways including: IGF, FGF, BMP, and Wnt. An endoproteolytic processing of GPC3 by proprotein convertases is required for the modulation of Wnt signaling (5). Direct interaction with FGF-basic has been observed and is mediated by the heparan sulfate chains (6).