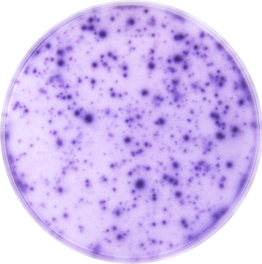
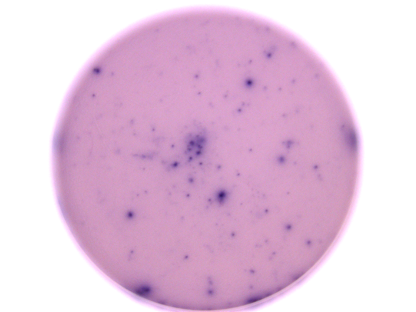
IFN-gamma (Interferon-gamma) is the prototype proinflammatory cytokine and is produced by a variety of immune cells under inflammatory conditions, notably by T cells and NK cells. It plays a key role in host defense by promoting the development and activation of Th1 cells, chemoattraction and activation of monocytes and macrophages, upregulation of antigen presentation molecules, and immunoglobulin class switching in B cells. It also exhibits antiviral, antiproliferative, and apoptotic effects. In addition, IFN-gamma functions as an anti-inflammatory mediator by promoting the development of regulatory T cells and inhibiting Th17 cell differentiation. IFN-gamma dimers signal through a receptor complex of two IFN-gamma R1 and two IFN-gamma R2 subunits.
Human IFN-gamma ELISpot Kit
R&D Systems | Catalog # EL285

Key Product Details
Species
Assay Type
Format
Assay Length
Sample Type
Product Summary for Human IFN-gamma ELISpot Kit
PRODUCT SUMMARY
ELISpot kits are highly sensitive, microplate-based assays for the detection of cytokine secreting cells. This kit is designed for the detection and enumeration of human IFN-gamma. Complete ELISpot kits are ready-to-run and require no assay development or refinement.
This ELISpot assay employs a capture antibody specific for human IFN-gamma, pre-coated onto a PVDF-backed microplate. Appropriately stimulated cells are pipetted directly into the wells and the immobilized antibody in the immediate vicinity of the secreting cells binds secreted human IFN-gamma. Following wash steps and incubation with a biotinylated detection antibody, alkaline-phosphatase conjugated to streptavidin is added. Unbound enzyme is subsequently removed by washing and a substrate solution (BCIP/NBT) is added. A blue-black colored precipitate forms at the sites of cytokine localization and appears as spots, with each individual spot representing an individual human IFN-gamma secreting cell. The spots can be counted with an automated ELISpot reader system or manually using a stereomicroscope.
PRODUCT FEATURES
- Detect and quantitate individual cells secreting human IFN-gamma
- High sensitivity - ELISpot assays can measure responses with frequencies well below 1 in 100,000 cells
- No in vitro expansion of cells required
- High-throughput - ELISpot assays use only a small number of primary cells
KIT CONTENTS
- Human IFN-gamma Microplate
- Biotinylated Detection Antibody
- Streptavidin conjugated to Alkaline Phosphatase
- Dilution Buffers
- Wash Buffer Concentrate
- BCIP/NBT Chromogen
- Human IFN-gamma Positive Control
OTHER REAGENTS REQUIRED
- Pipettes and pipette tips
- Deionized or distilled water
- Squirt bottle, manifold dispenser, or automated microplate washer
- 500 mL graduated cylinder
- 37 °C CO2 incubator
- Sterile culture media
- Dissection microscope or an automated ELISpot reader
Formulation, Preparation, and Storage
Shipping
Storage
Background: IFN-gamma
Long Name
Alternate Names
Entrez Gene IDs
Gene Symbol
Additional IFN-gamma Products
Product Documents for Human IFN-gamma ELISpot Kit
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human IFN-gamma ELISpot Kit
For research use only
⚠ WARNING: This product can expose you to chemicals including methanol, which is known to the State of California to cause reproductive toxicity with developmental effects. For more information, go to www.P65Warnings.ca.gov.Related Research Areas
Citations for Human IFN-gamma ELISpot Kit
Customer Reviews for Human IFN-gamma ELISpot Kit (11)
Have you used Human IFN-gamma ELISpot Kit?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
-
Sample Tested: SerumVerified Customer | Posted 09/26/2022
-
Sample Tested: Human T cellsVerified Customer | Posted 08/20/2021
-
Sample Tested: Human T cellVerified Customer | Posted 03/26/2021
-
Sample Tested: Human CD8 cellsVerified Customer | Posted 02/09/2021
-
Sample Tested: Activated human CD8 T cellsVerified Customer | Posted 09/30/2020Works really well as compared to kits from other companies. Easy to follow protocol.
-
Sample Tested: Peripheral blood mononuclear cells (PBMCs)Verified Customer | Posted 07/15/2020The kit is easy to use and produced excellent results. Couple things that need to be added are: Information regarding storage conditions of the developed kit (spots); In my experience, the kit once developed is pretty stable and can be stored @ 4C in the dark for at least few weeks. The positive control for the kit produces a lawn rather than spots, something to keep in mind.
-
Sample Tested: Peripheral blood T cellsVerified Customer | Posted 07/10/2020
-
Sample Tested: Human cellsVerified Customer | Posted 07/10/2020
-
Sample Tested: Serum and PlasmaVerified Customer | Posted 01/25/2020
-
Sample Tested: Cell Culture SamplesVerified Customer | Posted 05/14/2018
-
Sample Tested: Human PBMCVerified Customer | Posted 03/22/2018
There are no reviews that match your criteria.
Associated Pathways
 Hematopoietic Stem Cell Differentiation Pathways & Lineage-specific Markers
Hematopoietic Stem Cell Differentiation Pathways & Lineage-specific Markers
 IL-15 Signaling Pathways and their Primary Biological Effects in Different Immune Cell Types
IL-15 Signaling Pathways and their Primary Biological Effects in Different Immune Cell Types
 IL-21 Signaling Pathways and their Primary Biological Effects in Different Immune Cell Types
IL-21 Signaling Pathways and their Primary Biological Effects in Different Immune Cell Types
 Innate Lymphoid Cell Differentiation Pathways
Innate Lymphoid Cell Differentiation Pathways