EEA1 (Early Endosome Autoantigen 1; also known as Endosome-associated protein p162 and Zn-finger FYVE domain-containing protein 2) is a 170-180 kDa protein that serves as an identifying marker for early endosomes. It is ubiquitously expressed, and found in both the cytosol and on cellular membranes. Its activity has been described as that of a tethering factor which links endosomes to endocytic vesicles, allowing for their fusion via a SNARE complex. Normally, EEA1 exists as a homodimer in the cytoplasm and appears to make transient contacts with endosome membrane phosphatidylinositol. When endosome fusion is not required, EEA1 serves as a substrate for p97, promoting EEA1 dissociation and endosome independence. When endosome fusion is required, EEA1 interacts with NSF, resulting in its removal from a large endosome-associated complex and subsequent endosomal vesicle fusion. Human EEA1 is synthesized as a 1411 amino acid (aa) protein that contains one C2H2-type Zn finger region (aa 41-64) and one FYVE Zn finger domain (aa 1352-1410). There is one isoform variant that contains a nine aa substitution for aa 925-1411. Over aa 1249-1356, human EEA1 shares 99% aa sequence identity with mouse EEA1.
Human/Mouse/Rat EEA1 Alexa Fluor® 488‑conjugated Antibody
R&D Systems | Catalog # IC8047G

Key Product Details
Species Reactivity
Validated:
Cited:
Applications
Validated:
Cited:
Label
Antibody Source
Product Specifications
Immunogen
Asn1249-Gln1356
Accession # Q15075
Specificity
Clonality
Host
Isotype
Scientific Data Images for Human/Mouse/Rat EEA1 Alexa Fluor® 488‑conjugated Antibody
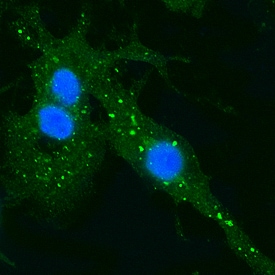
EEA1 in HeLa Human Cell Line.
EEA1 was detected in formaldehyde fixed HeLa human cervical epithelial carcinoma cell line using Sheep Anti-Human/Mouse/Rat EEA1 Alexa Fluor® 488‑conjugated Antigen Affinity-purified Polyclonal Antibody (Catalog # IC8047G) at 1:10 dilution overnight at 4 °C and counterstained with DAPI (blue). Specific staining was localized to endosomes. View our protocol for Fluorescent ICC Staining of Cells on Coverslips.Applications for Human/Mouse/Rat EEA1 Alexa Fluor® 488‑conjugated Antibody
Immunocytochemistry
Formulation, Preparation, and Storage
Purification
Formulation
Shipping
Stability & Storage
- 12 months from date of receipt, 2 to 8 °C as supplied.
Background: EEA1
Long Name
Alternate Names
Gene Symbol
UniProt
Additional EEA1 Products
Product Documents for Human/Mouse/Rat EEA1 Alexa Fluor® 488‑conjugated Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human/Mouse/Rat EEA1 Alexa Fluor® 488‑conjugated Antibody
This product is provided under an agreement between Life Technologies Corporation and R&D Systems, Inc, and the manufacture, use, sale or import of this product is subject to one or more US patents and corresponding non-US equivalents, owned by Life Technologies Corporation and its affiliates. The purchase of this product conveys to the buyer the non-transferable right to use the purchased amount of the product and components of the product only in research conducted by the buyer (whether the buyer is an academic or for-profit entity). The sale of this product is expressly conditioned on the buyer not using the product or its components (1) in manufacturing; (2) to provide a service, information, or data to an unaffiliated third party for payment; (3) for therapeutic, diagnostic or prophylactic purposes; (4) to resell, sell, or otherwise transfer this product or its components to any third party, or for any other commercial purpose. Life Technologies Corporation will not assert a claim against the buyer of the infringement of the above patents based on the manufacture, use or sale of a commercial product developed in research by the buyer in which this product or its components was employed, provided that neither this product nor any of its components was used in the manufacture of such product. For information on purchasing a license to this product for purposes other than research, contact Life Technologies Corporation, Cell Analysis Business Unit, Business Development, 29851 Willow Creek Road, Eugene, OR 97402, Tel: (541) 465-8300. Fax: (541) 335-0354.
For research use only
Citations for Human/Mouse/Rat EEA1 Alexa Fluor® 488‑conjugated Antibody
Customer Reviews
There are currently no reviews for this product. Be the first to review Human/Mouse/Rat EEA1 Alexa Fluor® 488‑conjugated Antibody and earn rewards!
Have you used Human/Mouse/Rat EEA1 Alexa Fluor® 488‑conjugated Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
