Nidogen-1 (also entactin) is a 150 kDa, secreted, monomeric glycoprotein that serves as a major linking component of basement membranes (1-4). It is synthesized as a 1247 amino acid (aa) precursor with a 28 aa signal sequence and a 1219 aa mature protein. The molecule is modular in structure with five distinct regions. There are three globular domains (G1-3) separated by a mucin region and an extended rod-shaped segment (5-7). The N-terminal globular domain (G1) is 200 aa in length and seemingly unrelated to any known motif (8). The mucin region is nearly 160 aa in length and presumably O-glycosylated (2, 8). G2 and G3 are both approximately 300 aa in length. G2 is described as a Nidogen ( beta -barrel) domain, while C-terminal G3 assumes a beta -propeller configuration (1). The 250 aa rod-shaped segment has multiple EGF-like motifs and two thyroglobulin type 1 domains. Functionally, G1 is reported to bind type IV collagen (2, 7). The mucin region contains a short peptide that ligates alpha 3 beta 1 integrins (9, 10). G2 interacts with perlecan, and an RGD motif in the rod-shaped segment serves as a binding site for alpha v beta 3 integrins (9, 10). Finally, G3 is associated with laminin binding (2, 7). As a full-length molecule, the multiple extracellular matrix-binding sites of Nidogen-1 are well positioned to serve as anchor sites for basement membrane molecules. Nidogen-1 also undergoes proteolytic processing by at least two MMPs, MMP-7 and MMP-19 (10, 11). While this destroys the integrity of Nidogen-associated matrices, it also generates peptide fragments that are capable of inducing neutrophil chemotaxis and phagocytosis (10). Nidogen-2 is related to Nidogen-1 (≈ 50% aa identity) and shares many of the same adhesive properties as Nidogen-1 (12). Both bind perlecan plus collagens I and IV. Nidogen‑2, however, does not bind fibulin-1 or 2, and shows only modest interaction with laminin. Thus, although coexpressed, Nidogen-2 serves as only a partial substitute for Nidogen-1 (2, 12). Human Nidogen-1 shares 85% aa sequence identity with both mouse and rat Nidogen-1, and 88% aa sequence identity with canine Nidogen-1.
Human Nidogen‑1/Entactin Antibody
R&D Systems | Catalog # AF2570


Key Product Details
Species Reactivity
Validated:
Human
Cited:
Human, Mouse, Rabbit
Applications
Validated:
Immunohistochemistry, Western Blot
Cited:
Immunohistochemistry, Immunohistochemistry-Frozen, Western Blot, Immunocytochemistry
Label
Unconjugated
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
Mouse myeloma cell line NS0-derived recombinant human Nidogen‑1/Entactin
Leu29-Lys1114 (Gln1113Arg)
Accession # AAH45606
Leu29-Lys1114 (Gln1113Arg)
Accession # AAH45606
Specificity
Detects human Nidogen‑1/Entactin in direct ELISAs and Western blots.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Scientific Data Images for Human Nidogen‑1/Entactin Antibody
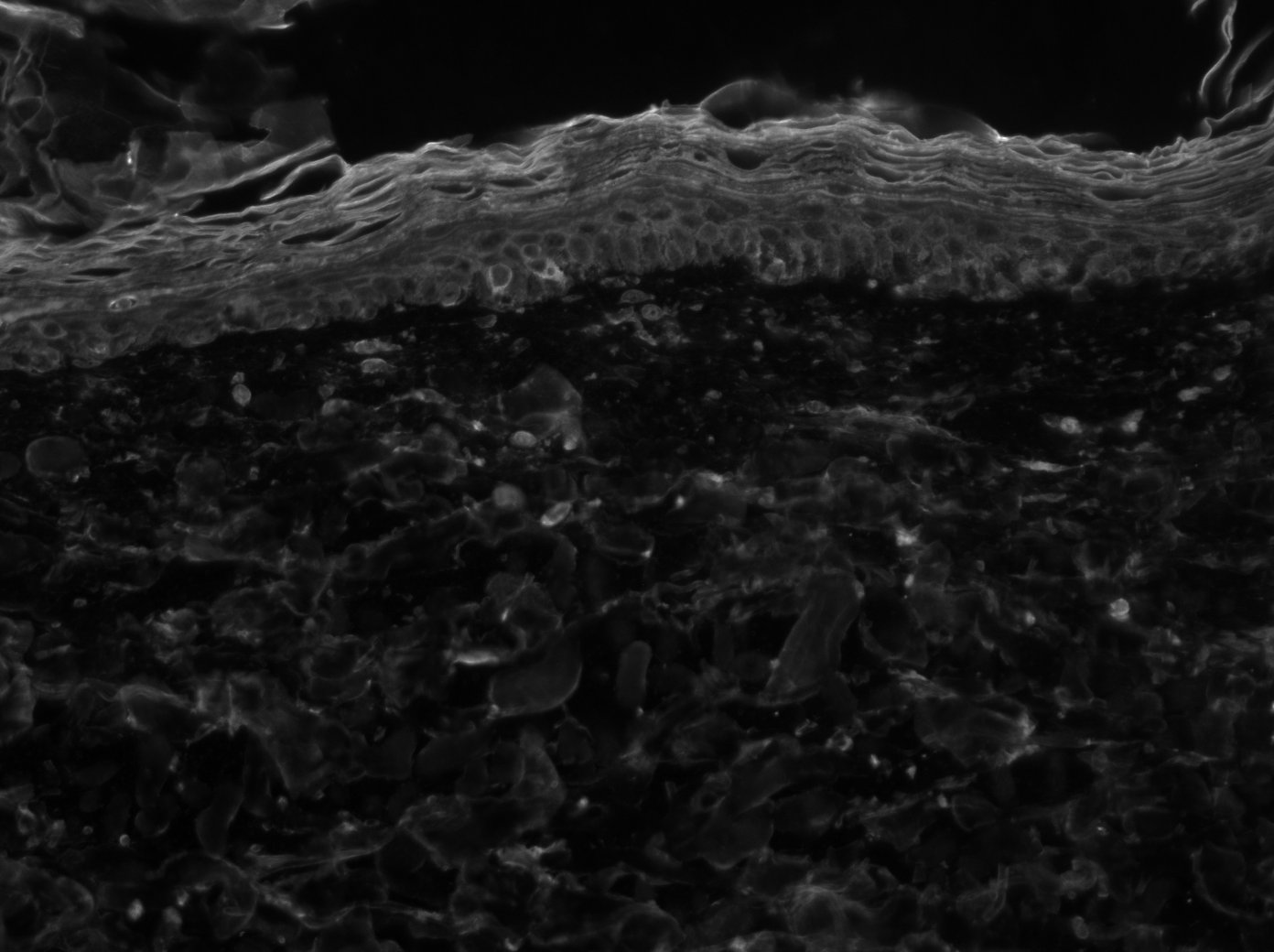
Nidogen‑1/Entactin in Human Heart.
Nidogen‑1/Entactin was detected in immersion fixed paraffin-embedded sections of human heart using Goat Anti-Human Nidogen‑1/Entactin Antigen Affinity-purified Polyclonal Antibody (Catalog # AF2570) at 15 µg/mL overnight at 4 °C. Tissue was stained using the Anti-Goat HRP-DAB Cell & Tissue Staining Kit (brown; Catalog # CTS008) and counterstained with hematoxylin (blue). Specific labeling was localized to the sarcolemma of cardiomyocytes. View our protocol for Chromogenic IHC Staining of Paraffin-embedded Tissue Sections.Detection of Human Nidogen-1/Entactin by Western Blot
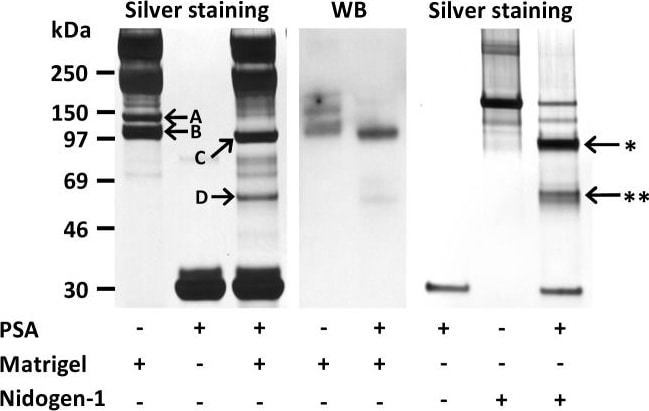
Degradation of nidogen-1 by PSA.PSA cleaved both mouse nidogen-1 in Matrigel and human recombinant nidogen-1. Mass spectrometry analysis identified nidogen-1 in the silver stained gel bands (arrows, left panel). Nidogen-1 bands of 140 kDa (A) and 110 kDa (B) disappeared and fragments of 90 kDa (C) and 55 kDa (D) appeared after 48 h incubation of diluted Matrigel with PSA at 37°C. Nidogen-1 cleavage by PSA in Matrigel was visualized by Western blotting with anti-nidogen-1 polyclonal antibody (middle panel). PSA (1 µM) cleaved recombinant human nidogen-1 (0.5 µM) into two fragments 85 kDa (arrow with *) and 55 kDa (arrow with **) during 20 h incubation at 37°C (right panel). Image collected and cropped by CiteAb from the following publication (https://dx.plos.org/10.1371/journal.pone.0107819), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human Nidogen-1/Entactin by Western Blot
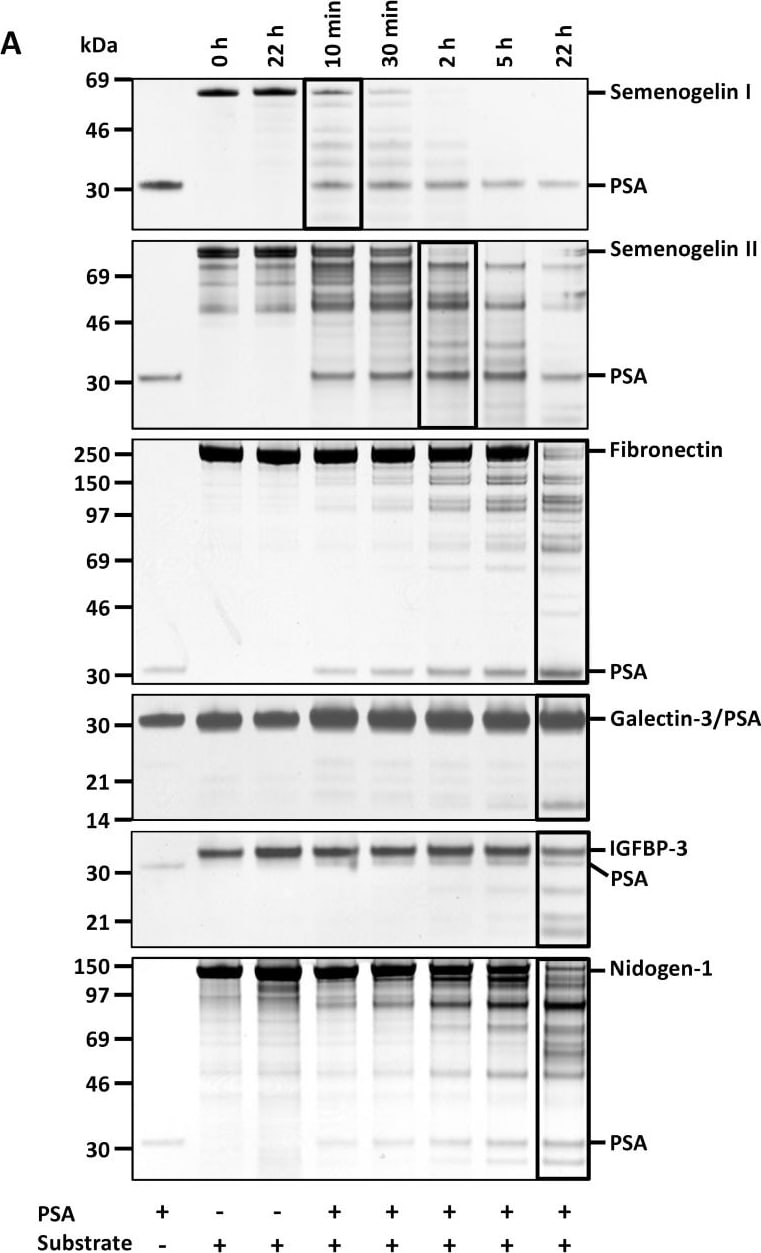
Cleavage of protein substrates by PSA.(A) Characterization of the proteolytic activity of PSA (0.2 µM) during 22 h incubation towards different protein substrates (1 µM each, except 0.5 µM semenogelin I) by SDS-PAGE and silver staining. Approximate molecular weights of the proteins are: PSA (28 kDa), semenogelin I (50 kDa), semenogelin II (63 kDa), fibronectin (220 kDa), galectin-3 (26 kDa), IGFBP-3 (30 kDa) and nidogen-1 (130 kDa). The lanes in which ∼50% of the proteins were cleaved are bordered. (B) 1 µM MMP-3 (22 kDa), but not PSA, cleaved 1 µM plasminogen (88 kDa). Also 0.5 M fibronectin was incubated with 1 µM PSA as a control (SDS-PAGE with silver staining). Image collected and cropped by CiteAb from the following publication (https://dx.plos.org/10.1371/journal.pone.0107819), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human Nidogen‑1/Entactin Antibody
Application
Recommended Usage
Immunohistochemistry
5-15 µg/mL
Sample: Immersion fixed paraffin-embedded sections of human heart
Sample: Immersion fixed paraffin-embedded sections of human heart
Western Blot
0.1 µg/mL
Sample: Recombinant Human Nidogen‑1/Entactin (Catalog # 2570-ND)
Sample: Recombinant Human Nidogen‑1/Entactin (Catalog # 2570-ND)
Reviewed Applications
Read 2 reviews rated 3.5 using AF2570 in the following applications:
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: Nidogen-1/Entactin
References
- Hohenester, E. and J. Engel (2002) Matrix Biol. 21:115.
- Miosge, N. et al. (2001) Histochem. J. 33:523.
- Charonis, A. et al. (2005) Curr. Med. Chem. 12:1495.
- Timpl, R. and J.C. Brown (1996) BioEssays 18:123.
- Nagayoshi, T. et al. (1989) DNA 8:581.
- Zimmerman, K. et al. (1995) Genomics 27:245.
- Fox, J.W. et al. (1991) EMBO J. 10:3137.
- Mayer, U. et al. (1995) Eur. J. Biochem. 227:681.
- Gresham, H.D. et al. (1996) J. Biol. Chem. 271:30587.
- Dong, L-J. et al. (1995) J. Biol. Chem. 270:15383.
- Titz, B. et al. (2004) Cell. Mol. Life Sci. 61:1826.
- Kohfeldt, K. et al. (1998) J. Mol. Biol. 282:99.
Alternate Names
Entactin-1, NID1, Nidogen1
Entrez Gene IDs
4811 (Human)
Gene Symbol
NID1
UniProt
Additional Nidogen-1/Entactin Products
Product Documents for Human Nidogen‑1/Entactin Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human Nidogen‑1/Entactin Antibody
For research use only
Citations for Human Nidogen‑1/Entactin Antibody
Customer Reviews for Human Nidogen‑1/Entactin Antibody (2)
3.5 out of 5
2 Customer Ratings
Have you used Human Nidogen‑1/Entactin Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
Showing
1
-
2 of
2 reviews
Showing All
Filter By:
-
Application: Immunohistochemistry-ParaffinSample Tested: human skinSpecies: HumanVerified Customer | Posted 04/11/2019
-
Application: Immunohistochemistry-ParaffinSample Tested: See PMID 23680364Species: HumanVerified Customer | Posted 01/07/2015
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...