Vascular Endothelial (VE)-Cadherin (VE-CAD), also called 7B4 and Cadherin‑5 (CDH5), is a member of the cadherin family of cell adhesion molecules. Cadherins are calcium-dependent transmembrane proteins which bind to one another in a homophilic manner. On their cytoplasmic side, they associate with the three catenins, alpha, beta, and gamma (plakoglobin). This association links the cadherin protein to the cytoskeleton. Without association with the catenins, the cadherins are non-adhesive. Cadherins play a role in development, specifically in tissue formation. They may also help to maintain tissue architecture in the adult. VE-Cadherin has been shown to play important roles in vasculogenesis and angiogenesis. VE-Cadherin is a classical cadherin molecule. Classical cadherins consist of a large extracellular domain which contains DXD and DXNDN repeats responsible for mediating calcium-dependent adhesion, a single-pass transmembrane domain, and a short carboxy-terminal cytoplasmic domain responsible for interacting with the catenins. Human VE-Cadherin is a 784 amino acid (aa) residue protein with a 25 aa signal sequence and a 759 aa propeptide. The mature protein begins at amino acid 48 and has a 546 aa extracellular domain, a 27 aa transmembrane domain, and a 164 aa cytoplasmic domain. The human and mouse mature VE-Cadherin proteins share approximately 74% homology.
Human VE‑Cadherin APC‑conjugated Antibody
R&D Systems | Catalog # FAB9381A

Key Product Details
Species Reactivity
Validated:
Cited:
Applications
Validated:
Cited:
Label
Antibody Source
Product Specifications
Immunogen
Asp48-Gln593
Accession # P33151
Specificity
Clonality
Host
Isotype
Scientific Data Images for Human VE‑Cadherin APC‑conjugated Antibody
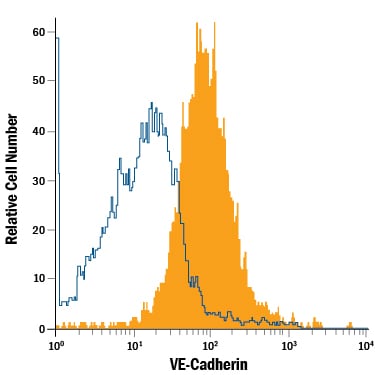
Detection of VE‑Cadherin in HUVEC Human Cells by Flow Cytometry.
HUVEC human umbilical vein endothelial cells were stained with Mouse Anti-Human VE-Cadherin APC-conjugated Monoclonal Antibody (Catalog # FAB9381A, filled histogram) or isotype control antibody (Catalog # IC0041A, open histogram). Cells were stained in a buffer containing Ca2+and Mg2+. View our protocol for Staining Membrane-associated Proteins.Applications for Human VE‑Cadherin APC‑conjugated Antibody
Flow Cytometry
Sample: HUVEC human umbilical vein endothelial cells stained in a buffer containing Ca2+ and Mg2+.
Spectra Viewer
Plan Your Experiments
Use our spectra viewer to interactively plan your experiments, assessing multiplexing options. View the excitation and emission spectra for our fluorescent dye range and other commonly used dyes.
Spectra ViewerFlow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Formulation
Shipping
Stability & Storage
- 12 months from date of receipt, 2 to 8 °C as supplied.
Background: VE-Cadherin
References
- Shimoyama, Y. et al. (1989) J. Cell Biol. 109:1787.
- Bussemakers, M.J.G. et al. (1993) Mol. Biol. Reports 17:123.
- Overduin, M. et al. (1995) Science 267:386.
- Takeichi, M. (1991) Science 251:1451.
- Nose, A. et al. (1987) EMBO J. 6:3655.
- Carmeliet, P. et al. (1999) Cell 98:147.
- Gory-Faure, S. et al. (1999) Development 126:2093.
Long Name
Alternate Names
Gene Symbol
UniProt
Additional VE-Cadherin Products
Product Documents for Human VE‑Cadherin APC‑conjugated Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human VE‑Cadherin APC‑conjugated Antibody
For research use only
Citations for Human VE‑Cadherin APC‑conjugated Antibody
Customer Reviews for Human VE‑Cadherin APC‑conjugated Antibody
There are currently no reviews for this product. Be the first to review Human VE‑Cadherin APC‑conjugated Antibody and earn rewards!
Have you used Human VE‑Cadherin APC‑conjugated Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Liperfluo
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
FAQs for Human VE‑Cadherin APC‑conjugated Antibody
-
Q: Why does the staining protocol with this Cadherin antibody use buffers containing Ca2+ and Mg2+?
A: The staining protocol with this and other Cadherin antibodies uses buffer containing Ca2+ and Mg2+ because Cadherin function is Calcium-dependent.
Associated Pathways
 Blood-Brain Barrier and Immune Cell Transmigration: ICAM-1/CD54 Signaling Pathways
Blood-Brain Barrier and Immune Cell Transmigration: ICAM-1/CD54 Signaling Pathways
 Blood-Brain Barrier and Immune Cell Transmigration: Pathways Overview
Blood-Brain Barrier and Immune Cell Transmigration: Pathways Overview
 Blood-Brain Barrier and Immune Cell Transmigration: VCAM-1/CD106 Signaling Pathways
Blood-Brain Barrier and Immune Cell Transmigration: VCAM-1/CD106 Signaling Pathways
 Blood-Brain Barrier and Immune Cell Transmigration: VEGF Signaling Pathways
Blood-Brain Barrier and Immune Cell Transmigration: VEGF Signaling Pathways

 Mesenchymal Stem Cell Differentiation Pathways & Lineage-specific Markers
Mesenchymal Stem Cell Differentiation Pathways & Lineage-specific Markers
 VEGF - VEGF R2 Signaling Pathways
VEGF - VEGF R2 Signaling Pathways