LAMP-1/CD107a Antibody - BSA Free
Novus Biologicals | Catalog # NB120-19294

![Western Blot: LAMP-1/CD107a Antibody [NB120-19294] Western Blot: LAMP-1/CD107a Antibody [NB120-19294]](https://resources.rndsystems.com/images/products/LAMP-1-CD107a-Antibody-Western-Blot-NB120-19294-img0005.jpg)
Key Product Details
Species Reactivity
Validated:
Human, Mouse, Rat
Cited:
Human, Mouse, Rat, Rabbit
Applications
Validated:
Immunohistochemistry, Immunohistochemistry-Paraffin, Western Blot, Immunocytochemistry/ Immunofluorescence
Cited:
Immunohistochemistry-Paraffin, Western Blot, Immunocytochemistry/ Immunofluorescence, IF/IHC
Label
Unconjugated
Antibody Source
Polyclonal Rabbit IgG
Format
BSA Free
Loading...
Product Specifications
Immunogen
Synthetic peptide corresponding to residues C K(407) R S H A G Y Q T I(416) of human LAMP1.
Reactivity Notes
Mouse reactivity reported in scientific literature (PMID: 22453828 ). Rat reactivity reported in scientific literature (PMID: 22544351).
Localization
Type I membrane protein. Lysosomal. This protein shuttles between lysosomes, endosomes, and the plasma membrane.
Marker
Late Endosome Marker
Specificity
LAMP1 - Lysosome Marker
Clonality
Polyclonal
Host
Rabbit
Isotype
IgG
Scientific Data Images for LAMP-1/CD107a Antibody - BSA Free
Western Blot: LAMP-1/CD107a Antibody [NB120-19294]
Western Blot: LAMP-1/CD107a Antibody [NB120-19294] - Analysis of 20 ug of HeLa cell lysate and 5 ul of Molecular Weight Protein Ladder per well.Immunocytochemistry/ Immunofluorescence: LAMP-1/CD107a Antibody [NB120-19294]
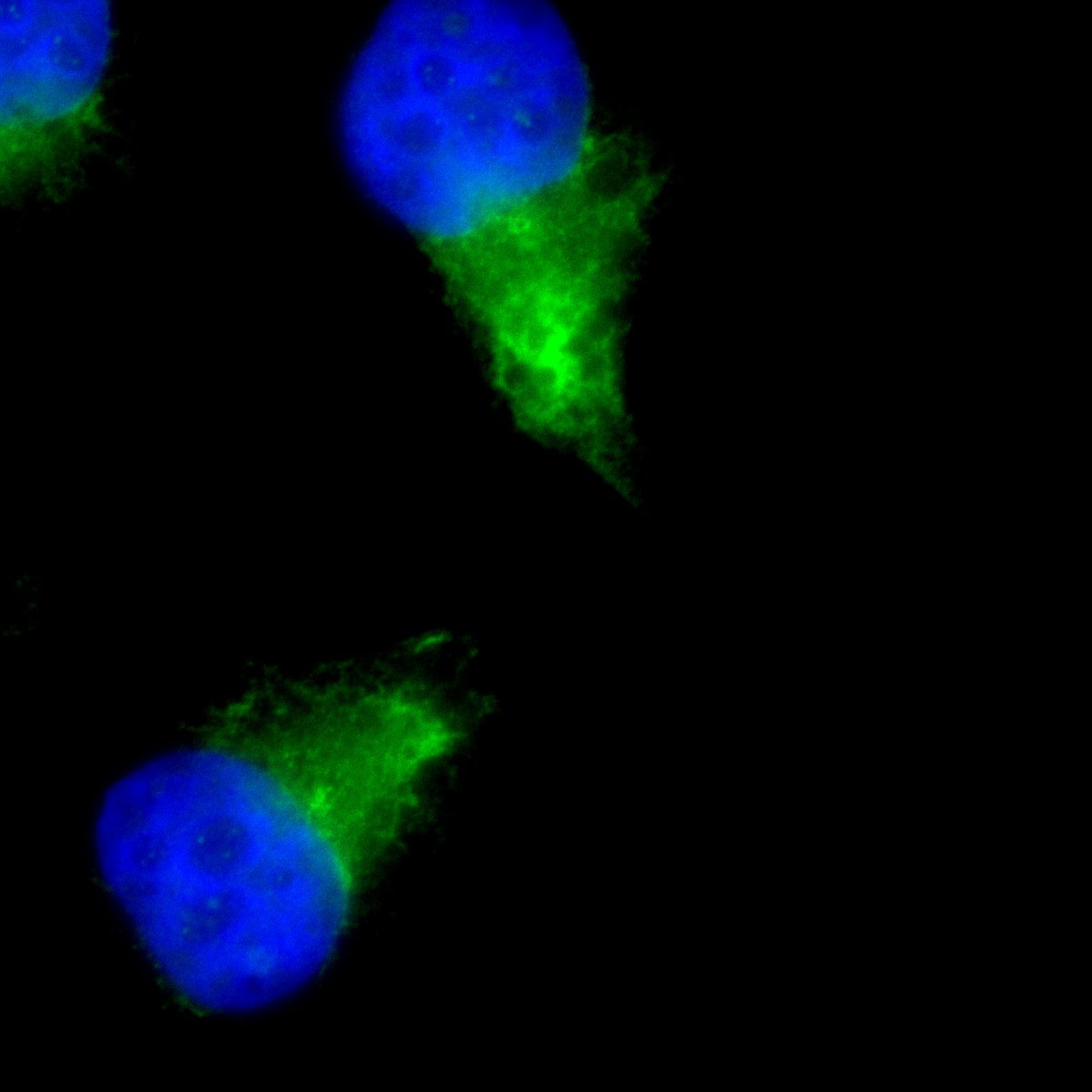
LAMP-1-CD107a-Antibody-Immunocytochemistry-Immunofluorescence-NB120-19294-img0006.jpgImmunocytochemistry/ Immunofluorescence: LAMP-1/CD107a Antibody [NB120-19294]
Immunocytochemistry/Immunofluorescence: LAMP-1/CD107a Antibody [NB120-19294] - Lamp1 (red) in HeLa cells.Immunocytochemistry/ Immunofluorescence: LAMP-1/CD107a Antibody [NB120-19294]
LAMP-1-CD107a-Antibody-Immunocytochemistry-Immunofluorescence-NB120-19294-img0007.jpgImmunocytochemistry/ Immunofluorescence: LAMP-1/CD107a Antibody [NB120-19294] -
Autophagy associated protein immunoreactivity in HIV-infected brain tissue. (A) Representative images from five randomly selected fields of cells each examined in duplicate frontal lobe white matter sections for the indicated subject groups. The indicated proteins were labeled red & microglia with the cell-type-specific marker Iba1 (green). Blue staining indicates cell nuclei. Arrow heads indicate examples of higher Iba1 immunoreactivity whereas arrows indicate more focal (punctal) vs. diffuse (filamentous) patterns of autophagy associated protein expression. Scale bar = 10 μm. (B) Quantification of relative Iba1 immunoreactivity from (A). F(3,20) = 6.450, p = 0.0031; ∗p < 0.05 when compared to all other subject groups. Error bars show the SEM for the average values of 2–6 regions from each subject group across the six autophagy associated proteins examined. (C) Quantification of the indicated autophagy associated protein relative immunoreactivity from (A). Beclin 1: F(3,12) = 11.29, p = 0.0008; LC3B: F(3,12) = 1.994, p = 0.1687; APG7/ATG7: F(3,12) = 84.20, p = < 0.0001; ATG5: F(3,12) = 6.218, p = 0.0086; p62/SQSTM1: F(3,12) = 87.04, p = < 0.0001; LAMP1: F(3,12) = 8.317, p = 0.0029. ∗p < 0.05 when compared to HIV-negative; #p < 0.05 when compared to HIV-positive; & Ωp < 0.05 when compared to HIV-positive/NCI subjects. Error bars show the SEM for four regions from each subject group. Image collected & cropped by CiteAb from the following publication (http://journal.frontiersin.org/Article/10.3389/fmicb.2015.00653/abstract), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: LAMP-1/CD107a Antibody [NB120-19294] -
Differences in neuronal autophagy & dendrite varicosity following HIV-1 Tat protein & morphine treatment. (A) Representative images of neurons transfected with a fluorescent reporter plasmid to monitor autophagic flux at 8 h following the indicated treatments. GFP (green) & GFP + mRFP (yellow) fluorescence are observed prior to the fusion of autophagosomes with lysosomes whereas only mRFP (red) fluorescence is present in post-fusion autolysosomes. DIC, differential interference contrast microscopy image. DAPI (blue) staining indicates cell nuclei. (B) Quantification of autolysosomes (red puncta) from (A). F(3,13) = 8.756, p = 0.0019; ∗p < 0.05 when compared to all other groups. (C) Western blotting analysis of the indicated autophagy associated protein levels at 24 h following the indicated treatments. GAPDH was used as a loading control. Blots are representative of three independent experiments. (D) Quantification of dendrite beading from (A). F(3,77) = 6.429, p = 0.0006; ∗p < 0.05 when compared to control cells. Error bars show the SEM. Image collected & cropped by CiteAb from the following publication (http://journal.frontiersin.org/Article/10.3389/fmicb.2015.00653/abstract), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunocytochemistry/ Immunofluorescence: LAMP-1/CD107a Antibody [NB120-19294] -
Immunocytochemistry/ Immunofluorescence: LAMP-1/CD107a Antibody [NB120-19294] - EsxG-EsxH alters phagosomal GAL3, ubiquitin, & LAMP1. (A, C, & E) IF images of GAL3 (A), ubiquitin (FK2 antibody) (C), & LAMP1 (E) in BMDMs that were infected with DsRed-expressing H37Rv (WT) or the delta esxH mutant for 3 h. Images are maximum-intensity projections. Scale bar, 10 μm. Boxed areas in the merged image are shown in higher magnification in the rightmost panel. Mtb, M. tuberculosis. (B, D, & F) Automated image analysis was used to quantify the MFI of GAL3 (B), ubiquitin (D), & LAMP1 (F) colocalized with individual bacilli from 5 fields of a 12-mm coverslip. Data are means ± SEM from one representative experiment from three (A, B, E, & F) or two (C & D) independent experiments. ****, P ≤ 0.0001, Student's t test. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/30482832), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunocytochemistry/ Immunofluorescence: LAMP-1/CD107a Antibody [NB120-19294] -
Immunocytochemistry/ Immunofluorescence: LAMP-1/CD107a Antibody [NB120-19294] - Characterization of selected mutant protein glycosylation status or localization.(A) Two independent, representative experiments to analyze the glycosylation status of GFP-NPC1 wild type & GFP-NPC1 P251C/L929C, as determined by immunoblot of indicated samples with anti-GFP antibodies after 6% SDS PAGE; samples were incubated at 70°C in SDS PAGE sample buffer for 10 min prior to loading. Molecular weight markers are shown at left in kilodaltons here & in all subsequent gels shown. (B), Localization of NPC1 P251/L929C/P249K/P259K in NPC1−/− HeLa cells as in Figure 1C. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/32410728), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunocytochemistry/ Immunofluorescence: LAMP-1/CD107a Antibody [NB120-19294] -
Immunocytochemistry/ Immunofluorescence: LAMP-1/CD107a Antibody [NB120-19294] - NPC1 delta loop mutant cannot rescue cholesterol export from lysosomes.(A) Cholesterol-cross-linked peptides (Hulce et al., 2013) are highlighted in red for two orientations of the crystal structure of N-terminal domain- & first transmembrane domain-deleted NPC1 (PDBID: 5u74). The disordered cytoplasmic loop residues 800–814 are shown as a blue dotted line. (B) Confocal immunofluorescence microscopy analysis of the localization of mouse NPC1-delta 807–811, NPC1-807-811Ala & LAMP1 proteins in HeLa cells (bar, 20 μm). White boxes in images indicate regions of cells enlarged in the insets shown at the lower right of each image. (C) Confocal immunofluorescence microscopy of cholesterol accumulation rescue. NPC1−/− HeLa cells were transfected with GFP-tagged mouse NPC1-delta 807–811 or mouse NPC1-807-811Ala plasmids for 48 h & assayed for cholesterol accumulation rescue as in Figure 1 (bar, 20 μm). (D) Quantitation of cholesterol accumulation rescue using flow cytometry. GFP-positive cells with similar expression levels were analyzed: 2480 NPC1; 427 NPC1-delta 807–811; 764 NPC1-807-811Ala; LAMP1 expressing control, 1753 cells counted. Shown are the normalized data from mean fluorescence intensity flow cytometry values. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/32410728), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunocytochemistry/ Immunofluorescence: LAMP-1/CD107a Antibody [NB120-19294] -
Immunocytochemistry/ Immunofluorescence: LAMP-1/CD107a Antibody [NB120-19294] - Cathepsin D (CathD) & B (CathB) expression & localization in primary neurons. A. Western blot analyses of CathD & CathB protein levels after transfection. Primary cortical neurons were transfected with 23QHtt, 23QHtt plus CathD, 23QHtt plus CathB, 145QmHtt, 145QmHtt plus CathD & 145QmHtt plus CathB constructs. Western blot analyses were performed with anti-CathD & anti-CathB antibodies. beta -actin western blots were used as loading controls. Relative expression levels were quantified by band intensity. Positions of molecular weight markers were indicated. Quantification of the western blots are shown in the bar graphs. *p < 0.05 compared to without cathepsin transfection. B. Analyses of CathD & CathB enzymatic activities after transfection. Primary cortical neurons were transfected with CathD, CathB & as described in A. CathD & CathB activities were assayed by CathD or CathB activity assay kit. *p < 0.05 compared to without cathepsin transfection. C & D. Colocalization of transfected CathD (C) & CathB (D) with LAMP-1. CathD & CathB transfected neurons were examined by co-immunostaining of LAMP-1 & CathD or CathB. Yellow colored cytoplasmic spots are indicative of co-localization of transfected cathepsins & LAMP-1. Scale bar = 10 micron. Image collected & cropped by CiteAb from the following publication (https://molecularneurodegeneration.biomedcentral.com/articles/10.1186/1…), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: LAMP-1/CD107a Antibody [NB120-19294] -
Western Blot: LAMP-1/CD107a Antibody [NB120-19294] - Effects on autophagic activity & dendritic length of neurons exposed to supernatant from HIV-1-infected microglia in combination with morphine. (A) Representative images of neurons with the indicated treatments. Sup, supernatant from uninfected [HIV(-)] & HIV-1-infected [HIV(+)] microglia. Cells were immunolabeled with antibodies to the autophagic activity marker p62/SQSTM1 (red) & the neuronal cell-type-specific marker MAP2 (green). DAPI (blue) staining indicates cell nuclei. (B) Quantification of p62/SQSTM1 immunoreactivity from (A). Data are presented as the percentage of control cells which was set at 100; F(5,24) = 5.882, p = 0.0011; ∗p < 0.05 when compared to HIV(+)Sup + morphine treatment. (C) Western blotting analysis of p62/SQSTM1 & LAMP1 expression levels for the indicated treatments. GAPDH was used as a loading control. Blots are representative of three independent experiments. (D) Measurement of dendrite length from (A). F(5,24) = 26.15, p = < 0.0001; Φp < 0.05 when compared to morphine; Ψp < 0.05 when compared to HIV(-)Sup; Ωp < 0.05 when compared to HIV(-)Sup + morphine; #p < 0.05 when compared to HIV(+)Sup; & ∗p < 0.05 when compared to HIV(+)Sup + morphine treatment. Error bars show the SEM for five randomly selected fields totaling at least 100 cells from each group. Image collected & cropped by CiteAb from the following publication (http://journal.frontiersin.org/Article/10.3389/fmicb.2015.00653/abstract), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunocytochemistry/ Immunofluorescence: LAMP-1/CD107a Antibody [NB120-19294] -
Immunocytochemistry/ Immunofluorescence: LAMP-1/CD107a Antibody [NB120-19294] - NPC1 Disulfide bond-locked MLD & CTD fails to rescue cholesterol export from lysosomes.(A) Partial NPC1 structure; inset, close-up view of the MLD/CTD interface. The amino acid residues mutated to cysteines for disulfide bond formation are shown & highlighted in red. (B) Confocal immunofluorescence microscopy analysis of mouse NPC1-A521C/K1013C & LAMP1 proteins in HeLa cells (bar, 20 μm). White boxes in images indicate regions of cells enlarged in the insets shown at the lower right of each image. (C) Confocal immunofluorescence microscopy of cholesterol accumulation rescue for NPC1-A521C or mouse NPC1-A521C/K1013C. (D) Flow cytometry of the rescue experiment analyzed in (C). GFP-positive cells with similar expression levels were analyzed: 17746 NPC1-/- cells; 1315 NPC1 wild type; 1137 NPC1-A521C/K1013C cells; 837 NPC1-A521C cells; cell numbers were normalized for comparison.Extracted ion chromatograms from LC-MS analysis of proteolyzed A521C/K1013C NPC1.Protein was carbamidomethylated in the presence or absence of reducing agent prior to deglycosylation & proteolysis. In both samples, blue traces represent m/z = 596.2818 (corresponding to the NPC1 peptide APCSLNDTSLL carbamidomethylated at the engineered cysteine A521C & deamidated at the N524 glycosylation site). Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/32410728), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Applications for LAMP-1/CD107a Antibody - BSA Free
Application
Recommended Usage
Immunocytochemistry/ Immunofluorescence
1:10 - 1:500
Immunohistochemistry
1:10 - 1:500
Immunohistochemistry-Paraffin
1:10 - 1:500
Western Blot
1:100 - 1:2000
Application Notes
ICC usage was reported and validated in scientific literature. Reactivity for this application may vary lot to lot.
Reviewed Applications
Read 2 reviews rated 3.5 using NB120-19294 in the following applications:
Formulation, Preparation, and Storage
Purification
Immunogen affinity purified
Formulation
0.02M tris (pH 7.4) and 0.1M glycine
Format
BSA Free
Preservative
0.1% Sodium Azide
Concentration
1 mg/ml
Shipping
The product is shipped with polar packs. Upon receipt, store it immediately at the temperature recommended below.
Stability & Storage
Store at -20C. Avoid freeze-thaw cycles.
Background: LAMP-1/CD107a
LAMP-1 plays an important role in autophagy-mediated ATP-release during apoptosis where lysosomes containing intracellular ATP migrate to the plasma membrane and, during exocytosis, LAMP-1 is exposed to the cell surface (5). Studies have found that knockdown of LAMP-1 blocks the ATP release from the cell (5). Furthermore, an absence of LAMP-1 and LAMP-2 leads to an accumulation of lysosomal cholesterol (6). Lysosomal membrane dysfunction or defects has also been associated with disease development (6,7). For example, one feature of pancreatitis is autophagy impairment which is caused by lysosomal dysfunction and a corresponding decrease in lysosomal-membrane associated proteins LAMP-1 and LAMP-2 (7).
References
1. Eskelinen E. L. (2006). Roles of LAMP-1 and LAMP-2 in lysosome biogenesis and autophagy. Molecular aspects of medicine, 27(5-6), 495-502. https://doi.org/10.1016/j.mam.2006.08.005
2. Cheng, X. T., Xie, Y. X., Zhou, B., Huang, N., Farfel-Becker, T., & Sheng, Z. H. (2018). Revisiting LAMP1 as a marker for degradative autophagy-lysosomal organelles in the nervous system. Autophagy, 14(8), 1472-1474. https://doi.org/10.1080/15548627.2018.1482147
3. Krzewski, K., & Coligan, J. E. (2012). Human NK cell lytic granules and regulation of their exocytosis. Frontiers in immunology, 3, 335. https://doi.org/10.3389/fimmu.2012.00335
4. Uniprot (P11279)
5. Wang, Y., Martins, I., Ma, Y., Kepp, O., Galluzzi, L., & Kroemer, G. (2013). Autophagy-dependent ATP release from dying cells via lysosomal exocytosis. Autophagy, 9(10), 1624-1625. https://doi.org/10.4161/auto.25873
6. Schwake, M., Schr0der, B., & Saftig, P. (2013). Lysosomal membrane proteins and their central role in physiology. Traffic (Copenhagen, Denmark), 14(7), 739-748. https://doi.org/10.1111/tra.12056
7. Gukovsky, I., Pandol, S. J., Mareninova, O. A., Shalbueva, N., Jia, W., & Gukovskaya, A. S. (2012). Impaired autophagy and organellar dysfunction in pancreatitis. Journal of gastroenterology and hepatology, 27 Suppl 2(Suppl 2), 27-32. https://doi.org/10.1111/j.1440-1746.2011.07004.x
Long Name
Lysosome-associated Membrane Glycoprotein 1
Alternate Names
CD107a, LAMP1
Gene Symbol
LAMP1
UniProt
Additional LAMP-1/CD107a Products
Product Documents for LAMP-1/CD107a Antibody - BSA Free
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Product Specific Notices for LAMP-1/CD107a Antibody - BSA Free
This product is for research use only and is not approved for use in humans or in clinical diagnosis. Primary Antibodies are guaranteed for 1 year from date of receipt.
Citations for LAMP-1/CD107a Antibody - BSA Free
Customer Reviews for LAMP-1/CD107a Antibody - BSA Free (2)
3.5 out of 5
2 Customer Ratings
Have you used LAMP-1/CD107a Antibody - BSA Free?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
Showing
1
-
2 of
2 reviews
Showing All
Filter By:
-
Application: ImmunocytochemistrySample Tested: MDA-MB-231Species: HumanVerified Customer | Posted 12/15/2017green lamp1 1/100 DAPI
-
Application: immunofluroscent stainingSample Tested: Primary mouse hepatocytesSpecies: MouseVerified Customer | Posted 12/08/2017white-nuclei; green-iNOS; red-prohibition; blue-LAMP1
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...
Associated Pathways


![Immunocytochemistry/ Immunofluorescence: LAMP-1/CD107a Antibody [NB120-19294] Immunocytochemistry/ Immunofluorescence: LAMP-1/CD107a Antibody [NB120-19294]](https://resources.rndsystems.com/images/products/LAMP-1-CD107a-Antibody-Immunocytochemistry-Immunofluorescence-NB120-19294-img0006.jpg)
![Immunocytochemistry/ Immunofluorescence: LAMP-1/CD107a Antibody [NB120-19294] Immunocytochemistry/ Immunofluorescence: LAMP-1/CD107a Antibody [NB120-19294]](https://resources.rndsystems.com/images/products/LAMP-1-CD107a-Antibody-Immunocytochemistry-Immunofluorescence-NB120-19294-img0003.jpg)
![Immunocytochemistry/ Immunofluorescence: LAMP-1/CD107a Antibody [NB120-19294] Immunocytochemistry/ Immunofluorescence: LAMP-1/CD107a Antibody [NB120-19294]](https://resources.rndsystems.com/images/products/LAMP-1-CD107a-Antibody-Immunocytochemistry-Immunofluorescence-NB120-19294-img0007.jpg)
![Immunocytochemistry/ Immunofluorescence: LAMP-1/CD107a Antibody [NB120-19294] - LAMP-1/CD107a Antibody](https://resources.rndsystems.com/images/products/nb120-19294_rabbit-polyclonal-lamp-1-cd107a-antibody-210202423452423.jpg)
![Western Blot: LAMP-1/CD107a Antibody [NB120-19294] - LAMP-1/CD107a Antibody](https://resources.rndsystems.com/images/products/nb120-19294_rabbit-polyclonal-lamp-1-cd107a-antibody-210202423454885.jpg)
![Immunocytochemistry/ Immunofluorescence: LAMP-1/CD107a Antibody [NB120-19294] - LAMP-1/CD107a Antibody](https://resources.rndsystems.com/images/products/nb120-19294_rabbit-polyclonal-lamp-1-cd107a-antibody-310202415363712.jpg)
![Immunocytochemistry/ Immunofluorescence: LAMP-1/CD107a Antibody [NB120-19294] - LAMP-1/CD107a Antibody](https://resources.rndsystems.com/images/products/nb120-19294_rabbit-polyclonal-lamp-1-cd107a-antibody-310202415382428.jpg)
![Immunocytochemistry/ Immunofluorescence: LAMP-1/CD107a Antibody [NB120-19294] - LAMP-1/CD107a Antibody](https://resources.rndsystems.com/images/products/nb120-19294_rabbit-polyclonal-lamp-1-cd107a-antibody-310202416205170.jpg)
![Immunocytochemistry/ Immunofluorescence: LAMP-1/CD107a Antibody [NB120-19294] - LAMP-1/CD107a Antibody](https://resources.rndsystems.com/images/products/nb120-19294_rabbit-polyclonal-lamp-1-cd107a-antibody-31020241621824.jpg)
![Western Blot: LAMP-1/CD107a Antibody [NB120-19294] - LAMP-1/CD107a Antibody](https://resources.rndsystems.com/images/products/nb120-19294_rabbit-polyclonal-lamp-1-cd107a-antibody-31020241621816.jpg)
![Immunocytochemistry/ Immunofluorescence: LAMP-1/CD107a Antibody [NB120-19294] - LAMP-1/CD107a Antibody](https://resources.rndsystems.com/images/products/nb120-19294_rabbit-polyclonal-lamp-1-cd107a-antibody-31020241621838.jpg)