Key Product Details
Validated by
Biological Validation
Species Reactivity
Validated:
Mouse
Cited:
Human, Mouse, Rat, Transgenic Mouse
Applications
Validated:
Immunohistochemistry, Western Blot, Neutralization
Cited:
Immunohistochemistry, Western Blot, Flow Cytometry, Immunocytochemistry, Immunoprecipitation
Label
Unconjugated
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
Mouse myeloma cell line NS0-derived recombinant mouse Cathepsin B
His18-Phe339
Accession # P10605
His18-Phe339
Accession # P10605
Specificity
Detects mouse Cathepsin B in direct ELISAs and Western blots. In direct ELISAs, approximately 15% cross-reactivity with recombinant human Cathepsin B is observed and less than 5% cross-reactivity with recombinant mouse Cathepsin H is observed.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Scientific Data Images for Mouse Cathepsin B Antibody
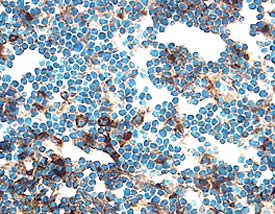
Cathepsin B in Mouse Thymus.
Cathepsin B was detected in perfusion fixed frozen sections of mouse thymus using 5 µg/mL Goat Anti-Mouse Cathepsin B Antigen Affinity-purified Polyclonal Antibody (Catalog # AF965) overnight at 4 °C. Tissue was stained with the Anti-Goat HRP-DAB Cell & Tissue Staining Kit (brown; Catalog # CTS008) and counterstained with hematoxylin (blue). View our protocol for Chromogenic IHC Staining of Frozen Tissue Sections.Detection of Mouse Cathepsin B by Immunocytochemistry/ Immunofluorescence
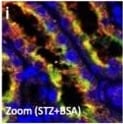
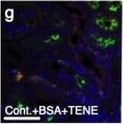
EMT in BSA-induced damaged tubule was associated with increased levels of DPP-4, integrin beta 1 and CAV1; TENE treatment ameliorated these alterations. (a–e) Multiplex immunofluorescence microscopy analysis of the EMT program and association with CAV1. Formaldehyde-fixed, paraffin-embedded (FFPE) kidney samples were labeled with epithelial markers for E-cadherin, alpha SMA and CAV1. An immunofluorescence analysis was performed by confocal microscopy. (d) The enlarged image of the inset shown in (c). The alpha SMA-positive damaged tubular cells were surrounded by alpha SMA-positive interstitial cells (f–j). Multiplex immunofluorescence was performed to analyze the crosstalk among DPP-4, integrin beta 1 and CAV1 in the BSA-injected diabetic mice. (i) The enlarged image of the inset shown in (h). DPP-4, integrin beta 1, and CAV1 were localized at the same location (likely the luminal side of the proximal tubule). The crosstalk occurred more frequently in the damaged tubular cells. Representative images from n = 7 in each group are shown. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/31101909), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse Cathepsin B by Immunohistochemistry
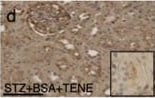
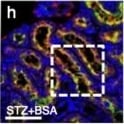
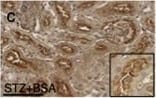
BSA-injected diabetic mice exhibited high tubular levels of DPP-4, CAV1 and EMT program; TENE treatment ameliorated these alterations. Immunohistochemical analysis of (a–d) DPP-4, (e–h) CAV1, (i–l) snail and (m–p) AQP-1 from the BSA-injected control or diabetic mice with or without the TENE treatment. Scale bar, 50 μm. Representative images from n = 7 in each group are shown. Each group was analyzed with an unpaired two-tailed t-test. *P < 0.05, **P < 0.01. Data are presented as mean ± s.e.m (q–t). Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/31101909), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse Cathepsin B by Immunocytochemistry/ Immunofluorescence
EMT in BSA-induced damaged tubule was associated with increased levels of DPP-4, integrin beta 1 and CAV1; TENE treatment ameliorated these alterations. (a–e) Multiplex immunofluorescence microscopy analysis of the EMT program and association with CAV1. Formaldehyde-fixed, paraffin-embedded (FFPE) kidney samples were labeled with epithelial markers for E-cadherin, alpha SMA and CAV1. An immunofluorescence analysis was performed by confocal microscopy. (d) The enlarged image of the inset shown in (c). The alpha SMA-positive damaged tubular cells were surrounded by alpha SMA-positive interstitial cells (f–j). Multiplex immunofluorescence was performed to analyze the crosstalk among DPP-4, integrin beta 1 and CAV1 in the BSA-injected diabetic mice. (i) The enlarged image of the inset shown in (h). DPP-4, integrin beta 1, and CAV1 were localized at the same location (likely the luminal side of the proximal tubule). The crosstalk occurred more frequently in the damaged tubular cells. Representative images from n = 7 in each group are shown. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/31101909), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse Cathepsin B by Immunocytochemistry/ Immunofluorescence
Alteration of the size of lysosomes and the expression of LAMP-1 in in Rab7 delta pan pancreatic acinar cells. (a–d) Immunofluorescence images of wild (a,c) and Rab7 delta pan (b,d) pancreases stained with anti-LAMP1(a,b) or anti-cathepsin B (c,d) antibodies (red). DAPI was used for nuclear staining (blue). Bars: 20 µm. (e) Quantification of the positive signals in immunofluorescence images of LAMP-1 (left panel) and cathepsin B (right panel). *P < 0.05. (f) WB of LAMP-1 using total pancreas homogenate of wild and Rab7 delta pan mice. An antibody against LAMP-1 N-terminal (top panel) and an antibody against LAMP-1 C-terminal (middle panel) were utilized. Anti-LAMP-1 N-terminal antibody revealed the shifting of intense bands to a lower position (top panel, arrow head) than that of full-length LAMP-1 (120 kDa) in Rab7 delta pan pancreas. In contrast, anti-LAMP-1 C-terminal antibody revealed bands at 120 kDa only in both wild and Rab7 delta pan pancreas (middle panel). beta -actin was used as an internal loading control (bottom panel). Image collected and cropped by CiteAb from the following open publication (https://www.nature.com/articles/s41598-017-02988-3), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse Cathepsin B by Immunocytochemistry/ Immunofluorescence
Alteration of the size of lysosomes and the expression of LAMP-1 in in Rab7 delta pan pancreatic acinar cells. (a–d) Immunofluorescence images of wild (a,c) and Rab7 delta pan (b,d) pancreases stained with anti-LAMP1(a,b) or anti-cathepsin B (c,d) antibodies (red). DAPI was used for nuclear staining (blue). Bars: 20 µm. (e) Quantification of the positive signals in immunofluorescence images of LAMP-1 (left panel) and cathepsin B (right panel). *P < 0.05. (f) WB of LAMP-1 using total pancreas homogenate of wild and Rab7 delta pan mice. An antibody against LAMP-1 N-terminal (top panel) and an antibody against LAMP-1 C-terminal (middle panel) were utilized. Anti-LAMP-1 N-terminal antibody revealed the shifting of intense bands to a lower position (top panel, arrow head) than that of full-length LAMP-1 (120 kDa) in Rab7 delta pan pancreas. In contrast, anti-LAMP-1 C-terminal antibody revealed bands at 120 kDa only in both wild and Rab7 delta pan pancreas (middle panel). beta -actin was used as an internal loading control (bottom panel). Image collected and cropped by CiteAb from the following open publication (https://www.nature.com/articles/s41598-017-02988-3), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse Cathepsin B by Immunocytochemistry/ Immunofluorescence
EMT in BSA-induced damaged tubule was associated with increased levels of DPP-4, integrin beta 1 and CAV1; TENE treatment ameliorated these alterations. (a–e) Multiplex immunofluorescence microscopy analysis of the EMT program and association with CAV1. Formaldehyde-fixed, paraffin-embedded (FFPE) kidney samples were labeled with epithelial markers for E-cadherin, alpha SMA and CAV1. An immunofluorescence analysis was performed by confocal microscopy. (d) The enlarged image of the inset shown in (c). The alpha SMA-positive damaged tubular cells were surrounded by alpha SMA-positive interstitial cells (f–j). Multiplex immunofluorescence was performed to analyze the crosstalk among DPP-4, integrin beta 1 and CAV1 in the BSA-injected diabetic mice. (i) The enlarged image of the inset shown in (h). DPP-4, integrin beta 1, and CAV1 were localized at the same location (likely the luminal side of the proximal tubule). The crosstalk occurred more frequently in the damaged tubular cells. Representative images from n = 7 in each group are shown. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/31101909), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse Cathepsin B by Immunohistochemistry
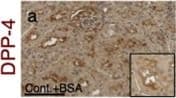
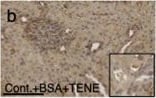
BSA-injected diabetic mice exhibited high tubular levels of DPP-4, CAV1 and EMT program; TENE treatment ameliorated these alterations. Immunohistochemical analysis of (a–d) DPP-4, (e–h) CAV1, (i–l) snail and (m–p) AQP-1 from the BSA-injected control or diabetic mice with or without the TENE treatment. Scale bar, 50 μm. Representative images from n = 7 in each group are shown. Each group was analyzed with an unpaired two-tailed t-test. *P < 0.05, **P < 0.01. Data are presented as mean ± s.e.m (q–t). Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/31101909), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse Cathepsin B by Western Blot
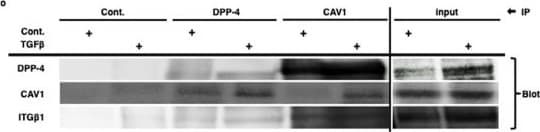
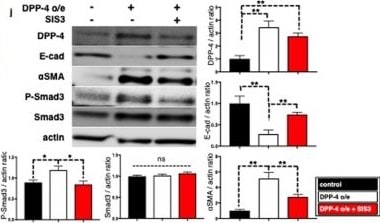
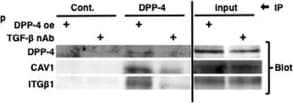
TENE treatment suppressed the crosstalk among DPP-4, integrin beta 1 and CAV1 via inhibition of TGF-beta /smad3 signaling pathway in vitro. Duolink in situ analysis of (a-c) DPP-4/integrin beta 1, (d–f) DPP-4/CAV1 and (g–i) integrin beta 1/CAV1 in HK-2 cells with or without TGF-beta 1 (10 ng/ml) was performed by confocal microscopy (×1260). Scale bar: 50 μm in each panel. (j) Representative western blot analysis. As a densitometric analysis, each protein level was normalized with actin. n = 6 per group were analyzed. (k–n) Duolink in situ analysis of integrin beta 1/CAV1 in DPP-4 overexpressed HK-2 cells with or without TENE and SIS3. (o) Immunoprecipitation analysis revealed TGF-beta treatment increased crosstalk among DPP-4, integrin beta 1 (ITG beta 1) and CAV1. (p) Immunoprecipitation assay revealed TGF-beta neutralization suppressed crosstalk among DPP-4, integrin beta 1 and CAV1 induced by DPP-4 overexpression. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/31101909), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse Cathepsin B by Immunohistochemistry
BSA-injected diabetic mice exhibited high tubular levels of DPP-4, CAV1 and EMT program; TENE treatment ameliorated these alterations. Immunohistochemical analysis of (a–d) DPP-4, (e–h) CAV1, (i–l) snail and (m–p) AQP-1 from the BSA-injected control or diabetic mice with or without the TENE treatment. Scale bar, 50 μm. Representative images from n = 7 in each group are shown. Each group was analyzed with an unpaired two-tailed t-test. *P < 0.05, **P < 0.01. Data are presented as mean ± s.e.m (q–t). Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/31101909), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse Cathepsin B by Western Blot
TENE treatment suppressed the crosstalk among DPP-4, integrin beta 1 and CAV1 via inhibition of TGF-beta /smad3 signaling pathway in vitro. Duolink in situ analysis of (a-c) DPP-4/integrin beta 1, (d–f) DPP-4/CAV1 and (g–i) integrin beta 1/CAV1 in HK-2 cells with or without TGF-beta 1 (10 ng/ml) was performed by confocal microscopy (×1260). Scale bar: 50 μm in each panel. (j) Representative western blot analysis. As a densitometric analysis, each protein level was normalized with actin. n = 6 per group were analyzed. (k–n) Duolink in situ analysis of integrin beta 1/CAV1 in DPP-4 overexpressed HK-2 cells with or without TENE and SIS3. (o) Immunoprecipitation analysis revealed TGF-beta treatment increased crosstalk among DPP-4, integrin beta 1 (ITG beta 1) and CAV1. (p) Immunoprecipitation assay revealed TGF-beta neutralization suppressed crosstalk among DPP-4, integrin beta 1 and CAV1 induced by DPP-4 overexpression. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/31101909), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse Cathepsin B by Western Blot
TENE treatment suppressed the crosstalk among DPP-4, integrin beta 1 and CAV1 via inhibition of TGF-beta /smad3 signaling pathway in vitro. Duolink in situ analysis of (a-c) DPP-4/integrin beta 1, (d–f) DPP-4/CAV1 and (g–i) integrin beta 1/CAV1 in HK-2 cells with or without TGF-beta 1 (10 ng/ml) was performed by confocal microscopy (×1260). Scale bar: 50 μm in each panel. (j) Representative western blot analysis. As a densitometric analysis, each protein level was normalized with actin. n = 6 per group were analyzed. (k–n) Duolink in situ analysis of integrin beta 1/CAV1 in DPP-4 overexpressed HK-2 cells with or without TENE and SIS3. (o) Immunoprecipitation analysis revealed TGF-beta treatment increased crosstalk among DPP-4, integrin beta 1 (ITG beta 1) and CAV1. (p) Immunoprecipitation assay revealed TGF-beta neutralization suppressed crosstalk among DPP-4, integrin beta 1 and CAV1 induced by DPP-4 overexpression. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/31101909), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse Cathepsin B by Immunohistochemistry
BSA-injected diabetic mice exhibited high tubular levels of DPP-4, CAV1 and EMT program; TENE treatment ameliorated these alterations. Immunohistochemical analysis of (a–d) DPP-4, (e–h) CAV1, (i–l) snail and (m–p) AQP-1 from the BSA-injected control or diabetic mice with or without the TENE treatment. Scale bar, 50 μm. Representative images from n = 7 in each group are shown. Each group was analyzed with an unpaired two-tailed t-test. *P < 0.05, **P < 0.01. Data are presented as mean ± s.e.m (q–t). Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/31101909), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse Cathepsin B by Immunocytochemistry/ Immunofluorescence
EMT in BSA-induced damaged tubule was associated with increased levels of DPP-4, integrin beta 1 and CAV1; TENE treatment ameliorated these alterations. (a–e) Multiplex immunofluorescence microscopy analysis of the EMT program and association with CAV1. Formaldehyde-fixed, paraffin-embedded (FFPE) kidney samples were labeled with epithelial markers for E-cadherin, alpha SMA and CAV1. An immunofluorescence analysis was performed by confocal microscopy. (d) The enlarged image of the inset shown in (c). The alpha SMA-positive damaged tubular cells were surrounded by alpha SMA-positive interstitial cells (f–j). Multiplex immunofluorescence was performed to analyze the crosstalk among DPP-4, integrin beta 1 and CAV1 in the BSA-injected diabetic mice. (i) The enlarged image of the inset shown in (h). DPP-4, integrin beta 1, and CAV1 were localized at the same location (likely the luminal side of the proximal tubule). The crosstalk occurred more frequently in the damaged tubular cells. Representative images from n = 7 in each group are shown. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/31101909), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse Cathepsin B by Immunocytochemistry/ Immunofluorescence
EMT in BSA-induced damaged tubule was associated with increased levels of DPP-4, integrin beta 1 and CAV1; TENE treatment ameliorated these alterations. (a–e) Multiplex immunofluorescence microscopy analysis of the EMT program and association with CAV1. Formaldehyde-fixed, paraffin-embedded (FFPE) kidney samples were labeled with epithelial markers for E-cadherin, alpha SMA and CAV1. An immunofluorescence analysis was performed by confocal microscopy. (d) The enlarged image of the inset shown in (c). The alpha SMA-positive damaged tubular cells were surrounded by alpha SMA-positive interstitial cells (f–j). Multiplex immunofluorescence was performed to analyze the crosstalk among DPP-4, integrin beta 1 and CAV1 in the BSA-injected diabetic mice. (i) The enlarged image of the inset shown in (h). DPP-4, integrin beta 1, and CAV1 were localized at the same location (likely the luminal side of the proximal tubule). The crosstalk occurred more frequently in the damaged tubular cells. Representative images from n = 7 in each group are shown. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/31101909), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse Cathepsin B by Immunocytochemistry/ Immunofluorescence
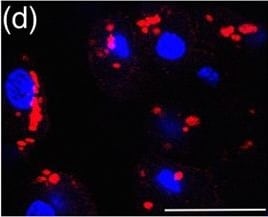
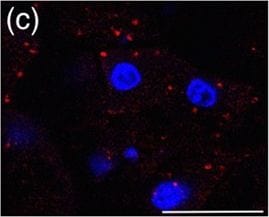
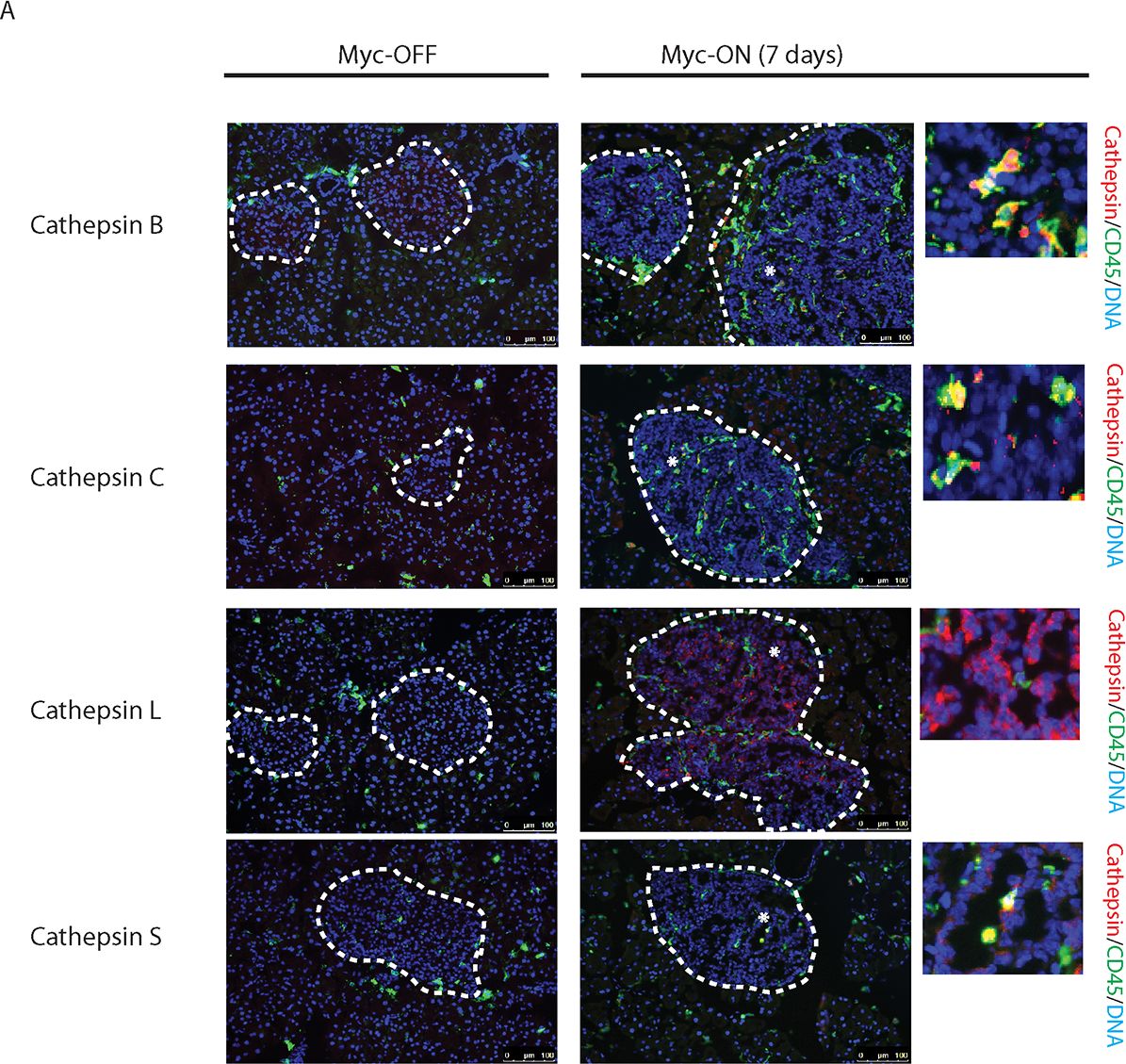
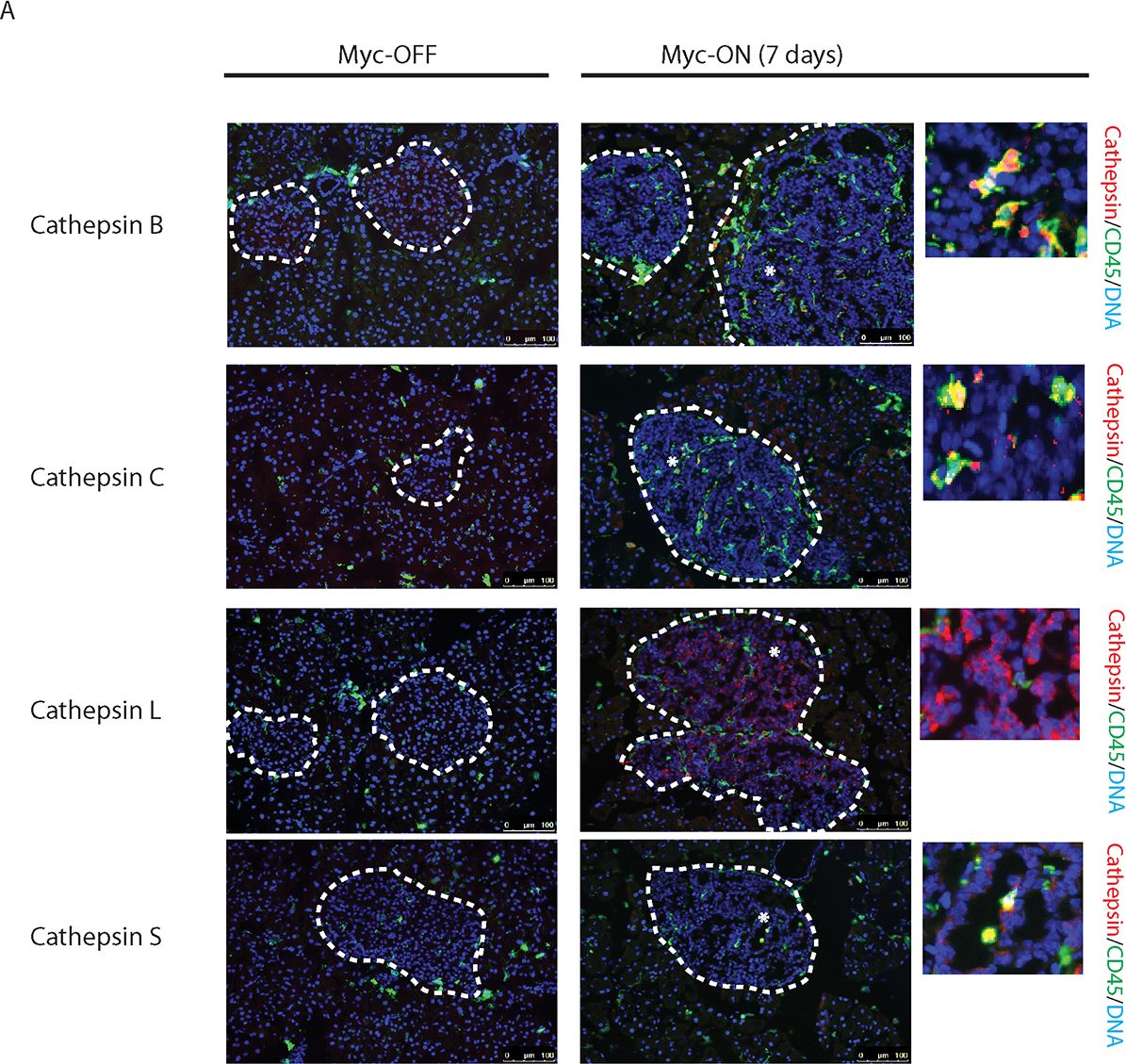
Myc induces cathepsin L expression in beta-cells of pancreatic Islets.(A) Immunohistochemical analyses for CTS B, C, L or S expression (all in red) in combination with staining for the pan-leukocyte marker CD45 (green) in pancreatic islet tumors from the MycERTAM;Bcl-xL animals. Pancreata were harvested from the MycERTAM;Bcl-xL mice treated for 7 d with TAM (Myc-On, 7 days) or control vehicle in place of TAM (Myc-OFF). The islet area is indicated by dotted line. The asterisks indicate the area of tumor represented in the insets. The panels are representatives of at least three animals assayed at each data point, all immunohistochemical analyses were done in duplicate; eight randomized fields per analysis were examined. Scale bars, 100μm. (B) Immunohistochemical analysis for cathepsin L expression in beta-cells of pancreatic islets from MycERTAM;Bcl-xL animals identified by insulin expression. Pancreata were collected from the animals described above. Scale bars represent 25μm. The panels are representatives of three animals assayed at each data point, all immunohistochemical analyses were done in duplicate; ten randomized fields per analysis were examined. Image collected and cropped by CiteAb from the following open publication (https://dx.plos.org/10.1371/journal.pone.0120348), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse Cathepsin B by Immunocytochemistry/ Immunofluorescence
Myc induces cathepsin L expression in beta-cells of pancreatic Islets.(A) Immunohistochemical analyses for CTS B, C, L or S expression (all in red) in combination with staining for the pan-leukocyte marker CD45 (green) in pancreatic islet tumors from the MycERTAM;Bcl-xL animals. Pancreata were harvested from the MycERTAM;Bcl-xL mice treated for 7 d with TAM (Myc-On, 7 days) or control vehicle in place of TAM (Myc-OFF). The islet area is indicated by dotted line. The asterisks indicate the area of tumor represented in the insets. The panels are representatives of at least three animals assayed at each data point, all immunohistochemical analyses were done in duplicate; eight randomized fields per analysis were examined. Scale bars, 100μm. (B) Immunohistochemical analysis for cathepsin L expression in beta-cells of pancreatic islets from MycERTAM;Bcl-xL animals identified by insulin expression. Pancreata were collected from the animals described above. Scale bars represent 25μm. The panels are representatives of three animals assayed at each data point, all immunohistochemical analyses were done in duplicate; ten randomized fields per analysis were examined. Image collected and cropped by CiteAb from the following open publication (https://dx.plos.org/10.1371/journal.pone.0120348), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Cathepsin B by Western Blot
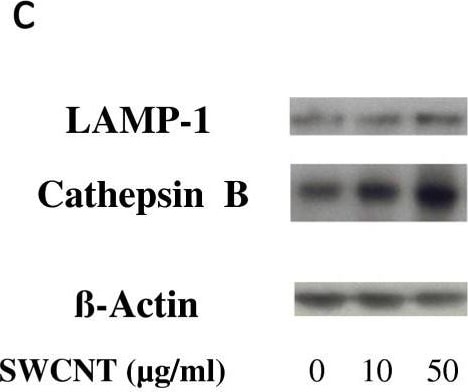
Evaluation of lysosomal activation in SWCNT-exposed cells. Panel a: Epi-fluorescence images of macrophages unexposed (upper line) or exposed (lower line) to 50 μg/ml SWCNT for 24 hours stained with Acridine Orange (for lysosomes, in red) and DAPI (RNA/DNA, in green). Merge: combination of Acridine Orange and DAPI staining (overlapping appeared yellow). Panel b: Cathepsin activity in macrophages exposed to SWCNT for 3 hours. *: p < 0.001 between groups. Panel c: Western Blot images of LAMP-1 (120 kDa) and Cathepsin B (37 kDa) expression in macrophages exposed for 24 hours to SWCNT. beta -Actin is given as internal standard. Panel d: quantification of LAMP-1 and Cathepsin B expression, normalized to beta -Actin expression. *: p < 0.05 between groups. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/23800198), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse Cathepsin B by Immunocytochemistry/ Immunofluorescence
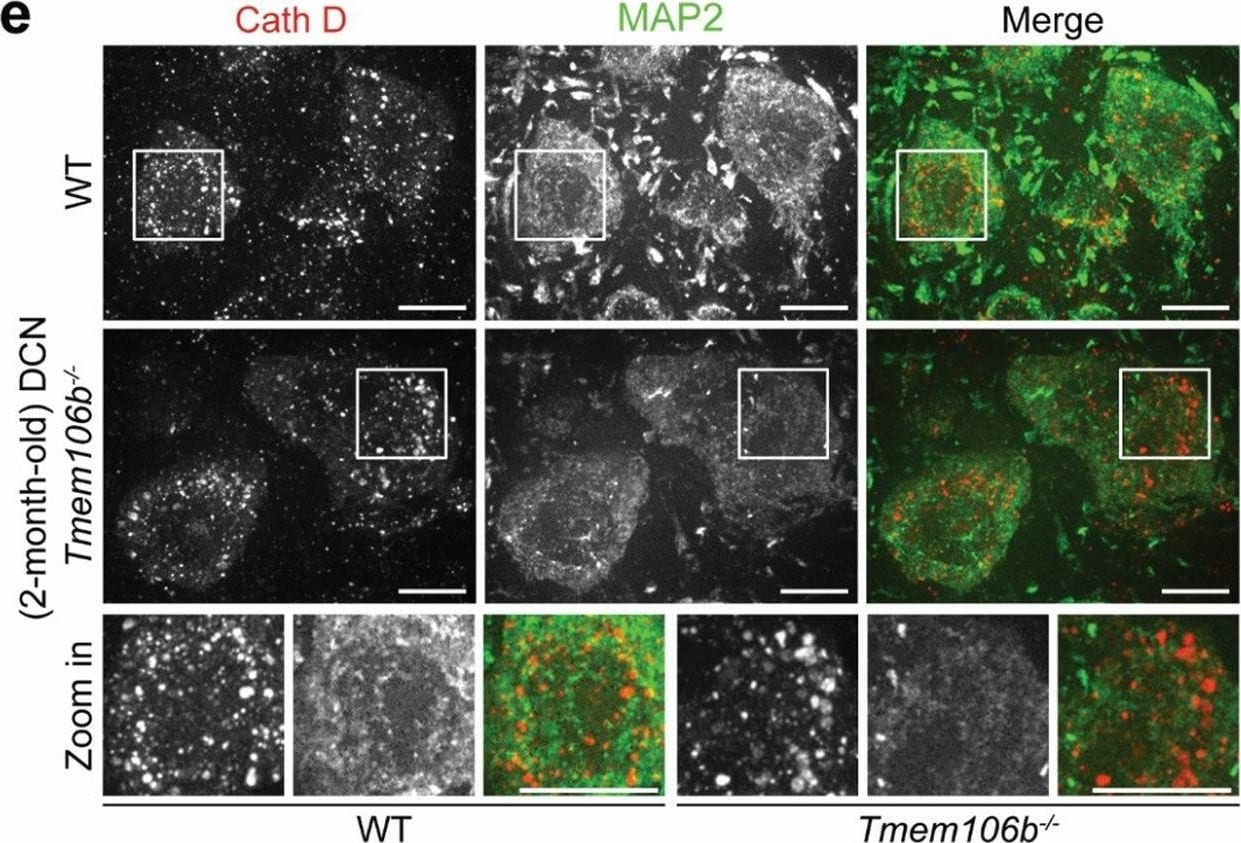
Distinct lysosomal phenotypes among different types of neurons in the cerebellum of young Tmem106b−/− mice. a, b Immunostaining of PVALB and Cathepsin D (Cath D) in cerebellar sections from 2-month‐old WT and Tmem106b−/− mice. Representative images from the molecular cell layer are shown. The intensity of Cath D in PVALB-positive interneurons was quantified in b. n = 3. ****, p < 0.0001, unpaired t-test. Scale bar = 10 μm. c, d Immunostaining of Cathepsin D (Cath D) and Hoechst in cerebellar sections from 2-month‐old WT and Tmem106b−/− mice, and images were captured from granule cell layer. The intensity of Cath D in the granule cell layer was quantified in d. n = 3. **, p < 0.001, unpaired t-test. Scale bar = 10 μm. e, f Immunostaining of Cathepsin D (Cath D) and MAP2 in cerebellar sections from 2-month‐old WT and Tmem106b−/− mice. Representative images from the DCN region are shown. The intensity of Cath D in MAP2-positive DCN neurons was quantified in f. Zoom-in image shows the enlarged lysosomes in 2-month‐old Tmem106b−/− mice compared with WT mice. n = 3. ****, p < 0.0001, unpaired t-test. Scale bar = 10 μm Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/35287730), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Cathepsin B by Immunohistochemistry
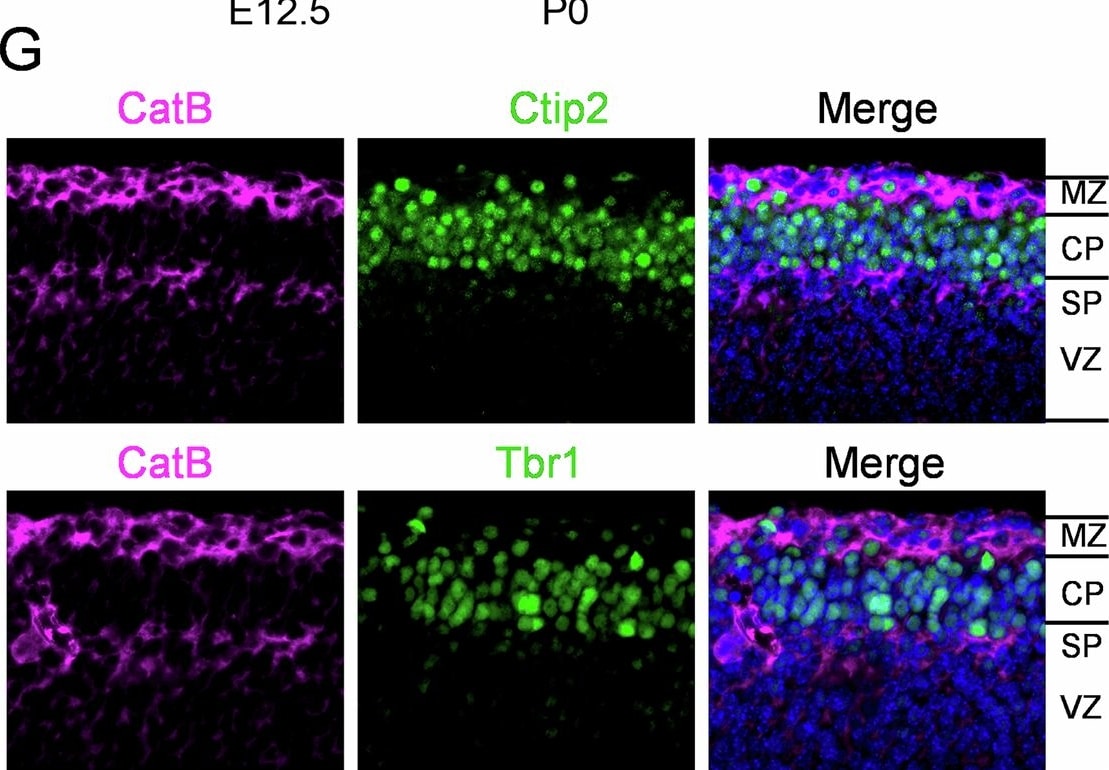
CatB is expressed highly in the developing mouse cortex. A CatB protein levels in mouse cortex at indicated embryonic (E), postnatal (P), or adult stages assessed by Western blot. Representative blots from three independent experiments (n = 3 mice per time point). B, C Quantification of pro-form (B) and mature-form (C) CatB expression normalized to Actin (n = 3 mice per group). D Schematic illustration of cell migration from ventricular zone (VZ) to preplate (PP) in E12.5 embryos. E Fluorescent images of CatB (magenta) and Hoechst (blue) in WT mice at E12.5, E14.5 and P0. Scale bar, 100 μm. F Quantification of CatB staining intensity in (E), normalized to VZ/SVZ at each time point. Images were taken from 3 mice per stage, 3 brain sections per mouse. G Immunofluorescent staining of CatB (magenta) with Ctip2 (green) or Trb1(green) in the cortex of WT mice at E12.5. Scale bar, 50 μm. *P < 0.05, **P < 0.01. Error bars represent mean ± SEM. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/40702210), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Mouse Cathepsin B Antibody
Application
Recommended Usage
Immunohistochemistry
5-15 µg/mL
Sample: Perfusion fixed frozen sections of mouse thymus
Sample: Perfusion fixed frozen sections of mouse thymus
Western Blot
0.1 µg/mL
Sample: Recombinant Mouse Cathepsin B (Catalog # 965-CY)
Sample: Recombinant Mouse Cathepsin B (Catalog # 965-CY)
Reviewed Applications
Read 1 review rated 5 using AF965 in the following applications:
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: Cathepsin B
References
- Mort, J.S. (2004) in Handbook of Proteolytic Enzymes (Barrett, A.J. et al. eds.) p. 1079, Academic Press, San Diego.
- Vancompernolle, K. et al. (1998) FEBS Lett. 438:150.
- Jutras, I. and T.L. Reudelhuber (1998) FEBS Lett. 443:48.
- Taggart, C.C. et al. (2001) J. Biol. Chem. 276:33345.
- Berquin, I.M. and B.F. Sloane (1996) Adv. Exp. Med. Biol. 389:281.
Alternate Names
CTSB
Gene Symbol
CTSB
UniProt
Additional Cathepsin B Products
Product Documents for Mouse Cathepsin B Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Mouse Cathepsin B Antibody
For research use only
Related Research Areas
Citations for Mouse Cathepsin B Antibody
Customer Reviews for Mouse Cathepsin B Antibody (1)
5 out of 5
1 Customer Rating
Have you used Mouse Cathepsin B Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Showing
1
-
1 of
1 review
Showing All
Filter By:
-
Application: Immunohistochemistry-ParaffinSample Tested: Kidney paraffin sectionSpecies: MouseVerified Customer | Posted 12/19/2014
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...
Associated Pathways