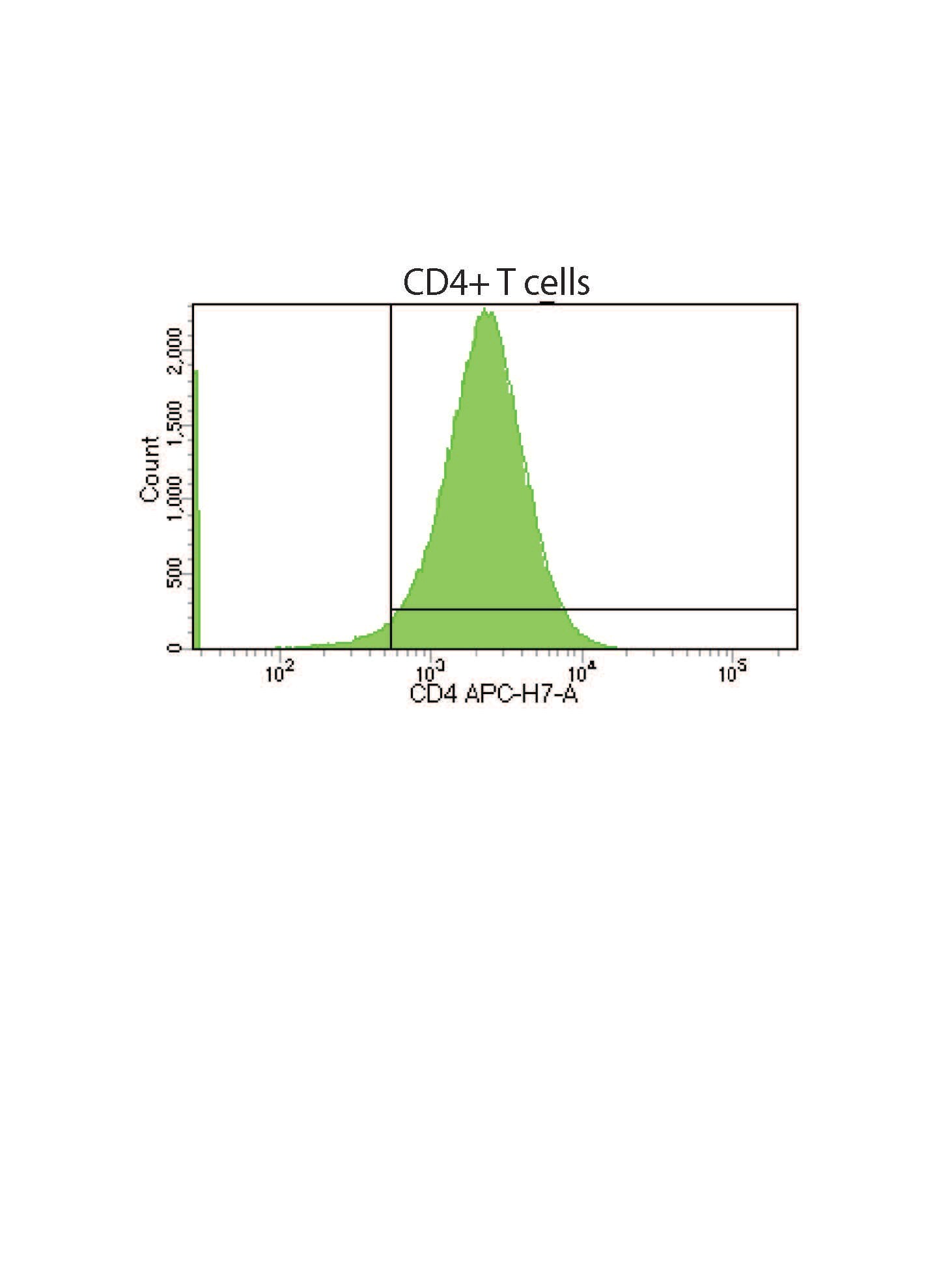
CD4 is a transmembrane glycoprotein that is expressed predominantly on thymocytes and a subset of mature T lymphocytes. It is a standard phenotype marker for the identification of T cell populations. CD4 is expressed along with CD8 on double positive T cells during their development in the thymus. Either CD4 or CD8 expression is then lost, giving rise to single positive (SP) CD4+ or CD8+ mature T cells. CD4+ SP cells, also known as T helper cells, further differentiate into multiple subsets of CD4+ cells including Th1, Th2, Th9, Th17,Th22, Tfh, and Treg cells which regulate humoral and cellular immunity. In human, CD4 is additionally expressed on macrophages, neutrophils, monocytes, NK cells, and neurons and glial cells in the brain. CD4 binds directly to MHC class II molecules on antigen presenting cells. This interaction contributes to the formation of the immunological synapse which is focused around the TCR-MHC class II-antigenic peptide interaction. CD4 also functions as a chemotactic receptor for IL-16 and, in human, as a coreceptor for the gp120 surface glycoprotein of HIV-1.
Mouse CD4+ T Cell Enrichment Column, Small, Pack of 4
R&D Systems | Catalog # MCD4C-1000

Key Product Details
Species
Product Summary for Mouse CD4+ T Cell Enrichment Column, Small, Pack of 4
Kit Summary
For the isolation of mouse CD4+ T cells.
Key Benefits
- Yields a highly pure (84-91%) population of CD4+ T cells
- Separates cells based on gravitational flow
- Negative selection reduces sources of experimental variation
Why Isolate CD4+ T cells?
CD4+ T cells develop in the thymus and differentiate into subsets of specialized effector T lymphocytes in response to distinct environmental cues and the activation of specific transcription factors. The isolation of CD4+ T cells is an important step in the preparation of multiple subsets including Th1, Th2, Th17, Th9, Th22, and regulatory T cells. CD4+ T cell subsets secrete characteristic combinations of cytokines which enables them to exert diverse functions including the recruitment and activation of additional immune cells, the dampening ongoing immune responses, and the maintenance of immunologic memory.
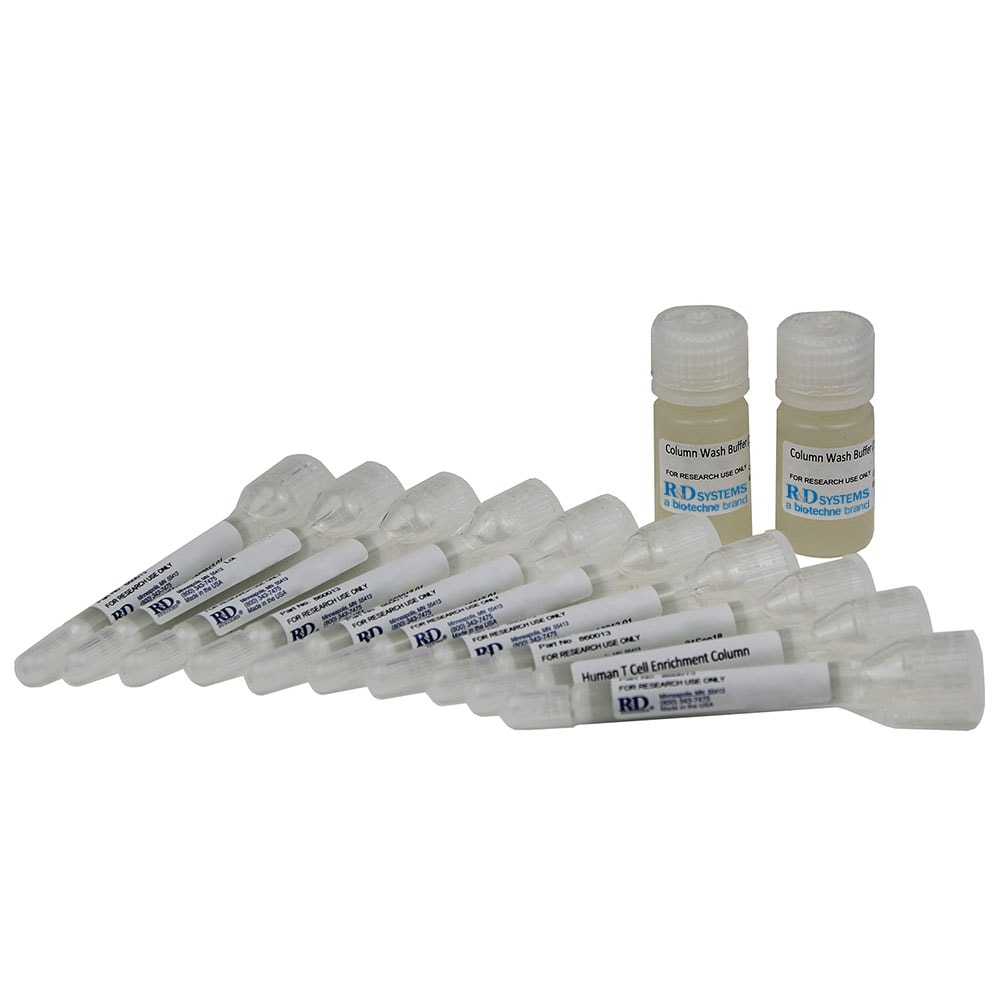
Kit Contents
The Mouse CD4+ T Cell Enrichment Column Kit, Small contains the following components to isolate mouse CD4+ T cells.
- Mouse CD4+ T Cell Subset Columns
- Monoclonal Antibody Cocktail
- Column Buffer Concentrate (10X)
Formulation, Preparation, and Storage
Shipping
Storage
Background: CD4
Alternate Names
Entrez Gene IDs
Gene Symbol
Additional CD4 Products
Product Documents for Mouse CD4+ T Cell Enrichment Column, Small, Pack of 4
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Mouse CD4+ T Cell Enrichment Column, Small, Pack of 4
For research use only
Citations for Mouse CD4+ T Cell Enrichment Column, Small, Pack of 4
Customer Reviews for Mouse CD4+ T Cell Enrichment Column, Small, Pack of 4 (1)
Have you used Mouse CD4+ T Cell Enrichment Column, Small, Pack of 4?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
-
Verified Customer | Posted 10/13/2017Have been using this kit in our laboratory for over 6 years in order to isolate CD4+ T cells for in vitro assays. Tim and time again these columns have yielded 90% or greater CD4+ T cell purity. I would recommend keeping lymph node and splenic cells separate when using the columns as they have a tendency to clump when put together before running through the columns.
There are no reviews that match your criteria.
Protocols
View specific protocols for Mouse CD4+ T Cell Enrichment Column, Small, Pack of 4 (MCD4C-1000):
Refer to the product datasheet for complete product details.
Briefly, mouse CD4+ T cells can be isolated using the following procedure:
- Incubate a single-cell suspension of leukocytes with the Monoclonal Antibody Cocktail
- Apply antibody-treated cell suspension to the CD4+ T Cell Subset Column
- Elute CD4+ T cells with 1X Column Buffer
Kit Contents
The Mouse CD4+ T Cell Enrichment Column Kit, Small (Catalog #MCD4C-1000) contains the following components to allow for the isolation of mouse CD4+ T cells.
- Mouse CD4+ T Cell Subset Columns
- Monoclonal Antibody Cocktail
- Column Buffer Concentrate (10X)
Other Supplies Required
Technical notes
- In order to best determine column performance, we advise that users retain a small portion of the starting cell population. Following cell selection with the column, it is then possible to perform immunophenotyping analysis on both starting and eluted cells. This information, when combined with the actual number of cells loaded and recovered, can then be used to calculate the percentage recovery of the target cell population.
- Care should be taken during cell preparation to reduce the number of platelets before loading the cells onto the column. Platelet contamination can be reduced by performing a final wash centrifugation of the cells at a reduced speed (150 - 200 x g).
- Some of the salts in the 10X column buffer solution may precipitate after storage at 4 °C. Should this be the case, do not carry out the 1:10 buffer dilution until all salts are in solution. This may be achieved by warming the 10X column buffer bottle in a 37 °C water bath for 5 - 10 minutes. Once there is no longer evidence of precipitates, the 10X column buffer may be diluted 1:10 to prepare the 1X column buffer for column processing.
Helpful Hints in Running T Cell Enrichment Columns
- Remove as many clumps as possible from the cell suspension before loading cells onto the column. Although the column is designed to filter out larger clumps of cells, too many clumps on the filter will reduce the column flow rate and cell recovery. In addition, leaving a large number of cells in a small volume of buffer for more than 30 minutes may promote cell clumping.
- If cells do not move into column after 15 minutes, the filter may have become clogged. If this occurs, move the white filter at the top of the column to the side with a sterile pipette. The cells should migrate into the column more easily.
- The column is designed so that the white filter at the top of the column bed will stop buffer flow and prevent the column from drying out. However leaving the open column exposed to air for more than 1 hour may cause the column bed to dry out and the CD4+ T cell enrichment to be compromised.
- Cell recovery after column processing is largely dependent on the total number of cells loaded. Optimal column performance is achieved by loading approximately 75 x106 cells. Loading fewer cells will negatively affect total cell recovery.
- If buffer does not drip out of column after initial removal of the bottom cap, try tapping the side of the column to remove any air locks that may be preventing the flow of buffer.
- The white filter at the top of the column bed may be found floating as a result of being dislodged during shipping. The column can still be used for cell processing after pushing the white filter onto the column bed with the back end of a sterile 2 mL pipette. The white filter should be positioned close to the column bed.
Procedure Overview
R&D Systems Protocol for Mouse CD4+ T Cell Enrichment Column, Small
Prepare a single cell suspension of mononuclear cells.
Wash the cells with excess sterile PBS.
Remove the red blood cells if necessary (e.g., using R&D Systems Mouse Erythrocyte Lysing Kit Catalog # WL2000)
Resuspend the cells in a small volume of 1X Column Wash Buffer.
Perform a cell count.
Adjust the cell concentration to 1-2 x 108 cells/mL with 1X Column Wash Buffer.

Mix 2 x 108 suspended cells with the contents of one vial of Monoclonal Antibody Cocktail.
Incubate for 15 minutes at room temperature.
Wash cells twice with 10 mL 1X Column Wash Buffer.
Resuspend cells in 2 mL of 1X Column Wash Buffer.
Load cell suspension onto the prepared column.
Incubate for 10 minutes at room temperature.
Elute CD4+ T cells with 10 mL of 1X Column Wash Buffer.
Collect the cells in a sterile 50 mL centrifuge tube.
Pellet the cells using centrifugation.
Resuspend CD4+ T cells in the appropriate buffer or culture medium for downstream applications.
FAQs
No product specific FAQs exist for this product.
View all FAQs for Cell Selection and Detection Kits