LBP (Lipopolysaccharide binding protein) is a 58‑62 kDa, single-chain glycoprotein member of the BPI/LBP family, BPI/PLUNC/PSP superfamily of lipid-binding proteins (1-3). It is secreted by a number of mammalian cell types, including hepatocytes (4), gingival keratinocytes (5), intestinal Paneth cells (6), and type II Greater alveolar cells (7). LBP is considered to be a class 1 APR (acute phase reactant) that is induced upon exposure to both IL-1 and IL-6 (8). These two cytokines appear upon immune cell exposure to pathogenic microbes. Following its synthesis and release, LBP is known to interact with bacterial wall components, lipopolysaccharide/LPS/Lipid A from Gram- (Gm-) bacteria, and lipoteichoic acid/LTA from Gm+ bacteria (9-13). In the case of LPS, this interaction appears to occur both in the bacterial cell wall, and within the intercellular space, where LPS micelles naturally form following bacterial death and cell wall dissolution (14-17). LBP is posited to induce disassembly of LPS micelles, allowing for LPS binding to LBP, and a heparin-mediated transfer of LPS from LBP to membrane-bound CD14 on the surface of monocytes/macrophages (15, 18). This CD14:LPS complex activates a TLR4:MD2 membrane complex, resulting in the production of NO and TNF-alpha (19). TNF-alpha serves as a chemoattractant for PMNs, and an initiator of coagulation that helps to wall-off and localize microbial elements (16). Notably, increased concentrations of LBP are also associated with parasitic infections (Trypanosoma), and may contribute to the immune response towards parasites (20). In addition to the above, LBP is also reported to transfer LPS to lipoproteins, particularly HDL and LDL (19, 21-23). For LDL, this transfer appears to be inhibitory to monocyte activation; for HDL, the effect may be either stimulatory or inhibitory, depending upon the circumstances (19). Mouse LBP is synthesized as a 481 amino acids (aa) precursor that contains a 25 aa signal sequence and a 456 aa mature region (aa 26-481) (24). It contains an N‑terminal LPS binding region plus a likely C-terminal LPS transfer region (24, 25). Mature mouse LBP shares 68% and 88% aa identity with human and rat LBP, respectively (11, 25).

Key Product Details
Species Reactivity
Mouse
Applications
Western Blot
Label
Unconjugated
Antibody Source
Polyclonal Sheep IgG
Loading...
Product Specifications
Immunogen
Mouse myeloma cell line NS0-derived recombinant mouse LBP
Gly25-Val481 (Ser102Arg, Tyr284His)
Accession # NP_032515
Gly25-Val481 (Ser102Arg, Tyr284His)
Accession # NP_032515
Specificity
Detects mouse LBP in direct ELISAs and Western blots. In direct ELISAs, approximately 25% cross-reactivity with recombinant human LBP is observed.
Clonality
Polyclonal
Host
Sheep
Isotype
IgG
Scientific Data Images for Mouse LBP Antibody
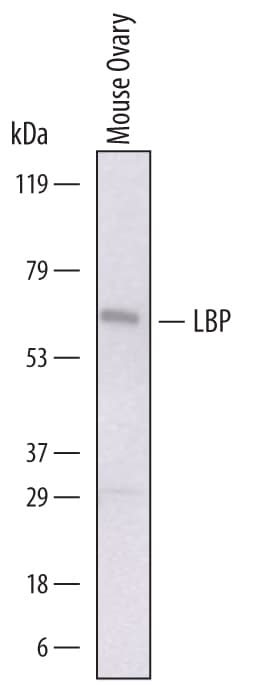
Detection of Mouse LBP by Western Blot.
Western blot shows lysates of mouse ovary tissue. PVDF membrane was probed with 1 µg/mL of Sheep Anti-Mouse LBP Antigen Affinity-purified Polyclonal Antibody (Catalog # AF6635) followed by HRP-conjugated Anti-Sheep IgG Secondary Antibody (Catalog # HAF016). A specific band was detected for LBP at approximately 65 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.Applications for Mouse LBP Antibody
Application
Recommended Usage
Western Blot
1 µg/mL
Sample: Mouse ovary tissue
Sample: Mouse ovary tissue
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Sterile PBS to a final concentration of 0.2 mg/mL. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: LBP
References
- Beamer, L.J. et al. (1998) Protein Sci. 7:906.
- Schroder, N.W.J. & R.R. Schumann (2005) J. Endotoxin Res. 11:237.
- Miyake, K. (2006) J. Endotoxin Res. 12:195.
- Grube, B.J. et al. (1994) J. Biol. Chem. 269:8477.
- Ren, L. et al. (2004) J. Periodont. Res. 39:242.
- Hansen, G.H. et al. (2009) Histochem. Cell Biol. 131:727.
- Dentener, M.A. et al. (2000) Am. J. Respir. Cell Mol. Biol. 23:146.
- Schumann, R.R. et al. (1996) Mol. Cell. Biol. 16:3490.
- Weber, J.R. et. al. (2003) Immunity 19:269.
- Schroder, N.W.J. et al. (2004) J. Immunol. 173:2683.
- Su, G.L. et al. (1994) J. Immunol. 153:743.
- Schroder, N.W.J. et al. (2003) J. Biol. Chem. 178:15587.
- Wright, S.D. et al. (1989) J. Exp. Med. 170:1231.
- Hallatschek, W. et al. (2004) Eur. J. Immunol. 34:1441.
- Schumann, R.R. & E. Latz (2000) Chem. Immunol. 74:42.
- Mannel, D.N. & B. Echtenacher (2000) Chem. Immunol. 74:141.
- Tsukamoto, H. et al. (2010) Int. Immunol. 22:271.
- Heinzelmann, M. & H. Bosshart (2005) J. Immunol. 174:2280.
- Gallay, P. et al. (1993) Infect. Immun. 61:378.
- Ngure, R.M. et al. (2009) Res. Vet. Sci. 86:394.
- Levels, J.H.M. et al. (2005) Infect. Immun. 73:2321.
- Hubacek, J.A. et al. (1997) Biochem. Biophys. Res. Commun. 236:427.
- Thompson, P.A. & R.L. Kitchens (2006) J. Immunol. 177:4880.
- Lengacher, S. et al. (1995-1996) J. Inflamm. 47:165.
- Schumann, R.R. et al. (1990) Science 249:1429.
Long Name
Lipopolysaccharide-binding Protein
Alternate Names
lipopolysaccharide binding protein, lipopolysaccharide-binding protein, LPS-binding protein, MGC22233
Gene Symbol
LBP
UniProt
Additional LBP Products
Product Documents for Mouse LBP Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Mouse LBP Antibody
For research use only
Related Research Areas
Citations for Mouse LBP Antibody
Customer Reviews for Mouse LBP Antibody
There are currently no reviews for this product. Be the first to review Mouse LBP Antibody and earn rewards!
Have you used Mouse LBP Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Cellular Response to Hypoxia Protocols
- R&D Systems Quality Control Western Blot Protocol
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...
