Nectins are a small family of Ca++-independent immunoglobulin (Ig)-like cell adhesion molecules (CAMs) that control cell adhesion, proliferation, and migration (1, 2, 3). The name Nectin derives from the Latin word necto, which means “to connect”. The Nectin family contains four members (Nectin-1 to -4), all of which show alternate splicing, a transmembrane (TM) region (except for Nectin-1 gamma which is secreted), and three extracellular Ig-domains. Nectins are highly homologous to the human receptor for poliovirus, and as such have been given the alternate name of poliovirus receptor-related proteins. They do not, however, appear to bind poliovirus (1). Mouse Nectin-2 is a 70 to 78 kDa type I TM glycoprotein that is found on a variety of cell types (4, 5). It has two splice forms (4, 6, 7). Nectin-2 alpha /PRR2 is a 65 kDa short form and is synthesized as a 467 amino acid precursor. It contains a 31 aa signal sequence, a 315 aa extracellular domain (ECD), a 28 aa TM segment, and a 93 aa cytoplasmic region. The ECD contains one N-terminal V-type Ig domain and two 85‑95 aa C2-type Ig-like domains (aa 153‑337) (8). The V-domain is believed to mediate Nectin binding to its ligands (9). A long, 78 kDa, 530 aa isoform of mouse Nectin-2 (Nectin-2 delta ) also exists. It has the same signal sequence and extracellular domain as Nectin-2 alpha (aa 1‑338), but differs in the TM segment (21 aa in length) and cytoplasmic region (159 aa in length) (4, 6, 7). Mouse Nectin-2 ECD (aa 32 - 338) shares 72%, 77% and 95% aa identity with the ECD in human, canine and rat Nectin-2, respectively. Nectin-2 is known to bind pseudorabies virus, and herpes simplex virus-2 (HSV-2). It also binds select HSV-1 strains. It does not bind poliovirus (1, 10, 11). As a cell adhesion molecule, Nectin-2 will form cis‑homodimers (same cell) and trans-homodimers (across cells). Nectin-2 will not cis‑dimerize with other Nectins, but will trans-heterodimerize with Nectin-3 and CD266/DNAM-1 (1, 3, 11, 12, 13). Nectin-2 is found concentrated at cell-to-cell interfaces, and is presumed to contribute to tight and adherens junction formation (14). Through its interaction with NK and T cell expressed DNAM-1, it also promotes lymphocyte cytotoxicity and cytokine secretion against both tumors and dendritic cells (DC) expressing Nectin-2 (15, 16). In the case of DC, this may be a mechanism whereby the immune system eliminates DC that are inefficient at antigen presentation. Nectin-2 is expressed on epithelium, endothelial cells, Sertoli cells, monocytes, dendritic cells, granulosa cells, mast cells, eosinophils and fibroblasts.
Mouse Nectin‑2/CD112 Antibody
R&D Systems | Catalog # MAB3869

Key Product Details
Species Reactivity
Validated:
Cited:
Applications
Validated:
Cited:
Label
Antibody Source
Product Specifications
Immunogen
Gln32-Gly351 (predicted)
Accession # P32507
Specificity
Clonality
Host
Isotype
Scientific Data Images for Mouse Nectin‑2/CD112 Antibody
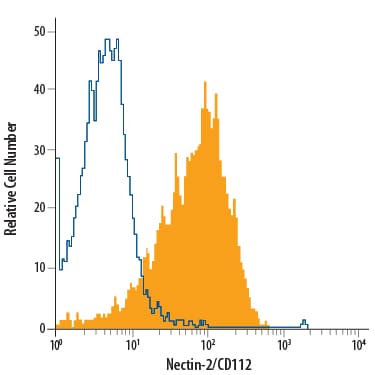
Detection of Nectin-2/CD112 in C2C12 Mouse Cell Line by Flow Cytometry.
C2C12 mouse myoblast cell line was stained with Rat Anti-Mouse Nectin-2/CD112 Monoclonal Antibody (Catalog # MAB3869, filled histogram) or isotype control antibody (Catalog # MAB006, open histogram), followed by Allophycocyanin-conjugated Anti-Rat IgG Secondary Antibody (Catalog # F0113).Applications for Mouse Nectin‑2/CD112 Antibody
CyTOF-ready
Flow Cytometry
Sample:
| C2C12 mouse myoblast cell line |
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Reconstitution
Sterile PBS to a final concentration of 0.5 mg/mL. For liquid material, refer to CoA for concentration.
Formulation
Shipping
Stability & Storage
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: Nectin-2/CD112
References
- Takai, Y. and H. Nakanishi (2003) J. Cell Sci. 116:17.
- Rikitake, Y. and Y. Takai (2008) Cell. Mol. Life Sci. 65:253.
- Sasisaka, T. et al. (2007) Curr. Opin. Cell Biol. 19:1.
- Aoki, J. et al. (1994) J. Biol. Chem. 269:8431.
- Takahashi, K. et al. (1999) J. Cell Biol. 145:539.
- Aoki, J. et al. (1997) Exp. Cell Res. 235:374.
- Lopez, M. et al. (1998) Blood 92:4602.
- Morrison, M.E. and V.R. Racaniello (1992) J. Virol. 66:2807.
- Struyf, F. et al. (2002) J. Virol. 76:12940.
- Delboy, M.G. et al. (2006) Virology J. 3:105.
- Irie, K. et al. (2004) Semin. Cell Dev. Biol. 15:643.
- Tahara-Hanaoka, S. et al. (2004) Int. Immunol. 16:533.
- Satoh-Horikawa, K. et al. (2000) J. Biol. Chem. 275:10291.
- Nakanishi, H. and Y. Takai (2004) Biol. Chem. 385:885.
- Tahara-Hanaoka, S. et al. (2006) Blood 107:1491.
- Pende, D. et al. (2006) Blood 107:2030.
Long Name
Alternate Names
Gene Symbol
UniProt
Additional Nectin-2/CD112 Products
Product Documents for Mouse Nectin‑2/CD112 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Mouse Nectin‑2/CD112 Antibody
For research use only
Related Research Areas
Citations for Mouse Nectin‑2/CD112 Antibody
Customer Reviews for Mouse Nectin‑2/CD112 Antibody
There are currently no reviews for this product. Be the first to review Mouse Nectin‑2/CD112 Antibody and earn rewards!
Have you used Mouse Nectin‑2/CD112 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Liperfluo
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- View all Protocols, Troubleshooting, Illustrated assays and Webinars

