Encoded by the THBD gene, Thrombomodulin is also known as CD141 antigen. The deduced amino acid (aa) sequence of mouse THBD predicts a signal peptide (aa 1 to 16) and a mature chain (aa 17 to 577) that consists of the following domains: C-type lectin (aa 31 to 167), EGF-like (aa 240 to 280, aa 283 to 323, aa 324 to 362, aa 364 to 404, aa 405 to 439, and aa 440 to 480), transmembrane (aa 518 to 541) and cytoplasmic (aa 542 to 577) (1). Predominantly synthesized by vascular endothelial cells, THBD inhibits coagulation and fibrinolysis (2‑4). It functions as a cell surface receptor and an essential cofactor for active thrombin, which in turn activates protein C and thrombin-activatable fibrinolysis inhibitor (TAFI), also known as carboxypeptidase B2 (CPB2). Activated protein C (APC), facilitated by protein S, degrades coagulation factors Va and VIIIa, which are required for thrombin activation. Activated CPB2 cleaves basic C-terminal amino acid residues from its substrates, including fibrin, preventing the conversion of plasminogen to plasmin. In addition, THBD gene polymorphisms are associated with human disease and THBD plays a role in thrombosis, stroke, arteriosclerosis, and cancer (5). For example, increased serum levels of THBD, due to protease cleavage, have been associated with smoking, atherosclerosis, liver cirrhosis, diabetes mellitus, cerebral and myocardial infarction, and multiple sclerosis (6). Over aa 17-517, mouse Thrombomodulin shares 84% and 66% aa sequence identity with rat and human thrombomoduli, respectively.
Mouse Thrombomodulin/BDCA‑3 APC‑conjugated Antibody
R&D Systems | Catalog # FAB3894A

Key Product Details
Species Reactivity
Applications
Label
Antibody Source
Product Specifications
Immunogen
Leu17-Ser517
Accession # P15306
Specificity
Clonality
Host
Isotype
Scientific Data Images for Mouse Thrombomodulin/BDCA‑3 APC‑conjugated Antibody
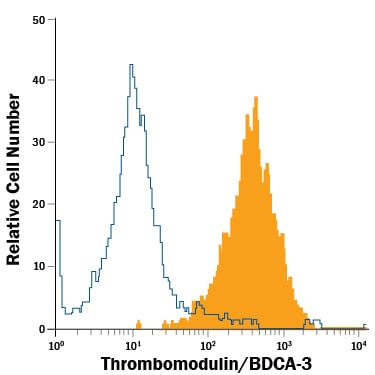
Detection of Thrombomodulin/BDCA‑3 in bEnd.3 Mouse Cell Line by Flow Cytometry.
bEnd.3 mouse endothelioma cell line was stained with Rat Anti-Mouse Thrombomodulin/BDCA-3 APC-conjugated Monoclonal Antibody (Catalog # FAB3894A, filled histogram) or isotype control antibody (Catalog # IC013A, open histogram). View our protocol for Staining Membrane-associated Proteins.Applications for Mouse Thrombomodulin/BDCA‑3 APC‑conjugated Antibody
Flow Cytometry
Sample: bEnd.3 mouse endothelioma cell line
Spectra Viewer
Plan Your Experiments
Use our spectra viewer to interactively plan your experiments, assessing multiplexing options. View the excitation and emission spectra for our fluorescent dye range and other commonly used dyes.
Spectra ViewerFlow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Formulation
Shipping
Stability & Storage
Background: Thrombomodulin/BDCA-3
References
- Dittman, W.A. and P.W. Majerus (1989) Nucleic Acids Res. 17:802.
- Van de Wouwer, M. et al. (2004) Arterioscler. Thromb. Vasc. Biol. 24:1374.
- Wu, K.K. et al. (2000) Ann Med. 32:73.
- Li, Y.H. et al. (2006) Cardiovasc. Hematol. Agents Med. Chem. 4:183.
- Weiler, H. and B.H. Isermann (2003) J. Thromb. Haemost. 1:1515.
- Califano, F. et al. (2000) Eur. Rev. Med. Pharmacol. Sci. 4:59.
Alternate Names
Gene Symbol
UniProt
Additional Thrombomodulin/BDCA-3 Products
Product Documents for Mouse Thrombomodulin/BDCA‑3 APC‑conjugated Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Mouse Thrombomodulin/BDCA‑3 APC‑conjugated Antibody
For research use only
Customer Reviews for Mouse Thrombomodulin/BDCA‑3 APC‑conjugated Antibody
There are currently no reviews for this product. Be the first to review Mouse Thrombomodulin/BDCA‑3 APC‑conjugated Antibody and earn rewards!
Have you used Mouse Thrombomodulin/BDCA‑3 APC‑conjugated Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Liperfluo
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Associated Pathways
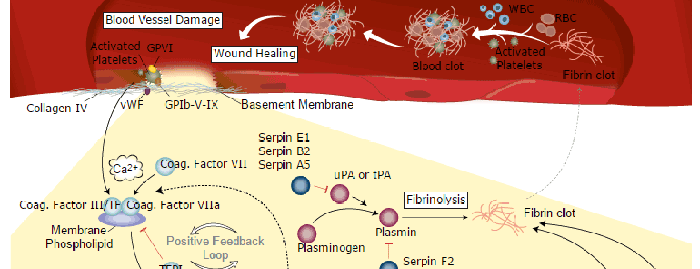 Dendritic Cell Lineage Development Pathways
Dendritic Cell Lineage Development Pathways