MycoProbe Mycoplasma Detection Kit
R&D Systems | Catalog # CUL001B

Product Summary for MycoProbe Mycoplasma Detection Kit
A complete kit to detect common antibiotic-resistant cell culture contaminants
Key Benefits
- Reduces experimental variation
- Accurate and highly sensitive
- Simple multiwell-based colorimetric assay
- Only takes 4.5 hours
Why should I be worried about mycoplasma contamination?
Mycoplasma contamination affects up to 80% of continuous cell cultures. If undetected, mycoplasma contamination can have significant effects on the quality and reliability of your cell culture preparations.
Mycoplasma contamination:
- Induces deleterious effects on cell culture quality.
- Alters the phenotypic characteristics of host cells.
- Increases experimental variation.
- Is common in eukaryotic cell cultures.
- Is resistant to antibiotics such as penicillin and streptomycin.
- Has multiple sources (i.e. personnel, reagents, other infected cells).
Mycoplasma are difficult to detect because they:
- Are not visible using standard microscopes.
- Are small enough to pass through 0.45 mm sterilization filters.
- Do not produce changes in culture medium color, pH, or turbidit
What is the best method to determine if my cultured cells are mycoplasma-contaminated?
To provide an accurate and highly sensitive tool for routine screening of mycoplasma contamination in cultured cells, R&D Systems developed the MycoProbe Mycoplasma Detection Assay. This assay detects Mycoplasma 16S ribosomal RNA (rRNA) using a colorimetric signal amplification system with sensitivity comparable to PCR. The MycoProbe assay is not susceptible to common problems encountered with PCR-based mycoplasma detection kits.
The MycoProbe Mycoplasma Detection Assay:
- Detects the eight mycoplasma species known to cause 95% of eukaryotic cell culture contamination.
- Is highly sensitive (comparable to PCR).
- Is compatible with high-throughput screening.
- Does not generate false positives from amplicon contamination.
- Can be used for cell culture supernatants or cultured cell pellets.
- Can be used with samples from fresh or frozen cells.
- Does not require cells to be cultured in antibiotic-free media.
- Includes a synthetic DNA oligonucleotide positive control.
- Generates results in 4.5 hours.
Alternative Methods for Mycoplasma Detection
|
Technique |
Advantage |
Disadvantage |
|---|---|---|
| Microbiological culture | Most sensitive |
|
| Fluorescent DNA staining | Efficient |
|
| ELISA of cell surface antigens | Common technique |
|
| PCR-based detection | High sensitivity |
|
| Biochemical activity | Common technique |
|
The MycoProbeTM Mycoplasma Detection Kit (Catalog # CUL001B) contains enough reagents to assay one 96-well plate for mycoplasma contamination.
- Cell Lysis Diluent Concentrate - 2 vials (1.7 mL/vial) of a 10-fold concentrated solution
- Hybridization Plate - One 96 well polystyrene microplate
- Streptavidin Plate - One 96 well polystyrene microplate (12 strips of 8 wells) coated with streptavidin
- Sample Diluent - 2 vials (21 mL/vial) of a buffered protein solution with preservatives
- Anti-digoxigenin Conjugate - 21 mL of a polyclonal antibody against digoxigenin conjugated to alkaline phosphatase with preservatives
- Capture Probes - 1.1 mL of a six-fold concentrated stock solution
- Detection Probes - 1.1 mL of a six-fold concentrated stock solution
- Positive Control - 1.1 mL of a solution containing a synthetic DNA oligonucleotide
- Wash Buffer Concentrate - 100 mL of a 10-fold concentrated solution with preservatives
- Substrate - 1 vial of lyophilized NADPH with stabilizers
- Substrate Diluent - 1 vial (7 mL) of a buffered solution with stabilizers
- Amplifier - 1 vial of lyophilized amplifier enzymes with stabilizers
- Amplifier Diluent - 1 vial (7 mL) of a buffered solution containing INT-violet with stabilizers
- Stop Solution - 6 mL of 2 N sulfuric acid
- Float Collar - Microplate float collar for water bath
- Plate Sealers - 12 adhesive strips
The Wash Buffer supplied in this kit contains sodium azide, which may react with lead and copper plumbing to form explosive metallic azides. Flush with large volumes of water during disposal.
The Stop Solution provided with this kit is an acid solution. Wear eye, hand, face, and clothing protection when using this material.
When handling cell culture samples, appropriate precautions should be taken to prevent exposure to mycoplasma and other hazardous biological agents.
Scientific Data Images for MycoProbe Mycoplasma Detection Kit
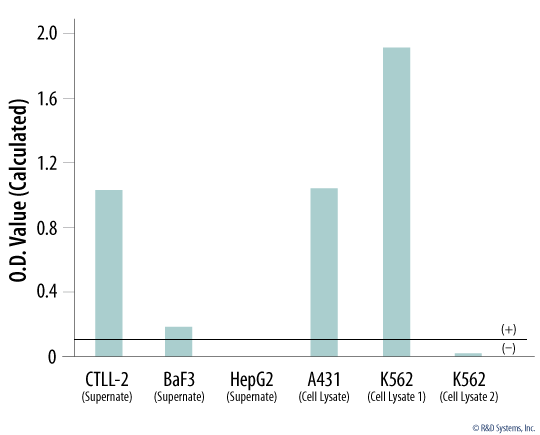
Mycoplasma Detection in Cell Line Supernates and Cell Lysates.
The presence of the eight mycoplasma species known to cause 95% of eukaryotic cell culture contamination was tested in supernates and cell lysates of the indicated cell lines using the MycoProbe Mycoplasma Detection Kit (Catalog # CUL001B). The average of the duplicate optical density (OD) readings for each control and sample was determined. The average negative control OD value was subtracted from all average sample OD values. A calculated positive control OD value of >0.10 indicated mycoplasma contamination (black line) in the CTLL-2, BaF3, A431, and K562 (cell lysate 1) samples. Abbreviations: CTLL-2 mouse cytotoxic T cell line, BaF3 mouse pro-B cell line, HepG2 human hepatocellular carcinoma cell line, A431 human epithelial carcinoma cell line, and K562 human chronic myelogenous leukemia cell line.Formulation, Preparation, and Storage
Shipping
Storage
Product Documents for MycoProbe Mycoplasma Detection Kit
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for MycoProbe Mycoplasma Detection Kit
For research use only
Related Research Areas
Citations for MycoProbe Mycoplasma Detection Kit
Customer Reviews for MycoProbe Mycoplasma Detection Kit (6)
Have you used MycoProbe Mycoplasma Detection Kit?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
-
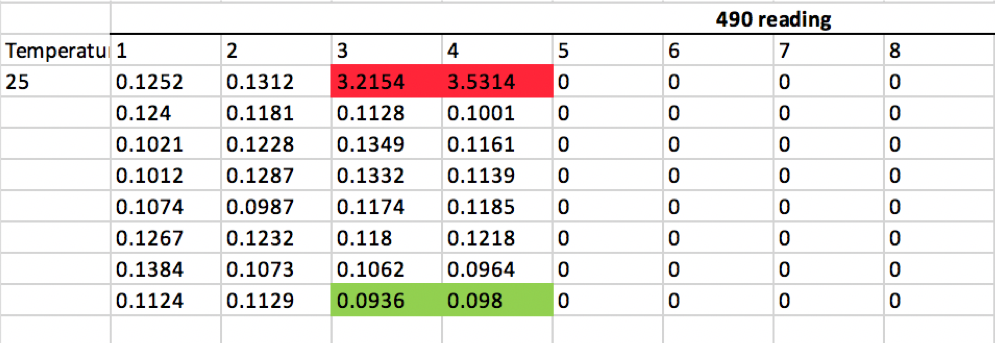
Verified Customer | Posted 05/20/2021This is a alternative to PCR for mycoplasma testing. I used it to test cell culture supernatants but can also be used for cell lysates. The positive control wells are in red and negative control are in green. 14 samples were measured in duplicate and after subtracting the negative control all were well below the lower threshold of 0.05. The kit assay requires about 6 hours to complete but mostly consists of incubation steps, one of which requires 65 degree water bath. The one downside is the kit is best for measuring large batches of samples all at once given the plate based format.
-
Verified Customer | Posted 05/05/2020This is the best kit I have ever used for testing mycoplasma, better than PCR and fluorescence based assays. It can test both cells and media, both frozen and fresh samples. It does not require 2 weeks antibiotic free culture time. It takes about 5 to 6 hours to finish. The only not very convenient step is the 65C incubation in water bath.
-
Verified Customer | Posted 12/06/2019
-
Verified Customer | Posted 04/03/2018
-
Verified Customer | Posted 07/07/2017
-
Verified Customer | Posted 05/13/2016Our lab has been using this kit for routine Mycoplasma testing of cultures for over 2 years now. The kit is highly sensitive and specific to Mycoplasma. The data is reproducible and more accurate than other tests, the size of the assay is adjustable (the kit comes as 12 8-well strips). You get reliable results in a few hours. Overall, great product and highly recommended.
There are no reviews that match your criteria.
Protocols
View specific protocols for MycoProbe Mycoplasma Detection Kit (CUL001B):
Refer to the product datasheet for complete product details.
Briefly, mycoplasma contamination can be evaluated in cell culture supernates or cell pellets using this straightforward procedure:
- Samples are lysed and hybridized with biotin-labeled capture oligonucleotide probes
- Digoxigenin-labeled detection probes target the eight most common mycoplasma contaminants
- Following signal amplification, multiwells are measured using a standard colorimetric plate reader
- Results are generated in 4.5 hours
View Graphic Procedure
Reagents Provided
Reagents supplied in the MycoProbe Mycoplasma Detection Kit (Catalog # CUL001B):
- Cell Lysis Diluent Concentrate - 2 vials (1.7 mL/vial) of a 10-fold concentrated solution
- Hybridization Plate - One 96 well polystyrene microplate
- Streptavidin Plate - One 96 well polystyrene microplate (12 strips of 8 wells) coated with streptavidin
- Sample Diluent - 2 vials (21 mL/vial) of a buffered protein solution with preservatives
- Anti-digoxigenin Conjugate - 21 mL of a polyclonal antibody against digoxigenin conjugated to alkaline phosphatase with preservatives
- Capture Probes - 1.1 mL of a six-fold concentrated stock solution
- Detection Probes - 1.1 mL of a six-fold concentrated stock solution
- Positive Control - 1.1 mL of a solution containing a synthetic DNA oligonucleotide
- Wash Buffer Concentrate - 100 mL of a 10-fold concentrated solution with preservatives
- Substrate - 1 vial of lyophilized NADPH with stabilizers
- Substrate Diluent - 1 vial (7 mL) of a buffered solution with stabilizers
- Amplifier - 1 vial of lyophilized amplifier enzymes with stabilizers
- Amplifier Diluent - 1 vial (7 mL) of a buffered solution containing INT-violet with stabilizers
- Stop Solution - 6 mL of 2 N sulfuric acid
- Float Collar - Microplate float collar for water bath
- Plate Sealers - 12 adhesive strips
Other Supplies Required
Reagents
- Deionized water, RNase-free
- Cell samples of interest
Materials
- Pipettes and pipette tips
- Squirt bottle or manifold dispenser
- 100 mL and 1,000 mL graduated cylinders for preparation of Wash Buffer
- Gloves and mask
Equipment
- Microplate reader capable of measuring absorbance at 490 nm with the correction wavelength set at 650 nm or 690 nm
- Horizontal orbital microplate shaker (0.12" orbit) capable of maintaining a speed of 500 + 50 rpm
- 65 + 1 °C water bath
- Vortex mixer
Procedure Overview
R&D Systems Protocol for Mouse Treg Cell Differentiation
- Wash the Hybridization Plate 2 times with Wash Buffer.
- Add diluted Probes, Positive Control, Sample Diluent (Negative Control), or sample to the designated wells.
- Incubate the plate for 60 minutes in a 65 °C water bath.
- Wash the Streptavidin Plate 2 times with Wash Buffer.
- Transfer 150 µL from each well of the Hybridization Plate to the Streptavidin Plate.
- Incubate for 60 minutes on a horizontal orbital shaker.
- Wash the Streptavidin Plate 4 times with Wash Buffer.
- Add Anti-Digoxigenin Conjugate to each well.
- Incubate for 60 minutes on a shaker.
- Wash the Streptavidin Plate 6 times with Wash Buffer.
- Add Substrate Solution to each well.
- Incubate for 60 minutes on a shaker.
- Do not wash.
- Add Amplifier Solution to each well.
- Incubate for 30 minutes on a shaker.
- Do not wash.
- Add Stop Solution to each well.
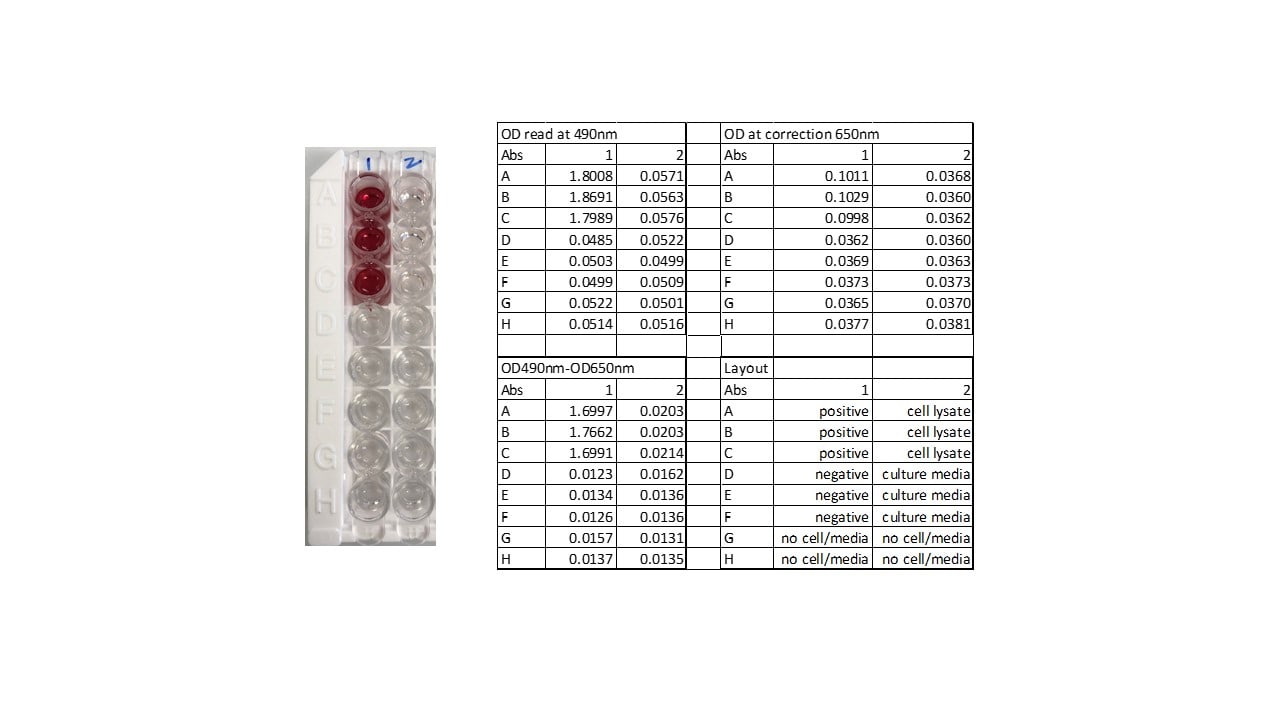
- Determine the optical density (OD) of each well within 30 minutes, using a microplate reader set to 490 nm.
: If wavelength correction is available, set to 650 nm or 690 nm. If wavelength correction is not available, subtract readings at 650 nm or 690 nm from the readings at 490 nm. This subtraction will correct for optical imperfections in the plate. Readings made directly at 490 nm without correction may be higher and less accurate.
Determine the average of the duplicate optical density (OD) readings for each control and sample. Subtract the average negative control OD value from all average OD values. The calculated positive control OD value should be > 1.5.
|
OD Values (Calculated) |
Result |
Interpretation |
|---|---|---|
| < 0.05 | Negative | No mycoplasma detected |
| 0.05 – 0.10 | Inconclusive | Sample is suspect for mycoplasma. Continue to culture for an additional 2 - 3 days and repeat the test. If sample gives a similar OD, then no mycoplasma are detected. |
| > 0.10 | Positive | Mycoplasma detected |
FAQs for MycoProbe Mycoplasma Detection Kit
-
Q: Is using partial plates an option for Cat. # CUL001B?
A: Cat. # CUL001B comes with a hybridization microplate and a streptavidin-coated microplate. The hybridization microplate is a solid, non-dismountable frame without removable strips, whereas the streptavidin-coated microplate is made of 12 strips of 8 wells. To run partial plates, cover the unused portion of the hybridization microplate with a plate sealer. Return unused wells of the streptavidin-coated microplate to the foil pouch containing the desiccant pack, reseal along entire edge of zip-seal. Unused partial microplates can be stored up to 3 months at 2-8 °C, as directed on the product datasheet.
-
Q: What are recommendations for cell culture numbers prior to evaluating supernatant in the kit?
A: For adherent cells, we recommend culturing cells to confluence prior to the assay. For suspension cells, we recommend culturing to a density of 0.5-1 x 10^6 cells/mL.
-
Q: Is using partial plates an option for Cat. # CUL001B?
A: Cat. # CUL001B comes with a hybridization microplate and a streptavidin-coated microplate. The hybridization microplate is a solid, non-dismountable frame without removable strips, whereas the streptavidin-coated microplate is made of 12 strips of 8 wells. To run partial plates, cover the unused portion of the hybridization microplate with a plate sealer. Return unused wells of the streptavidin-coated microplate to the foil pouch containing the desiccant pack, reseal along entire edge of zip-seal. Unused partial microplates can be stored up to 3 months at 2-8 °C, as directed on the product datasheet.
-
Q: What are recommendations for cell culture numbers prior to evaluating supernatant in the kit?
A: For adherent cells, we recommend culturing cells to confluence prior to the assay. For suspension cells, we recommend culturing to a density of 0.5-1 x 10^6 cells/mL.