MZ 1
Tocris Bioscience | Catalog # 6154


Key Product Details
Description
Product Description
MZ 1 is a cell penetrant Degrader (PROTAC®) based on (+)-JQ1 (Cat. No. 4499) conjugated to a von Hippel-Lindau (VHL) ligand. MZ 1 induces preferential degradation of BRD4 over BRD2 and BRD3 (DC50 values for degradation of BRD4 are 8 and 23 nM in H661 and H838 cells, respectively), while retaining high affinity for BRD2, BRD3 and BRD4 bromodomains (Kd = 13-60 nM). MZ 1 induces complete degradation of BRD4 at a concentration of 100 nM, whereas complete degradation of BRD2/3 is achieved at 2 μM. Potent cytotoxicity and antiproliferative effects are exhibited in AML cell lines (pEC50 = 7.6 in Mv4-11 cells).Negative control cis MZ 1 also available.
PROTAC® is a registered trademark of Arvinas Operations, Inc., and is used under license.
Scientific Data Images for MZ 1
Application of MZ 1 in HeLa Cells.
Western Blot data showing knockdown of BRD4 long isoform after MZ-1 (Catalog # 6154, 1 μM) treatment of HeLa cells. Protein quantification (relative to DMSO-only control) is shown beneath the corresponding lane. BRD4 antibody is CST#13440 used at 1:2000 dilution. Secondary is Anti-Rabbit HAF008, 1:1000, R&D Systems. GAPDH primary antibody is R&D Systems AF5718 used at 5μg/mL. Secondary is Anti-Goat HAF017, 1:1000, R&D Systems. Data courtesy of Jeff Cooper, Bio-Techne.Licensing Information
Sold under licence from the University of DundeeExternal Portal Information
Chemicalprobes.org is a portal that offers independent guidance on the selection and/or application of small molecules for research. The use of MZ 1 is reviewed on the Chemical Probes website.
Product Specifications for MZ 1
Molecular Weight
Formula
Storage
Purity
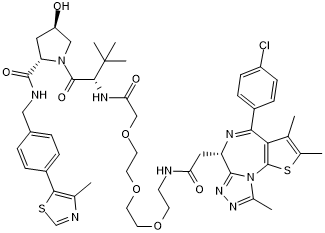
Chemical Name
CAS Number
PubChem ID
InChI Key
SMILES
The technical data provided above is for guidance only. For batch specific data refer to the Certificate of Analysis.
Solubility
| Solvent | Max Conc. mg/mL | Max Conc. mM | |
|---|---|---|---|
| Solubility | |||
| DMSO | 100.26 | 100 | |
| Ethanol | 100.26 | 100 |
Preparing Stock Solutions for MZ 1
The following data is based on the product molecular weight 1002.64.
Batch specific molecular weights may vary from batch to batch due to the degree of hydration, which all affect the solvent volumes required to prepare stock solutions.
| Concentration / Solvent Volume / Mass | 1 mg | 5 mg | 10 mg |
|---|---|---|---|
| 1 mM | 1.00 mL | 4.99 mL | 9.97 mL |
| 5 mM | 0.20 mL | 1.00 mL | 1.99 mL |
| 10 mM | 0.10 mL | 0.50 mL | 1.00 mL |
| 50 mM | 0.02 mL | 0.10 mL | 0.20 mL |
Calculators
Background References
References are publications that support the biological activity of the product. See our Citations tab to view 18 publications citing the usage of this product.
- Zhou A comprehensive review of BET-targeting PROTACs for cancer therapy. Bioorg.Med.Chem. 2022 PMID: 36202064
- Gadd Structural basis of PROTAC cooperative recognition for selective protein degradation. Nat.Chem.Biol. 2017 PMID: 28288108
- Wurz A " click chemistry platform" for the rapid synthesis of bispecific molecules for inducing protein degradation. J.Med.Chem. 2017 PMID: 28378579
- Zengerle Selective small molecule induced degradation of the BET bromodomain protein BRD4. ACS.Chem.Biol 2015 PMID: 26035625
Product Documents for MZ 1
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Product Specific Notices for MZ 1
For research use only
Citations for MZ 1
Customer Reviews for MZ 1
There are currently no reviews for this product. Be the first to review MZ 1 and earn rewards!
Have you used MZ 1?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review