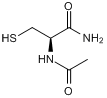
N-Acetylcysteine amide
Tocris Bioscience | Catalog # 5619

Key Product Details
Description
Product Description
N-Acetylcysteine amide is a glutathione (GSH) precursor and cell-permeable antioxidant. N-Acetylcysteine amide replenishes intracellular Glutathione (GSH). N-Acetylcysteine amide directly reduces intracellular Glutathione disulfide (GSSG) to GSH without glutathione peroxidase. N-Acetylcysteine amide has anti-inflammatory activity through regulation of activation of NF-κB and HIF-1α, as well as modulation of reactive oxygen species. N-Acetylcysteine amide improves neuronal mitochondrial bioenergetics, reduces tissue damage and enhances functional recovery following spinal cord injury in rats. N-Acetylcysteine amide also enhances behavioral recovery in rats following traumatic brain injury. Neuroprotective. N-Acetylcysteine amide can be used in a small molecule cocktail to generate 3D culture of lung alveolar cells (see protocol below).For more information about how N-Acetylcysteine amide may be used, see our protocol: 3D Culture of Lung Alveolar Cells
Product Specifications for N-Acetylcysteine amide
Molecular Weight
Formula
Storage
Chemical Name
CAS Number
PubChem ID
InChI Key
SMILES
The technical data provided above is for guidance only. For batch specific data refer to the Certificate of Analysis.
Solubility
| Solvent | Max Conc. mg/mL | Max Conc. mM | |
|---|---|---|---|
| Solubility | |||
| water | 16.22 | 100 | |
| DMSO | 16.22 | 100 |
Preparing Stock Solutions for N-Acetylcysteine amide
The following data is based on the product molecular weight 162.21.
Batch specific molecular weights may vary from batch to batch due to the degree of hydration, which all affect the solvent volumes required to prepare stock solutions.
| Concentration / Solvent Volume / Mass | 1 mg | 5 mg | 10 mg |
|---|---|---|---|
| 1 mM | 6.16 mL | 30.82 mL | 61.65 mL |
| 5 mM | 1.23 mL | 6.16 mL | 12.33 mL |
| 10 mM | 0.62 mL | 3.08 mL | 6.16 mL |
| 50 mM | 0.12 mL | 0.62 mL | 1.23 mL |
Calculators
Background References
References are publications that support the biological activity of the product. See our Citations tab to view 17 publications citing the usage of this product.
- Pandya N-acetylcysteine amide confers neuroprotection, improves bioenergetics and behavioral outcome following TBI. Exp.Neurol. 2014 PMID: 24792639
- Tu The anti-inflammatory and anti-oxidant mechanisms of the Keap1/Nrf2/ARE signaling pathway in chronic diseases. Aging Dis 2019 PMID: 31165007
- Patel N-acetylcysteine amide preserves mitochondrial bioenergetics and improves functional recovery following spinal trauma. Exp.Neurol. 2014 PMID: 24805071
Product Documents for N-Acetylcysteine amide
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Product Specific Notices for N-Acetylcysteine amide
For research use only
Citations for N-Acetylcysteine amide
Customer Reviews for N-Acetylcysteine amide (1)
Have you used N-Acetylcysteine amide?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
-
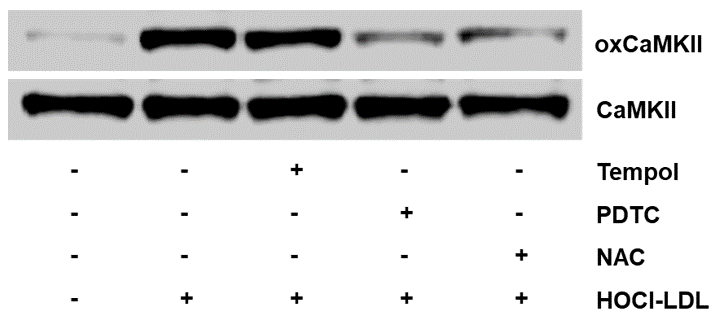
Species: MouseAssay Type: In VitroCell Line/Tissue: CardiomyocytesVerified Customer | Posted 10/22/2018HL-1 cardiomyocytes were pre-treated with Pyrrolidinedithiocarbamate (PDTC, 1 mM), Tempol (1 mM) or N-Acetylcysteine (NAC, 1 mM) for 30 min and stimulated with HOCl-LDL (200 µg/ml) for 2 h to follow CaMKII oxidation using Western blot.
There are no reviews that match your criteria.