Recombinant Human Active MAP3K8/Tpl2/COT Protein, CF
R&D Systems | Catalog # 4586-KS

Loading...
Key Product Details
- R&D Systems Sf 9 (baculovirus)-derived Recombinant Human Active MAP3K8/Tpl2/COT Protein (4586-KS)
- Quality control testing to verify active proteins with lot specific assays by in-house scientists
- All R&D Systems proteins are covered with a 100% guarantee
Source
Sf 9 (baculovirus)
Accession Number
Applications
Bioactivity
Loading...
Product Specifications
Source
Spodoptera frugiperda, Sf 9 (baculovirus)-derived human MAP3K8/Tpl2/COT protein
aa 30-397
aa 30-397
Purity
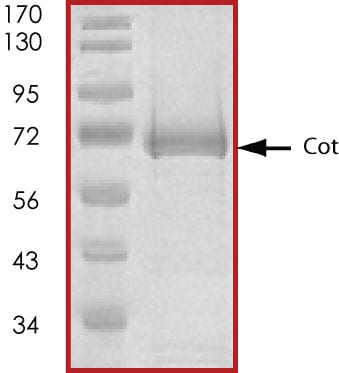
>75%, by SDS-PAGE under reducing conditions and visualized by Colloidal Coomassie® Blue stain at 5 μg per lane.
N-terminal Sequence Analysis
Using an N-terminal GST tag
SDS-PAGE
70 kDa
Activity
The specific activity of COT is typically 860-1164 nmol/min/mg using MEK1 and ERK1 substrates and a myelin basic protein (MBP) substrate.
Scientific Data Images for Recombinant Human Active MAP3K8/Tpl2/COT Protein, CF
Recombinant Human Active MAP3K8/Tpl2/COT Protein SDS-PAGE
The approximate molecular weight is 70 kDa and the purity is > 75%.Formulation, Preparation, and Storage
4586-KS
| Formulation | Supplied in 50 mM Tris-HCl (pH 7.5), 150 mM NaCl, 10 mM Glutathione, 0.1 mM EDTA, 0.25 mM DTT, 0.1 mM PMSF, and 25% Glycerol. |
| Shipping | The product is shipped with dry ice or equivalent. Upon receipt, store it immediately at the temperature recommended below. |
| Stability & Storage | This product is stable at ≤ ‑70 °C for up to 1 year from the date of receipt. For optimal storage, aliquot into smaller quantities after centrifugation and store at recommended temperature. Avoid repeated freeze-thaw cycles. |
Background: MAP3K8/Tpl2/COT
References
- Channavajhala, P.L. et al. (2003) J. Biol. Chem. 278:47089.
- Rodriguez, C. et al. (2006) Cell Signal. 18:1376.
Long Name
Mitogen-activated Protein Kinase Kinase Kinase 8
Alternate Names
COT, EST, ESTF, MAP3K8, Tpl-2
Gene Symbol
MAP3K8
UniProt
Additional MAP3K8/Tpl2/COT Products
Product Documents for Recombinant Human Active MAP3K8/Tpl2/COT Protein, CF
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Recombinant Human Active MAP3K8/Tpl2/COT Protein, CF
For research use only
Related Research Areas
Customer Reviews for Recombinant Human Active MAP3K8/Tpl2/COT Protein, CF
There are currently no reviews for this product. Be the first to review Recombinant Human Active MAP3K8/Tpl2/COT Protein, CF and earn rewards!
Have you used Recombinant Human Active MAP3K8/Tpl2/COT Protein, CF?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
View specific protocols for Recombinant Human Active MAP3K8/Tpl2/COT Protein, CF (4586-KS):
Materials
- Active Kinase - Active COT (0.1 μg/μL) diluted with Kinase Dilution Buffer III and assayed as outlined in Sample Activity Plot. Note: These are suggested working dilutions and it is recommended that the researcher perform a serial dilution of Active COT for optimal results.
- Kinase Assay Buffer I - 25 mM MOPS, pH 7.2, 12.5 mM beta -glycerolphosphate, 25 mM MgCl2, 5 mM EGTA, 2 mM EDTA. Add 0.25 mM DTT to the Kinase Assay Buffer prior to use.
- Kinase Dilution Buffer III - Kinase Assay Buffer I diluted at a 1:4 ratio (5X dilution) with a 50 ng/μL BSA solution.
- 10 mM ATP Stock Solution - Prepare ATP Stock Solution by dissolving 55 mg of ATP in 10 mL of Kinase Assay Buffer I. Store 200 μL aliquots at ≤ -20 °C.
- [33P]-ATP Assay Cocktail - Prepare 250 μM [33P]-ATP Assay Cocktail in a designated radioactive work area by combining 150 μL of 10 mM ATP Stock Solution, 100 μL of [33P]-ATP (1 mCi/100 μL), and 5.75 mL of Kinase Assay Buffer I. Store 1 mL aliquots at ≤ -20 °C.
- Substrate - Inactive MEK1 and ERK1 were activated in a coupled reaction. Myelin Basic Protein (MBP) diluted in distilled or deionized water to a final concentration of 1 mg/mL was subsequently used as a substrate for the activated ERK1.
- Thaw the Active COT, Kinase Assay Buffer I, and inactive MEK1 and ERK1 on ice. In a pre-cooled microfuge tube, add the following reaction components bringing the initial reaction volume up to 20 μL:
a. Diluted Active COT: 10 μL
b. Inactive MEK1 (0.2 μg/μL): 2 μL
c. Inactive ERK1 (0.2 μg/μL): 3 μL
d. Kinase Dilution Buffer: 5 μL - Start the reaction with the addition of 5 μL ATP (250 μM) and incubate in a water bath at 30 °C for 25 minutes.
- After the 25 minute incubation, remove 5 μL and add to the following reaction components bringing the initial reaction volume up to 20 μL:
a. Reaction Mixture: 5 μL
b. Distilled water (on ice): 10 μL
c. MBP Substrate (1 mg/mL; on ice): 5 μL - Set up the blank control as outlined in Step 3, excluding the addition of the substrate. Replace the substrate with an equal volume of distilled water.
- Initiate the reaction with the addition of 5 μL [33P]-ATP Assay Cocktail, bringing the final volume up to 25 μL and incubate the mixture in a water bath at 30 °C for 15 minutes.
- After the 15 minute incubation, terminate the reaction by spotting 20 μL of the reaction mixture onto individual pre-cut strips of phosphocellulose P81 paper.
- Air dry the pre-cut P81 strip and sequentially wash in a 1% phosphoric acid solution (add 10 mL of phosphoric acid to 990 mL of distilled or deionized water) with constant gentle stirring. It is recommended that the strips be washed a total of three times for approximately 10 minutes each.
- Count the radioactivity on the P81 paper in the presence of scintillation fluid in a scintillation counter.
- Determine the corrected cpm by subtracting the blank control value (see step 4) for each sample and calculate the kinase specific activity as outlined below.
Calculation of [33P]-ATP Specific Activity (SA) (cpm/pmol)
Specific Activity (SA) = cpm for 5 μL [33P]-ATP/pmole of ATP (in 5 μL of a 250 μM ATP stock solution; i.e. 1250 pmol)
Calculation of Kinase Specific Activity (SA) (pmol/minutes/μg or nmol/minutes/mg)
Corrected cpm from reaction / [(SA of 33P-ATP in cpm/pmol) x (Reaction time in minutes) x (Enzyme amount in μg or mg)] x [(Reaction volume) / (Spot Volume)]
Loading...
