Cathepsin L is a lysosomal cysteine protease expressed in most eukaryotic cells. Cathepsin L is known to hydrolyze a number of proteins, including the proform of urokinase-type plasminogen activator, which is activated by Cathepsin L cleavage (1). Cathepsin L has also been shown to proteolytically inactivate alpha 1-antitrypsin and secretory leucoprotease inhibitor, two major protease inhibitors of the respiratory tract (2). These observations, combined with the demonstration of increased Cathepsin L activity in the epithelial lining fluid of the lungs of emphysema patients, have led to the suggestion that the enzyme may be involved in the progression of this disease. Cathepsin L has also been identified as a major excreted protein of transformed fibroblasts, indicating the enzyme could be involved in malignant tumor growth (3). Human Cathepsin L activity is greatest under mildly acidic conditions, from pH 4.5 - 6.5. The stability of the enzyme decreases at higher pH values.
Recombinant Human Cathepsin L Protein, CF
R&D Systems | Catalog # 952-CY


Key Product Details
- R&D Systems NS0-derived Recombinant Human Cathepsin L Protein (952-CY)
- Quality control testing to verify active proteins with lot specific assays by in-house scientists
- All R&D Systems proteins are covered with a 100% guarantee
Source
Accession Number
Structure / Form
Applications
Product Specifications
Source
Glu113-Val333 & Ala114-Val333, both with a C-terminal 6-His tag
Purity
Endotoxin Level
N-terminal Sequence Analysis
Predicted Molecular Mass
SDS-PAGE
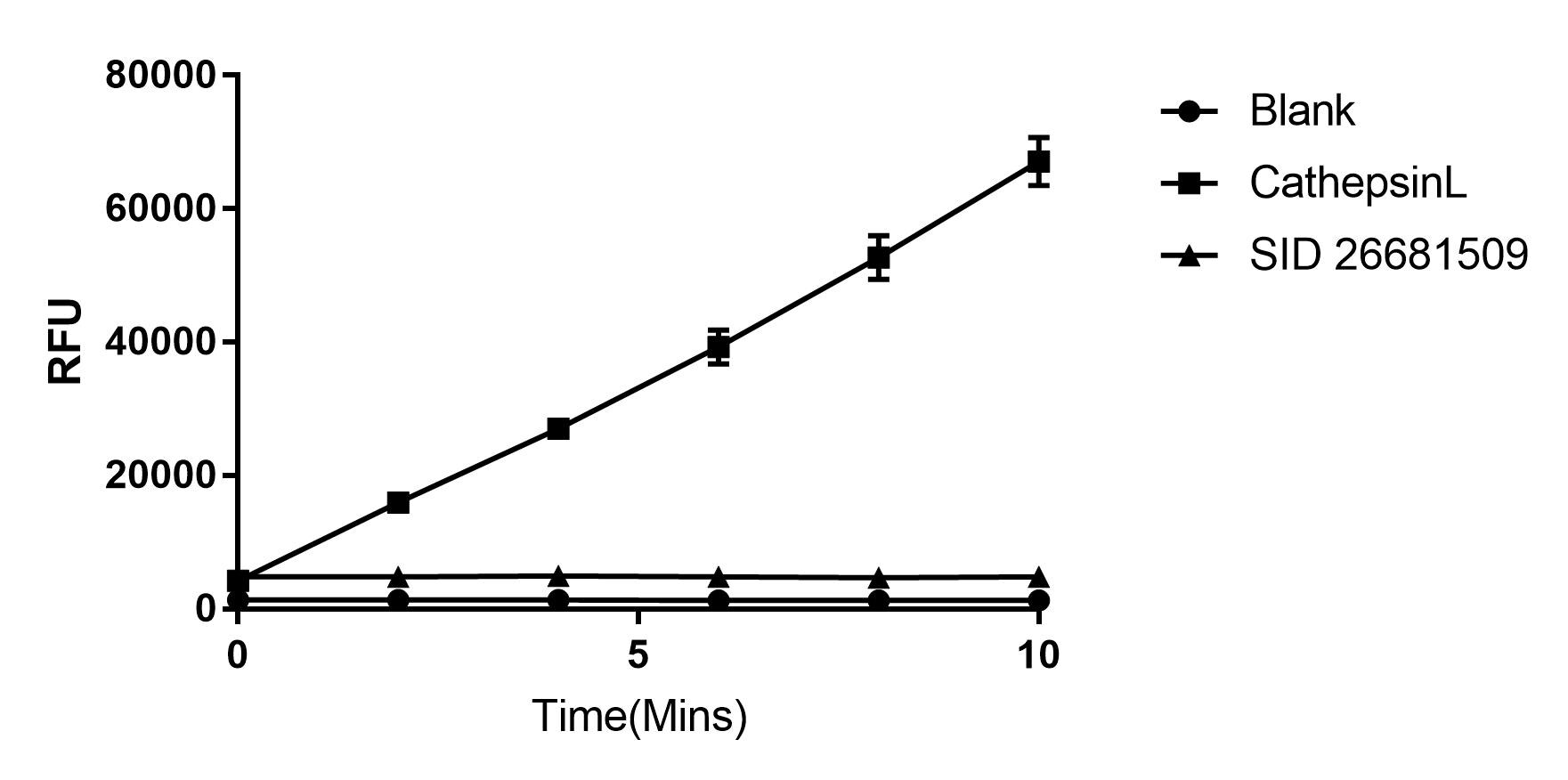
Activity
The specific activity is >25,000 pmol/min/µg, as measured under the described conditions.
Reviewed Applications
Read 2 reviews rated 5 using 952-CY in the following applications:
Formulation, Preparation, and Storage
952-CY
| Formulation | Supplied as a 0.2 μm filtered solution in Sodium Acetate and NaCl. |
| Shipping | The product is shipped with dry ice or equivalent. Upon receipt, store it immediately at the temperature recommended below. |
| Stability & Storage | Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
|
Background: Cathepsin L
References
- Goretzki, L. et al. (1992) FEBS Lett. 297:112.
- Taggart, C.C. et al. (2001) J. Biol. Chem. 276:33345.
- Gottesman, M.M. and F. Cabral (1981) Biochemistry 20:1659.
Alternate Names
Gene Symbol
UniProt
Additional Cathepsin L Products
Product Documents for Recombinant Human Cathepsin L Protein, CF
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Recombinant Human Cathepsin L Protein, CF
For research use only
Related Research Areas
Citations for Recombinant Human Cathepsin L Protein, CF
Customer Reviews for Recombinant Human Cathepsin L Protein, CF (2)
Have you used Recombinant Human Cathepsin L Protein, CF?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
-
Application: Enzymatic activity in vitroVerified Customer | Posted 09/23/2021
-
Application: Enzymatic activity in vitroVerified Customer | Posted 02/21/2017
There are no reviews that match your criteria.
Protocols
View specific protocols for Recombinant Human Cathepsin L Protein, CF (952-CY):
- Assay Buffer: 50 mM MES, 5 mM DTT, 1 mM EDTA, 0.005% (w/v) Brij-35, pH 6.0
- Recombinant Human Cathepsin L (rhCathepsin L) (Catalog # 952-CY)
- Fluorogenic Peptide Substrate VII: Z-Leu-Arg-AMC (Catalog # ES008)
- F16 Black Maxisorp Plate (Nunc, Catalog # 475515)
- Fluorescent Plate Reader (Model: SpectraMax Gemini EM by Molecular Devices) or equivalent
- Dilute rhCathepsin L to 40 µg/mL in Assay Buffer.
- Incubate diluted rhCathepsin L on ice for 15 minutes.
- Dilute incubated 40 µg/mL rhCathepsin L to 0.02 ng/µL in Assay Buffer.
- Dilute Substrate to 80 µM in Assay Buffer.
- Load 50 µL of 0.02 ng/µL rhCathepsin L into a black well plate, and start the reaction by adding 50 µL of 80 µM Substrate. Include a Substrate Blank containing 50 µL Assay Buffer and 50 µL of 80 µM Substrate without any rhCathepsin L.
- Read at excitation and emission wavelengths of 380 nm and 460 nm (top read), respectively, in kinetic mode for 5 minutes.
- Calculate specific activity:
|
Specific Activity (pmol/min/µg) = |
Adjusted Vmax* (RFU/min) x Conversion Factor** (pmol/RFU) |
| amount of enzyme (µg) |
*Adjusted for Substrate Blank
**Derived using calibration standard 7-Amino, 4-Methyl Coumarin (AMC) (Sigma, Catalog # A-9891).
Per Well:
- rhCathepsin L: 0.001 µg
- Substrate: 40 µM
FAQs for Recombinant Human Cathepsin L Protein, CF
-
Q: When the activity of Recombinant Human Cathepsin L Protein (Catalog # 952-CY) is measured, does it need to be incubated diluted on ice for 15 minutes as stated in the datasheet?
A: Yes, it is recommended to follow the protocol stated in the datasheet as closely as possible. The 15-minute incubation in assay buffer on ice that is mentioned in the protocol is used to bring the Cysteine active site into a reduced state, which is required for the enzyme to be optimally functional.