HGF, also known as scatter factor and hepatopoietin A, is a pleiotropic protein in the plasminogen subfamily of S1 peptidases. It is a multidomain molecule that includes an N-terminal PAN/APPLE‑like domain, four Kringle domains, and a serine proteinase-like domain that has no detectable protease activity (1‑5). Human HGF is secreted as an inactive 728 amino acid (aa) single chain propeptide. It is cleaved after the fourth Kringle domain by a serine protease to form bioactive
disulfide‑linked HGF with a 60 kDa alpha and 30 kDa beta chain. Alternate splicing generates human HGF isoforms that lack the proteinase‑like domain and different numbers of the Kringle domains. Human HGF shares 91%‑94% aa sequence identity with bovine, canine, feline, mouse, and rat HGF. HGF binds heparan-sulfate proteoglycans and the widely expressed receptor tyrosine kinase, HGF R/c-MET (6, 7). HGF-dependent c-MET activation is implicated in the development of many human cancers (8). HGF regulates epithelial morphogenesis by inducing cell scattering and branching tubulogenesis (9, 10). HGF induces the up‑regulation of integrin alpha 2 beta 1 in epithelial cells by a selective increase in alpha 2 gene transcription (11). This integrin serves as a collagen I receptor, and its blockade disrupts epithelial cell branching tubulogenesis (11, 12). HGF can also alter epithelium morphology by the induction of nectin‑1 alpha ectodomain shedding, an adhesion protein component of adherens junctions (13). In the thyroid, HGF induces the proliferation, motility, and loss of differentiation markers of thyrocytes and inhibits TSH‑stimulated iodine uptake (14). HGF promotes the motility of cardiac stem cells in damaged myocardium (15).
GMP
Recombinant Human HGF (NS0-expressed) GMP Protein, CF
R&D Systems | Catalog # 294-GMP

Loading...
Key Product Details
Source
NS0
Accession Number
Structure / Form
Disulfide-linked heterodimer
Applications
Bioactivity
Loading...
Product Specifications
Source
Mouse myeloma cell line, NS0-derived human HGF protein
Gln32-Ser728 (Asp384Asn, Asp416Asn, Leu622Ser, His645Arg, Val678Ile)
Manufactured and tested under cGMP guidelines.
Gln32-Ser728 (Asp384Asn, Asp416Asn, Leu622Ser, His645Arg, Val678Ile)
Manufactured and tested under cGMP guidelines.
Purity
>95%, by SDS-PAGE with silver staining, under reducing conditions.
Endotoxin Level
<0.10 EU per 1 μg of the protein by the LAL method.
N-terminal Sequence Analysis
Amino acid sequencing of the alpha chain was blocked, suggesting it is consistent with Gln32
Gln32-Arg-Lys-Arg-Arg-Asn-Thr-Ile-His-Glu ( alpha -chain)
Val495-Val-Asn-Gly-Ile-Pro-Thr-Arg-Thr-Asn ( beta -chain)
Gln32-Arg-Lys-Arg-Arg-Asn-Thr-Ile-His-Glu ( alpha -chain)
Val495-Val-Asn-Gly-Ile-Pro-Thr-Arg-Thr-Asn ( beta -chain)
Predicted Molecular Mass
alpha chain: 53.7 kDa
beta chain: 26 kDa
beta chain: 26 kDa
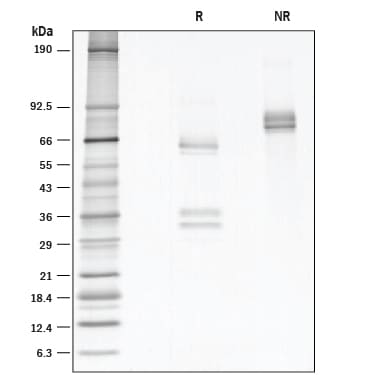
SDS-PAGE
71-85 kDa, non-reducing conditions
56-65 kDa, and 30-40 kDa, reducing conditions
56-65 kDa, and 30-40 kDa, reducing conditions
Activity
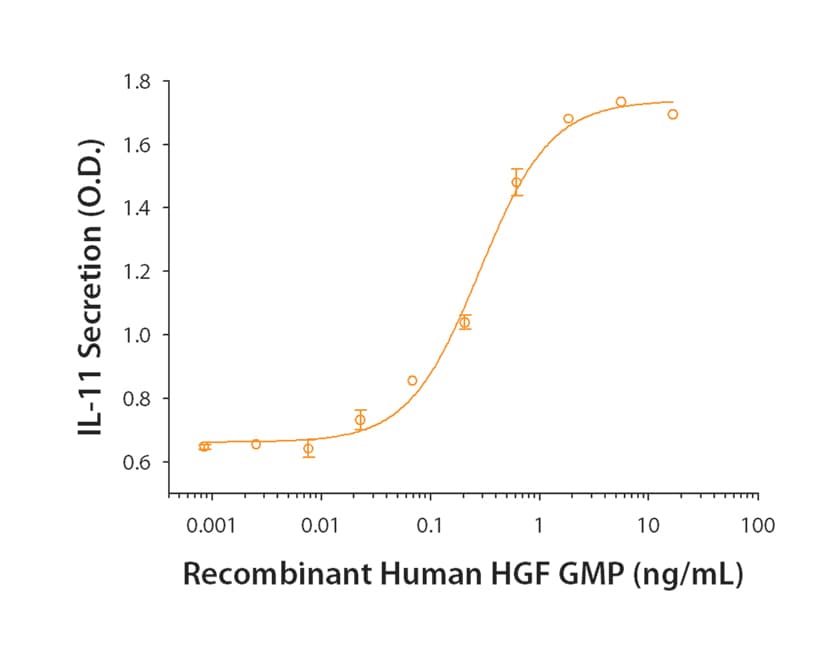
Measured by its ability to induce IL-11 secretion by Saos‑2 human osteosarcoma cells. Hjertner, O. et al. (1999) Blood 94:3883.
The ED50 for this effect is equal or less than 4.00 ng/mL.
The specific activity of recombinant human HGF is >1.0 x 106 IU/mg, which is calibrated against the human HGF WHO International Standard (NIBSC code: 96/564).
The ED50 for this effect is equal or less than 4.00 ng/mL.
The specific activity of recombinant human HGF is >1.0 x 106 IU/mg, which is calibrated against the human HGF WHO International Standard (NIBSC code: 96/564).
Host Cell Protein
<5 ng per µg of protein when tested by ELISA.
Mycoplasma
Negative for Mycoplasma.
Scientific Data Images for Recombinant Human HGF (NS0-expressed) GMP Protein, CF
Recombinant Human HGF (NS0-expressed) GMP Protein Bioactivity
Recombinant Human HGF GMP induces IL-11 secretion by Saos‑2 human osteosarcoma cells. The ED50 for this effect is equal or less than 4.00 ng/mL.Recombinant Human HGF (NS0-expressed) GMP Protein SDS-PAGE
1 μg/lane of GMP-grade Recombinant Human HGF (Catalog # 294-GMP) was resolved with SDS-PAGE under reducing (R) and non-reducing (NR) conditions and visualized by silver staining, showing R bands at 30-40 and 56-65 kDa and NR bands at 71-85 kDa, respectively.Formulation, Preparation, and Storage
294-GMP
| Formulation | Lyophilized from a 0.2 μm filtered solution in PBS. |
| Reconstitution | Reconstitute at 100 μg/mL in PBS. |
| Shipping | The product is shipped with polar packs. Upon receipt, store it immediately at the temperature recommended below. |
| Stability & Storage | Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
|
Calculators
Background: HGF
References
- Karihaloo, A. et al. (2005) Nephron Exp. Nephrol. 100:e40.
- Hammond, D.E. et al. (2004) Curr. Top. Microbiol. Immunol. 286:21.
- Rosario, M. and W. Birchmeier (2004) Dev. Cell 7:3.
- Lesk, A.M. and W.D. Fordham (1996) J. Mol. Biol. 258:501.
- Nakamura, T. et al. (1989) Nature 342:440.
- Mizuno, K., et al. (1994) J. Biol. Chem. 269:1131.
- Gheradi, E. et al. (2003) Proc. Natl. Acad. Sci. 100:12039.
- Corso, S. et al. (2005) Trends Mol. Med. 11:284.
- Maeshima, A. et al. (2000) Kid. Int. 58:1511.
- Montesano, R. et al. (1991) Cell 67:901.
- Chiu, S-J. et al. (2002) J. Biomed. Sci. 9:261.
- Saelman, E.U.M. et al. (1995) J. Cell Sci. 108:3531.
- Tanaka, Y. et al. (2002) Biochem. Biophys. Res. Commun. 299:472.
- Mineo, R. et al. (1994) Endocrinology 145:4355.
- Urbanek, K. et al. (2005) Circ. Res. 97:663.
Long Name
Hepatocyte Growth Factor
Alternate Names
DFNB39, F-TCF, Hepatopoietin A, HGFB, HPTA, SF
Entrez Gene IDs
Gene Symbol
HGF
UniProt
Additional HGF Products
Product Documents for Recombinant Human HGF (NS0-expressed) GMP Protein, CF
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Manufacturing Specifications
GMP ProteinsR&D Systems, a Bio-Techne Brand's GMP proteins are produced according to relevant sections of the following documents: USP Chapter 1043, Ancillary Materials for Cell, Gene and Tissue-Engineered Products and Eu. Ph. 5.2.12, Raw Materials of Biological Origin for the Production of Cell-based and Gene Therapy Medicinal Products.
R&D Systems' quality focus includes:
- Designed, manufactured and tested under an ISO 9001:2015 and ISO 13485:2016 certified quality system
- Documented and controlled manufacturing process
- Control of documentation and process changes by QA
- Personnel training programs
- Raw material inspection and vendor qualification/monitoring program
- Validated equipment, processes and test methods
- Equipment calibration and maintenance schedules using a Regulatory Asset Manager
- Facility/Utilities maintenance, contamination controls, safety and pest control programs
- Material review process for variances
- Robust product stability program following relevant ICH guidelines
- N-terminal amino acid analysis
- SDS-PAGE purity analysis
- Molecular weight analysis via mass spectrometry
- Endotoxin assessment per USP <85> and Ph. Eur. 2.6.14 guidelines
- Bioassay analysis
- Microbial testing per USP <71> and Ph. Eur. 2.6.1 guidelines
- Host cell protein assessment
- Host cell DNA assessment
- Mycoplasma assessment
Production records and facilities are available for examination by appropriate personnel on-site at R&D Systems in Minneapolis and St. Paul, Minnesota USA.
R&D Systems sells GMP grade products for preclinical or clinical ex vivo use. They are not for in vivo use. Please read the following End User Terms prior to using this product.
Product Specific Notices for Recombinant Human HGF (NS0-expressed) GMP Protein, CF
Full terms and conditions of sale can be found online in the Protein Sciences Segment T&Cs at: Terms & Conditions.
For preclinical, or clinical ex vivo use
Related Research Areas
Customer Reviews for Recombinant Human HGF (NS0-expressed) GMP Protein, CF
There are currently no reviews for this product. Be the first to review Recombinant Human HGF (NS0-expressed) GMP Protein, CF and earn rewards!
Have you used Recombinant Human HGF (NS0-expressed) GMP Protein, CF?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Loading...
Associated Pathways