Recombinant Human Lipoprotein Lipase/LPL Protein, CF
R&D Systems | Catalog # 9888-LL

Key Product Details
- R&D Systems CHO-derived Recombinant Human Lipoprotein Lipase/LPL Protein (9888-LL)
- Quality control testing to verify active proteins with lot specific assays by in-house scientists
- All R&D Systems proteins are covered with a 100% guarantee
Source
Accession Number
Applications
Product Specifications
Source
Ala28-Gly475 with a C-terminal 6-His tag
Endotoxin Level
N-terminal Sequence Analysis
Predicted Molecular Mass
SDS-PAGE
Activity
The specific activity is >2,500 pmol/min/μg, as measured under the described conditions.
Scientific Data Images for Recombinant Human Lipoprotein Lipase/LPL Protein, CF
Recombinant Human Lipoprotein Lipase/LPL Protein Bioactivity
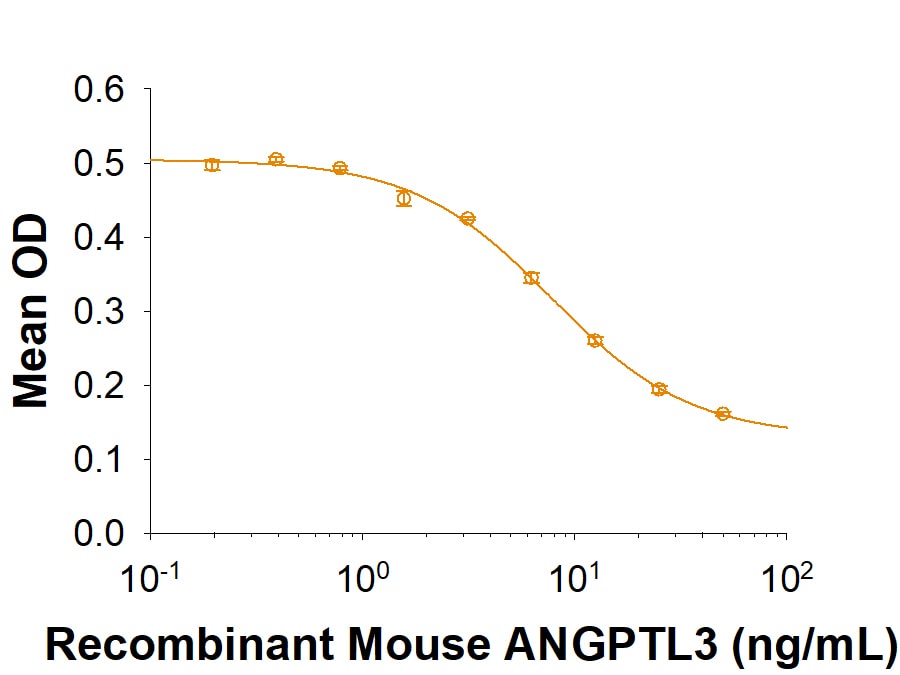
Recombinant Human LPL activity can be inhibited by Recombinant Mouse ANGPTL3. Recombinant Mouse ANGPTL3 (Catalog # 9899-AN) dose dependently inhibits Recombinant Human LPL (Catalog # 9888-LL) activity with a ND50of 2-10 µg/mL.Recombinant Human Lipoprotein Lipase/LPL Protein Enzyme Activity
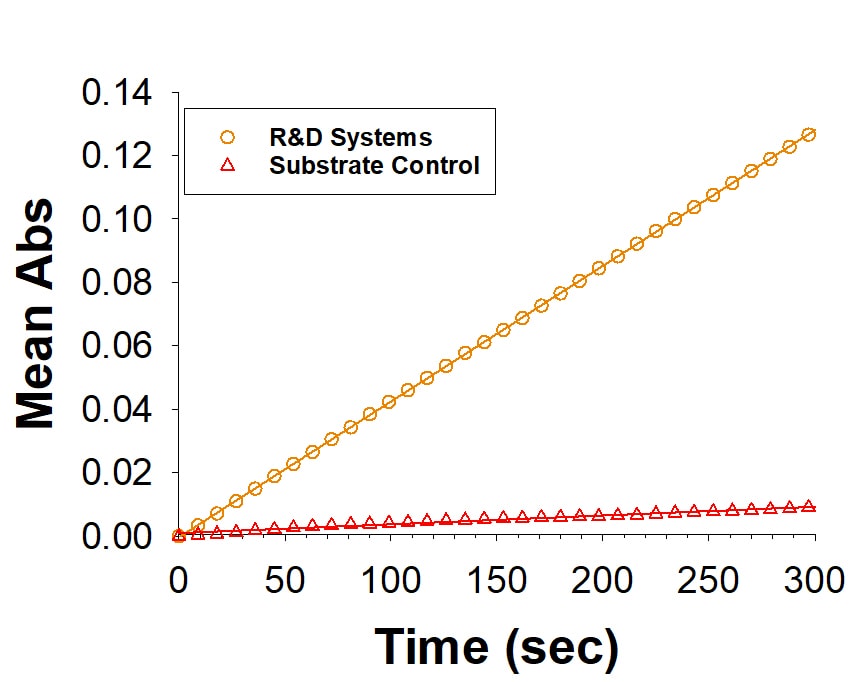
Recombinant Human Lipoprotein Lipase (Catalog # 9888-LL) is measured by its ability to hydrolyze 4-Nitrophenyl butyrate.Recombinant Human Lipoprotein Lipase/LPL Protein SDS-PAGE
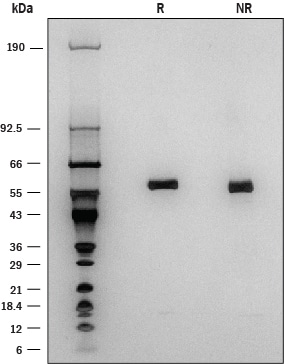
1 μg/lane of Recombinant Human Lipoprotein Lipase was resolved with SDS-PAGE under reducing (R) and non-reducing (NR) conditions and visualized by silver staining, showing a band at 60 kDa.Formulation, Preparation, and Storage
9888-LL
| Formulation | Supplied as a 0.2 μm filtered solution in Tris, NaCl, CHAPS and Glycerol. |
| Shipping | The product is shipped with dry ice or equivalent. Upon receipt, store it immediately at the temperature recommended below. |
| Stability & Storage | Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
|
Background: Lipoprotein Lipase/LPL
References
- Wang, H. and R. H. Eckel (2009) Am. J. Physiol. Endocrinol. Metab. 297:E271.
- Olivecrona, T.G. et al. (1985) J. Biol. Chem. 260:6888.
- Dugi, K. A. (1995) J. Biol. Chem. 270:25396.
- Keiper, T. et al. (2001) J. Lipid Res. 42:1180.
- Camps, L. et al. (1991) J. Lipid Res. 32:1877.
- Savonen, R. et al. (2015) J. Lipid Res. 56:588.
- Ping-Ping, H. et al. (2018) Clin Chim Acta 480:126.
- Takahashi, M. et al. (2013) J. Lipid Res. 54:1124.
- Qui, G. et al. (2007) J. Lipid Res. 48:385.
- Gagne, S. E. et al. (1999) Clin. Genet. 55:450.
- Jensen, M. K. et al. (2009) Am. Heart J. 157:384.
- Wang, C. et al. (2011) Thromb. Res. 128:e107.
- Zhang, W. S. et al. (2015) Int. J. Clin. Exp. Med. 8:9575.
- Ren, L. and X. Ren (2016) Neurosci. Lett. 619:73.
- Chan, L. Y. S. et al. (2002) Hum. Mutat. 20:232.
- Van Bockstaele, F. et al. (2007) Clin. Chem. 53:204.
- Kuemmerle, N.B. et al. (2011) Mol. Cancer Ther. 10:427.
- He, D. et al. (2016) J. Bioinform. Comput. Biol. 14:1650028.
- Takasu, S. et al. (2012) Biochem. Res. 2012:398697.
Alternate Names
Gene Symbol
UniProt
Additional Lipoprotein Lipase/LPL Products
Product Documents for Recombinant Human Lipoprotein Lipase/LPL Protein, CF
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Recombinant Human Lipoprotein Lipase/LPL Protein, CF
For research use only
Citations for Recombinant Human Lipoprotein Lipase/LPL Protein, CF
Customer Reviews for Recombinant Human Lipoprotein Lipase/LPL Protein, CF
There are currently no reviews for this product. Be the first to review Recombinant Human Lipoprotein Lipase/LPL Protein, CF and earn rewards!
Have you used Recombinant Human Lipoprotein Lipase/LPL Protein, CF?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
View specific protocols for Recombinant Human Lipoprotein Lipase/LPL Protein, CF (9888-LL):
- Assay Buffer: 50 mM Tris, pH 7.5
- Substrate Buffer: 50 mM Tris, 2% (v/v) Triton X-100, pH 7.5
- Recombinant Human LPL (rhLPL) (Catalog # 9888-LL)
- Substrate: 4-Nitrophenyl butyrate (Sigma, Catalog # N9876), 100 mM stock in acetone
- 96-well Clear Plate (Catalog # DY990)
- Plate Reader (Model: SpectraMax Plus by Molecular Devices) or equivalent
- Dilute rhLPL to 2 µg/mL in Assay Buffer.
- Load 100 µL of Assay Buffer to all wells.
- Add 50 μL of 2 µg/mL rhLPL to all wells.
- Dilute Substrate to 8 mM in Substrate Buffer.
- Add 50 μL of diluted Substrate to all wells. For Substrate Blank, load 150 μL of Assay Buffer followed by the addition of 50 μL of diluted Substrate.
- Read in kinetic mode for 5 minutes at an absorbance of 400 nm.
- Calculate specific activity:
Specific Activity (pmol/min/µg) = | Adjusted Vmax* (OD/min) x Conversion Factor** (pmol/OD) |
| amount of enzyme (µg) |
*Adjusted for Substrate Blank.
**Derived using calibration standard 4-Nitrophenol (4-NP) (Sigma, Catalog # 241326).
Note: the output of many spectrophotometers is in mOD.
Per Well:
- rhLPL: 0.1 μg
- Substrate: 2 mM
Associated Pathways