Recombinant Human PON1 His-tag Fc Chimera Protein, CF
R&D Systems | Catalog # 10175-PO

Key Product Details
- R&D Systems HEK293-derived Recombinant Human PON1 His-tag Fc Chimera Protein (10175-PO)
- Quality control testing to verify active proteins with lot specific assays by in-house scientists
- All R&D Systems proteins are covered with a 100% guarantee
Source
Accession Number
Applications
Product Specifications
Source
Leu16-Leu355
with an N-terminal 6-His tag and a C-terminal Fc tag
Purity
Endotoxin Level
N-terminal Sequence Analysis
Predicted Molecular Mass
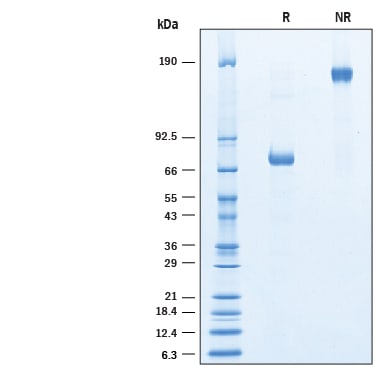
SDS-PAGE
Activity
The specific activity is >5000 pmol/min/μg, as measured under the described conditions.
Scientific Data Images for Recombinant Human PON1 His-tag Fc Chimera Protein, CF
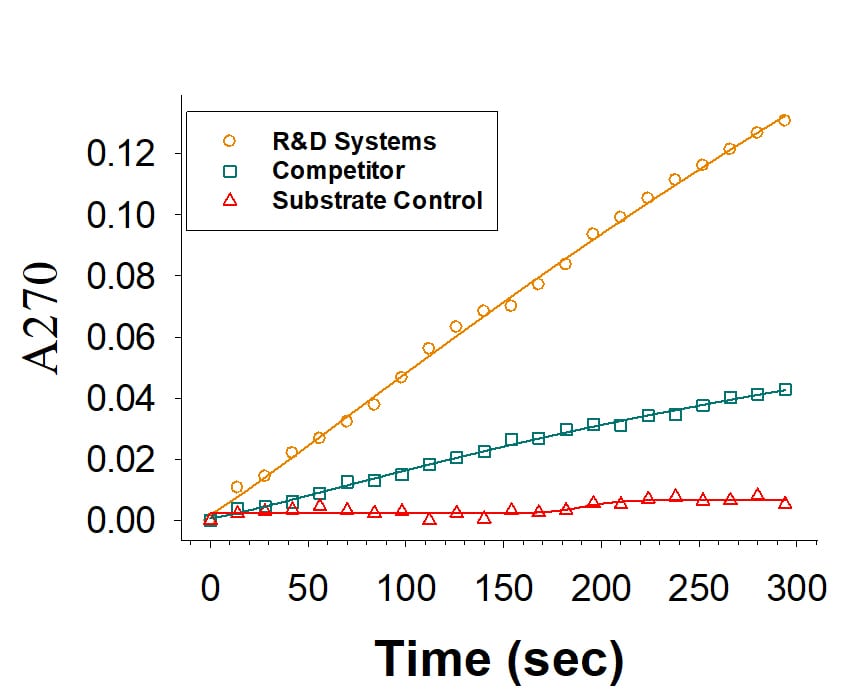
Recombinant Human PON1 His-tag Fc Chimera Protein Enzyme Activity
Recombinant Human PON-1 His-tag Fc Chimera (Catalog # 10175-PO) is measured by its ability to hydrolyze phenyl acetate. The activity (orange) is over 3-fold higher than the competitor's PON-1 (green).Recombinant Human PON1 His-tag Fc Chimera Protein SDS-PAGE
2 μg/lane of Recombinant Human PON1 His-tag Fc Chimera was resolved with SDS-PAGE under reducing (R) and non-reducing (NR) conditions and visualized by Coomassie® blue staining, showing a band at ~74 kDa under reducing conditions.Formulation, Preparation, and Storage
10175-PO
| Formulation | Supplied as a 0.2 μm filtered solution in Tris, CaCl2, NaCl, and Glycerol. |
| Shipping | The product is shipped with polar packs. Upon receipt, store it immediately at the temperature recommended below. |
| Stability & Storage | Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
|
Background: PON1
References
- Dragonov, D. I. et al. (2005) Lipid Res. 46:1239.
- Khersonsky, O. et al. (2005) Biochemistry. 44:6371.
- Ben-David, M. et al. (2012) J. Mol. Biol. 418:181.
- Harel, M. et al. (2004) Nature Struct. Mol. Biol. 11:412.
- Mackness, B. et al. (1998) Gen. Pharmac. 31:329.
- Aviram, M. et al. (2004) Free Radic. Biol. Med. 37:1304.
- Rozenberg, O. et al. (2008) Free Radic. Biol. Med. 44:1951.
- Koren-Gluzer, M. et al. (2011) Atherosclerosis. 219:532.
- Goswami, N. et al. (2009) Clin. Chim. Acta. 410:1.
- Mahrooz, A. (2016) Curr. Clin. Pharmacol. 11:259.
- Berrougui, H. et al. (2012) Free Radic. Biol. Med. 52:1372.
- Aharoni, S. et al. (2013) Atherosclerosis 228:353.
- Tavori, H. (2011) Free Rad. Biol. Med. 51:234.
- Wang, M. et al. (2012) DNA Cell. Biol. 31:975.
- Tang, W. H. W. et al. (2012) Arterioscler. Thromb. Vasc. Biol. 32:2803.
- Li, W. F. et al. (2000) Pharmacogenetics. 10:767.
- Shih, D. M. et al. (1998) Nature. 394:284.
- Ahmed, H. et al. (2017) Biomed. Pharmacother. 90:638.
- Gagliardi, S. et al. (2013) Neurotox. Res. 23:370.
Long Name
Alternate Names
Gene Symbol
UniProt
Additional PON1 Products
Product Documents for Recombinant Human PON1 His-tag Fc Chimera Protein, CF
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Recombinant Human PON1 His-tag Fc Chimera Protein, CF
For research use only
Related Research Areas
Customer Reviews for Recombinant Human PON1 His-tag Fc Chimera Protein, CF
There are currently no reviews for this product. Be the first to review Recombinant Human PON1 His-tag Fc Chimera Protein, CF and earn rewards!
Have you used Recombinant Human PON1 His-tag Fc Chimera Protein, CF?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
View specific protocols for Recombinant Human PON1 His-tag Fc Chimera Protein, CF (10175-PO):
- Assay Buffer: 50 mM Tris, 5 mM CaCl2, pH 8.0
- Deionized Water
- Recombinant Human PON1 His-tag Fc Chimera (rhPON1) (Catalog 10175-PO)
- Substrate: Phenyl Acetate (7.88 M) (Sigma, Catalog # 108723)
- UV Plate (Catalog # 3635)
- Plate Reader (Model: SpectraMax Plus by Molecular Devices) or equivalent
- Dilute rhPON1 to 10 µg/mL in Assay Buffer.
- Dilute Phenyl Acetate to 40 mM in deionized water.
- Load 50 µL of 10 µg/mL rhPON1 in a plate, and start the reaction by adding 50 µL of Substrate. Include a Substrate Blank containing 50 µL Assay Buffer and 50 µL of Substrate.
- Read plate at a wavelength of 270 nm (absorbance) in kinetic mode for 5 minutes.
- Calculate specific activity:
Specific Activity (pmol/min/µg) = | Adjusted Vmax* (OD/min) x well volume (L) x 1012 pmol/mol |
| ext. coeff** (M-1cm-1) x path corr.*** (cm) x amount of enzyme (µg) |
*Adjusted for Substrate Blank
**Using the extinction coefficient 1310 M-1cm-1
***Using the path correction 0.32 cm
Note: the output of many spectrophotometers is in mOD
Per Well:
- rhPON1: 0.5 µg
- Phenyl Acetate: 20 mM