Recombinant Human Renalase Protein, CF
R&D Systems | Catalog # 9887-RE

Loading...
Key Product Details
- R&D Systems E. coli-derived Recombinant Human Renalase Protein (9887-RE)
- Quality control testing to verify active proteins with lot specific assays by in-house scientists
- All R&D Systems proteins are covered with a 100% guarantee
Source
E. coli
Accession Number
Applications
Enzyme Activity
Loading...
Product Specifications
Source
E. coli-derived human Renalase protein
Ala2-Ile342
with substitution Glu37Asp, N-terminal Met and 6-His tag
Ala2-Ile342
with substitution Glu37Asp, N-terminal Met and 6-His tag
Purity
>95%, by SDS-PAGE visualized with Silver Staining and quantitative densitometry by Coomassie® Blue Staining.
Endotoxin Level
<1.0 EU per 1 μg of the protein by the LAL method.
N-terminal Sequence Analysis
Met
Predicted Molecular Mass
39 kDa
SDS-PAGE
39 kDa, reducing conditions
Activity
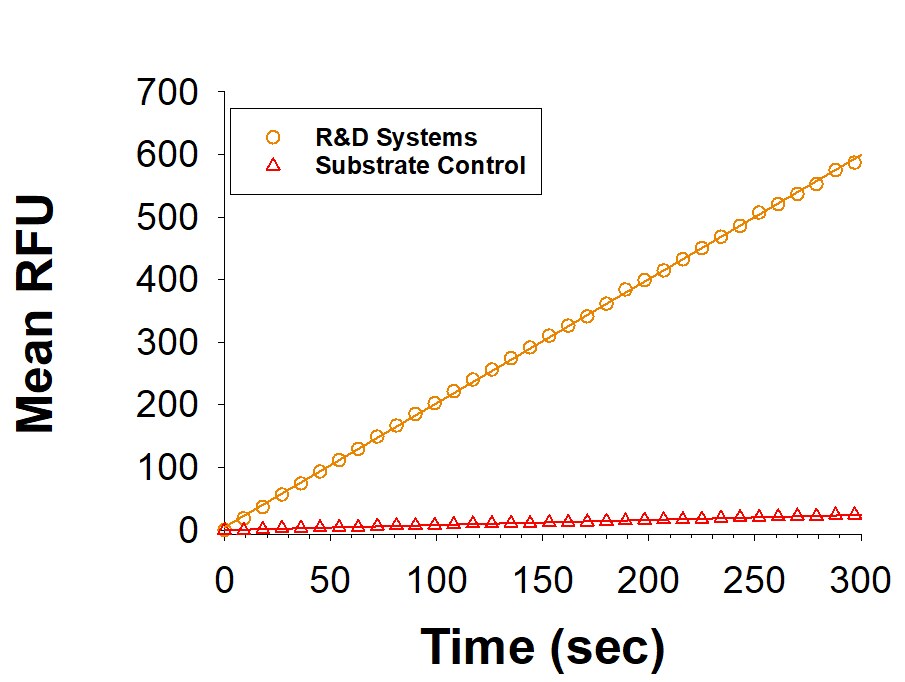
Measured by its ability to reduce Resazurin.
The specific activity is >3.5 pmol/min/μg, as measured under the described conditions.
The specific activity is >3.5 pmol/min/μg, as measured under the described conditions.
Scientific Data Images for Recombinant Human Renalase Protein, CF
Recombinant Human Renalase Protein Enzyme Activity
Recombinant Human Renalase (Catalog # 9887-RE) is measured by its ability to reduce Resazurin (Catalog # AR002).Formulation, Preparation, and Storage
9887-RE
| Formulation | Supplied as a 0.2 μm filtered solution in Tris, EDTA, DTT and Glycerol. |
| Shipping | The product is shipped with polar packs. Upon receipt, store it immediately at the temperature recommended below. |
| Stability & Storage | Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
|
Background: Renalase
References
- Xu, J. et al. (2005) J. Clin. Invest. 115:1275.
- Beaupre, B. A. et al. (2015) Biochemistry 54:795.
- Moran, G.R. et al. (2016) Biochim. Biophys. Acta 1864:177.
- Milani, M. et al. (2011) J. Mol. Biol. 411:463.
- Fedchenko, V. et al. (2016) Kidney Blood Press. Res. 41:593.
- Guo, X. et al. (2014) Curr. Opin. Nephrol. Hypertens. 23:513.
- Wu, Y. et al. (2011) Kidney International 79:853.
- Lee, H.T. et al. (2013) J. Am. Soc. Nephrol. 24:445.
- Buraczynska, M. et al. (2011) Neuromolecular Med. 13:321.
- Orlowska-Baranowska, E. et al. (2017) PLoS One 125:e0186729.
- Wang, L. et al. (2015) PLoS ONE 10:e0122932.
- Wang, L. et al. (2014) J. Am. Soc. Nephrol. 25:1226.
- Wang, Y. et al. (2017) J. Cell. Mol. Med. 21:1260.
Alternate Names
C10orf59, RNLS
Gene Symbol
RNLS
UniProt
Additional Renalase Products
Product Documents for Recombinant Human Renalase Protein, CF
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Recombinant Human Renalase Protein, CF
For research use only
Related Research Areas
Customer Reviews for Recombinant Human Renalase Protein, CF
There are currently no reviews for this product. Be the first to review Recombinant Human Renalase Protein, CF and earn rewards!
Have you used Recombinant Human Renalase Protein, CF?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
View specific protocols for Recombinant Human Renalase Protein, CF (9887-RE):
Materials
- Assay Buffer: 50 mM HEPES, 200 mM NaCl, 5 µM FAD (Flavin Adenine Dinucleotide), 0.05% Tween-20, pH 7.5
- Recombinant Human Renalase (rhRenalase) (Catalog # 9887-RE)
- beta -Nicotinamide adenine dinucleotide, reduced (NADH) (Sigma, Catalog # N8129), 20 mM stock in 0.1 M Sodium Borate, pH 9.0
- Resazurin (Catalog # AR002) (MW = 251.17 Da)
- F16 Black Maxisorp Plate (Nunc, Catalog # 475515)
- Fluorescent Plate Reader (Model: SpectraMax Gemini EM by Molecular Devices) or equivalent
- Dilute rhRenalase to 40 µg/mL in Assay Buffer.
- Prepare a Substrate Mixture containing 400 µM of NADH and 20 µM Resazurin in Assay Buffer. Mix well and use immediately.
- Load 50 µL of 40 µg/mL rhRenalase into a plate, and start reaction by adding 50 µL Substrate Mixture. Include a Substrate Blank containing 50 µL Assay Buffer and 50 µL Substrate Mixture.
- Read plate at excitation and emission wavelengths of 540 and 585 nm, respectively, in kinetic mode for five minutes.
- Calculate specific activity:
Specific Activity (pmol/min/µg) = | Adjusted Vmax* (RFU/min) x Conversion Factor** (pmol/RFU) |
| amount of enzyme (µg) |
*Adjusted for Substrate Blank.
**Derived using calibration standard Resorufin (Sigma, Catalog # R3257).
Per Well:
- rhRenalase: 2.0 µg
- NADH: 200 µM
- Resazurin: 10 µM
Loading...
