Recombinant Human Siglec-15 Fc Chimera Protein, CF
R&D Systems | Catalog # 9227-SL

Loading...
Key Product Details
- R&D Systems CHO-derived Recombinant Human Siglec-15 Fc Chimera Protein (9227-SL)
- Quality control testing to verify active proteins with lot specific assays by in-house scientists
- All R&D Systems proteins are covered with a 100% guarantee
Source
CHO
Accession Number
Structure / Form
Disulfide-linked homodimer
Applications
Bioactivity
Loading...
Product Specifications
Source
Chinese Hamster Ovary cell line, CHO-derived human Siglec-15 protein
| Human Siglec-15 (Phe20-Thr263) Accession # Q6ZMC9 |
IEGRMD | Human IgG1 (Pro100-Lys330) |
| N-terminus | C-terminus |
Purity
>90%, by SDS-PAGE visualized with Silver Staining and quantitative densitometry by Coomassie® Blue Staining.
Endotoxin Level
<0.10 EU per 1 μg of the protein by the LAL method.
N-terminal Sequence Analysis
Phe20
Predicted Molecular Mass
53 kDa
SDS-PAGE
53-64 kDa, reducing conditions
Activity
Measured by its ability to inhibit anti-CD3 antibody induced IL-2 or IFN-gamma secretion by human T cells.
The ED50 for this effect is 1-10 μg/mL.
The ED50 for this effect is 1-10 μg/mL.
Reviewed Applications
Read 1 review rated 5 using 9227-SL in the following applications:
Scientific Data Images for Recombinant Human Siglec-15 Fc Chimera Protein, CF
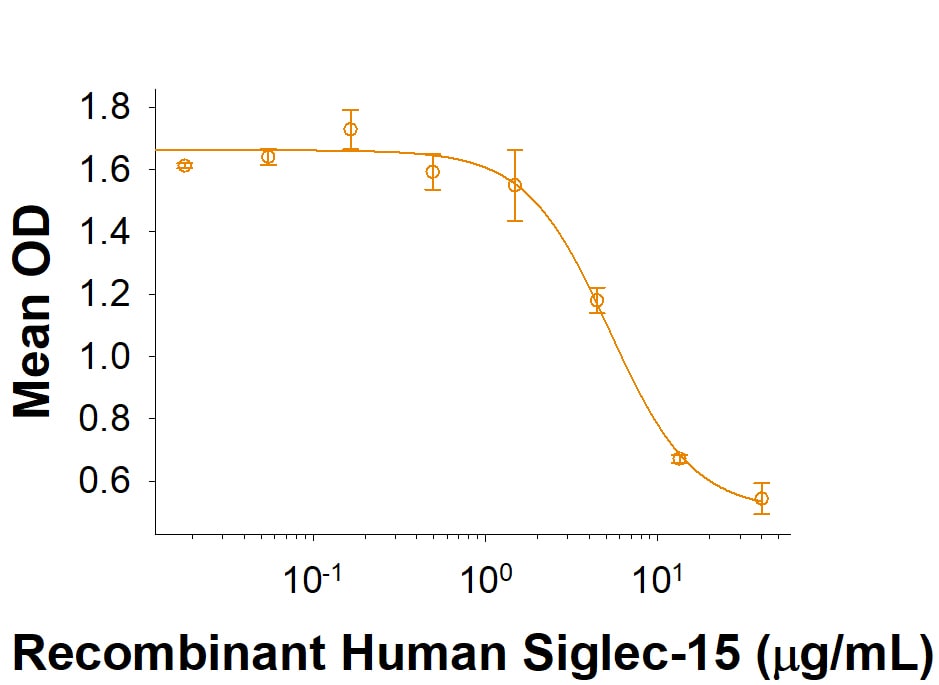
Recombinant Human Siglec-15 Fc Chimera Protein Bioactivity
Recombinant Human Siglec-15 Fc Chimera (Catalog # 9227-SL) inhibits anti-CD3 antibody induced IFN-gamma secretion by human T cells. The ED50 for this effect is 1-10 μg/mL.Formulation, Preparation, and Storage
9227-SL
| Formulation | Lyophilized from a 0.2 μm filtered solution in PBS, NaCl, CHAPS and Trehalose. |
| Reconstitution | Reconstitute at 200 μg/mL in water.
Loading...
|
| Shipping | The product is shipped at ambient temperature. Upon receipt, store it immediately at the temperature recommended below. |
| Stability & Storage | Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
|
Calculators
Background: Siglec-15
References
- Humphrey, M.B. and M.C. Nakamura (2015) Clin. Rev. Allergy Immunol. PMID 26573914.
- Angata, T. et al. (2007) Glycobiology 17:838.
- Takamiya, R. et al. (2013) Glycobiology 23:178.
- Kameda, Y. et al. (2013) J. Bone Miner. Res. 28:2463.
- Stuible, M. et al. (2014) J. Biol. Chem. 289:6498.
- Ishida-Kitagawa, N. et al. (2012) J. Biol. Chem. 287:17493.
Long Name
Sialic Acid Binding Ig-like Lectin 15
Alternate Names
CD33L3, HsT1361, Siglec15
Gene Symbol
SIGLEC15
UniProt
Additional Siglec-15 Products
Product Documents for Recombinant Human Siglec-15 Fc Chimera Protein, CF
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Recombinant Human Siglec-15 Fc Chimera Protein, CF
For research use only
Related Research Areas
Citations for Recombinant Human Siglec-15 Fc Chimera Protein, CF
Customer Reviews for Recombinant Human Siglec-15 Fc Chimera Protein, CF (1)
5 out of 5
1 Customer Rating
Have you used Recombinant Human Siglec-15 Fc Chimera Protein, CF?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Showing
1
-
1 of
1 review
Showing All
Filter By:
-
Application: SDS-PAGE ControlVerified Customer | Posted 06/17/2019
There are no reviews that match your criteria.
Loading...
