Recombinant Human SPR His-tag Protein, CF
R&D Systems | Catalog # 10209-SP

Key Product Details
- R&D Systems E. coli-derived Recombinant Human SPR His-tag Protein (10209-SP)
- Quality control testing to verify active proteins with lot specific assays by in-house scientists
- All R&D Systems proteins are covered with a 100% guarantee
Source
Accession Number
Applications
Product Specifications
Source
Met1-Lys261
with a C-terminal 6-His tag
Purity
Endotoxin Level
N-terminal Sequence Analysis
Predicted Molecular Mass
SDS-PAGE
Activity
The specific activity is >700 pmol/min/μg, as measured under the described conditions.
Scientific Data Images for Recombinant Human SPR His-tag Protein, CF
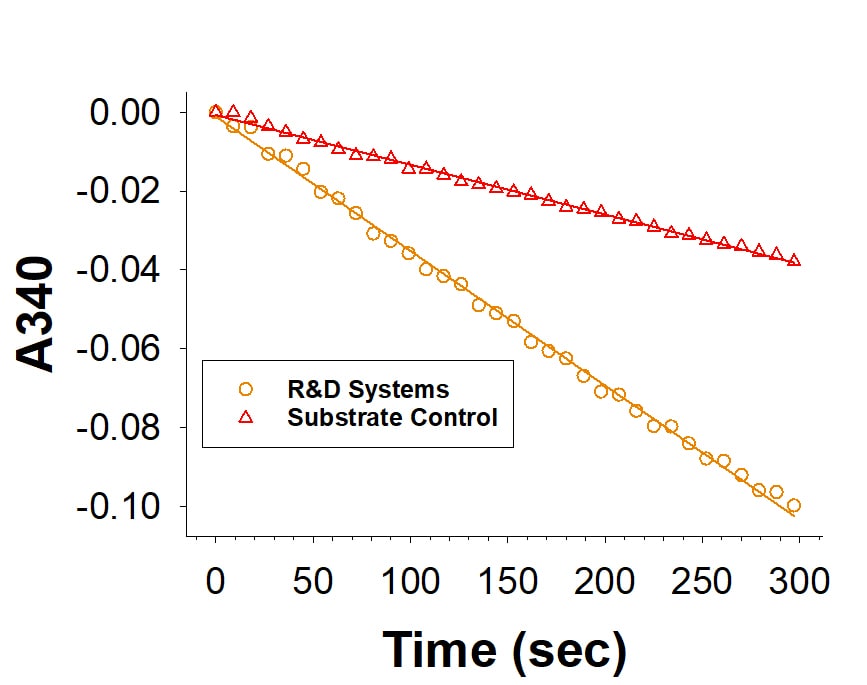
Recombinant Human SPR His-tag Protein Enzyme Activity
Recombinant Human SPR His-tag Protein (Catalog # 10209-SP) is measured by its ability to catalyze the reduction of phenanthrenequinone.Recombinant Human SPR His-tag Protein SDS-PAGE
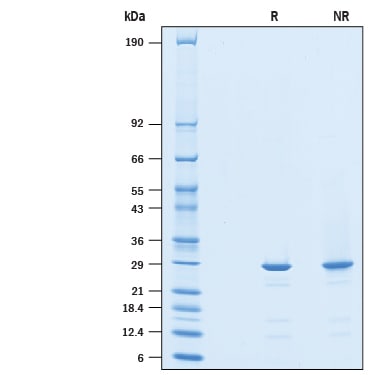
2 μg/lane of Recombinant Human SPR His-tag was resolved with SDS-PAGE under reducing (R) and non-reducing (NR) conditions and visualized by Coomassie® Blue staining, showing a band at 28 kDa under reducing conditions.Formulation, Preparation, and Storage
10209-SP
| Formulation | Supplied as a 0.2 μm filtered solution in Tris, NaCl and Glycerol. |
| Shipping | The product is shipped with polar packs. Upon receipt, store it immediately at the temperature recommended below. |
| Stability & Storage | Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
|
Background: SPR
References
- Werner, E.R. et al. (2011) Biochem. J 348:397.
- Bendall, J. K. et al. (2014) Antiox. Redox. Signal 20:3040.
- Yang, S. et al. (2013) J. Biol. Chem. 288:19222.
- Jornvall, H. et al. (1995) Biochemistry 34:6003.
- Auerbach, G. et al. (1997) EMBO J. 16:7219.
- Fujimoto, K. et al. (2001) Chem. Biol. Interact. 130:825.
- Katoh, S. (1971) Arch. Biochem. Biophys. 146:202.
- Bonafe, L. et al. (2001) Am. J. Hum. Genet 69:269.
- Farrugia, R. et al. (2007) Mol. Genet. Metab. 90:277.
- Abeling, N.G. et al. (2006) Mol. Genet. Metab. 89:116.
- Yang, S. et al. (2006) Am. J. Hum. Genet. 78:575.
- Latremoliere, A. et al. (2015) Neuron. 86:1393.
- Fujita, M. et al. (2019) Arthritis Rheumatol. [Epub ahead of print]
- Cronin, S.J.F. et al. (2018) Nature. 563:7732.
Long Name
Alternate Names
Gene Symbol
UniProt
Additional SPR Products
Product Documents for Recombinant Human SPR His-tag Protein, CF
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Recombinant Human SPR His-tag Protein, CF
For research use only
Customer Reviews for Recombinant Human SPR His-tag Protein, CF
There are currently no reviews for this product. Be the first to review Recombinant Human SPR His-tag Protein, CF and earn rewards!
Have you used Recombinant Human SPR His-tag Protein, CF?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
View specific protocols for Recombinant Human SPR His-tag Protein, CF (10209-SP):
- Assay Buffer: 50 mM Potassium Phosphate, 150 mM NaCl, pH 6.0
- Recombinant Human SPR His-tag (rhSPR) (Catalog # 10209-SP)
- Substrate: 9,10-Phenanthrenequinone (PQ) (Sigma, Catalog # 156507), 5 mM stock in N,N-Dimethylformamide (DMF)
- beta -Nicotinamide adenine dinucleotide phosphate reduced, tetrasodium salt ( beta -NADPH) (Sigma, Catalog# N7505), 10 mM stock in deionized water
- 96-well clear plate (Catalog # DY990)
- Fluorescent Plate Reader (Model: SpectraMax Plus by Molecular Devices) or equivalent
- Dilute rhSPR to 10 µg/mL in Assay Buffer.
- Prepare Substrate Mixture containing 200 µM PQ and 800 µM beta -NADPH in Assay Buffer.
- Load 50 µL of 10 µg/mL rhSPR into a plate, and start the reaction by adding 50 µL of Substrate Mixture. Include a Substrate Blank containing 50 µL Assay Buffer and 50 µL of Substrate Mixture.
- Read at an absorbance of 340 nm, respectively, in kinetic mode of 5 minutes.
- Calculate specific activity:
Specific Activity (pmol/min/µg) = | Adjusted Vmax* (OD/min) x well volume (L) x 1012 pmol/mol x -1 |
| ext. coeff** (M-1cm-1) x path corr.*** (cm) x amount of enzyme (µg) |
*Adjusted for Substrate Blank
**Using the extinction coefficient 6270 M-1cm-1
***Using the path correction 0.32 cm
Note: the output of many spectrophotometers is in mOD
Per Well:
- rhSPR: 0.5 µg
- PQ: 100 µM
- beta -NADPH: 400 µM