Signal regulatory protein alpha (SIRP alpha, designated CD172a), also called SHPS-1 (SHP substrate 1) and previously, MyD-1 (Myeloid/Dendritic-1), is a homodimeric, 100-105 kDa type I transmembrane glycoprotein that belongs to the SIRP/SHPS (CD172) family of the immunoglobulin superfamily (1-5). SIRPs are paired receptors, with similar extracellular domains but differing C-termini and functions (1, 2). The 513 amino acid (aa) mouse SIRP alpha contains a 342 aa extracellular domain (ECD) with one V‑type and two C1 type Ig domains and many potential N‑glycosylation sites. It has a 117 aa cytoplasmic sequence with ITIM motifs that recruit tyrosine phosphatases SHP-1 and SHP-2 when phosphorylated (4). Mouse and human SIRP alpha have at least 30 described polymorphisms, including the human SIRP alpha prominent variant BIT (Brain Ig like molecule with Tyrosine-based activation motifs, also called SIRP alpha 2 or PTPNS) (2). In mouse, one splice variant lacks aa 147-364, which eliminates the C type Ig domains, while another lacks only aa 425-428 (6). Mouse SIRP alpha ECD shares 61%, 75%, 62%, 61%, and 59% aa sequence identity with human, rat, equine, bovine, and porcine SIRP alpha, respectively, and shares 62% aa identity with mouse SIRP beta 1 (2). SIRP alpha is expressed mainly on myeloid cells, including macrophages, neutrophils, dendritic and Langerhans cells (3 ‑ 7). It is also found on neurons, smooth muscle and endothelial cells (8-10). SIRP alpha shows adhesion to the ubiquitous CD47/IAP (integrin associated protein), while SIRP gamma binds more weakly and SIRP beta 1 does not bind at all (1, 2). Mouse and human SIRP alpha are allelic in nature, and variation(s) in the V-type Ig-like domain likely impacts its binding to CD47 (11). SIRP alpha engagement generally produces a negative regulatory signal (4). Low SIRP alpha recognition of CD47, which occurs on aged erythrocytes or platelets or xenogenic cells, promotes clearance of CD47low cells from circulation (12 ‑ 14). SIRP alpha recognition of surfactants SP‑A and SP‑D in the lung can inhibit alveolar macrophage cytokine production (15). The CD47 integrin-SIRP alpha interaction is reported to promote macrophage fusion during osteoclastogenesis (16).
Recombinant Mouse SIRP alpha/CD172a Fc Chimera Protein, CF
R&D Systems | Catalog # 7154-SA

Key Product Details
- R&D Systems NS0-derived Recombinant Mouse SIRP alpha/CD172a Fc Chimera Protein (7154-SA)
- Quality control testing to verify active proteins with lot specific assays by in-house scientists
- All R&D Systems proteins are covered with a 100% guarantee
Source
Accession Number
Structure / Form
Applications
Product Specifications
Source
| Mouse SIRP alpha /CD172a (Met1-Asn373) (Gly365Asp) Accession # P97797 |
IEGRMDP | Mouse IgG2A (Glu98-Lys330) |
| N-terminus | C-terminus |
Purity
Endotoxin Level
N-terminal Sequence Analysis
Predicted Molecular Mass
SDS-PAGE
Activity
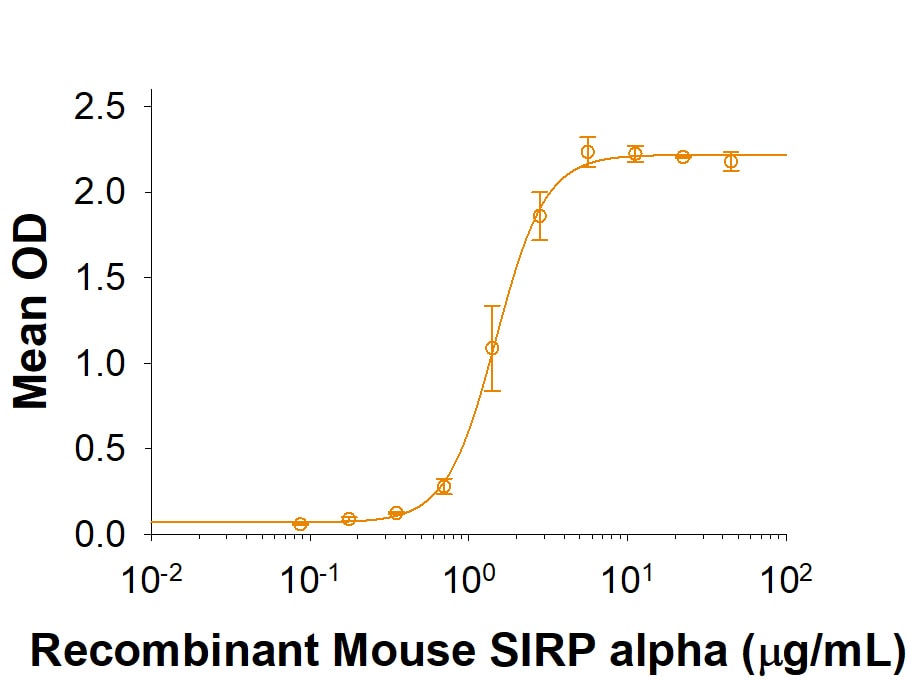
The ED50 for this effect is 0.4-1.6 μg/mL.
Optimal dilutions should be determined by each laboratory for each application.
Scientific Data Images for Recombinant Mouse SIRP alpha/CD172a Fc Chimera Protein, CF
Recombinant Mouse SIRP alpha/CD172a Fc Chimera Protein Bioactivity
Recombinant Mouse SIRP alpha /CD172a Fc Chimera (Catalog # 7154-SA) supports the adhesion of mouse red blood cells. The ED50 for this effect is 0.4‑1.6 µg/mL.Formulation, Preparation, and Storage
7154-SA
| Formulation | Lyophilized from a 0.2 μm filtered solution in PBS. |
| Reconstitution | Reconstitute at 500 μg/mL in PBS.
Loading...
|
| Shipping | The product is shipped at ambient temperature. Upon receipt, store it immediately at the temperature recommended below. |
| Stability & Storage | Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
|
Calculators
Background: SIRP alpha/CD172a
References
- Barclay, A.N. (2009) Curr. Opin. Immunol. 21:47.
- van Beek, E.M. et al. (2005) J. Immunol. 175:7781.
- Liu, Y. et al. (2005) J. Biol. Chem. 280:36132.
- Sano, S-I. et al. (1999) Biochem. J. 344:667.
- Lee, W.Y. et al. (2010) J. Biol. Chem. 285:37953.
- Swissprot Accession # P97797.
- Miyashita, M. et al. (2004) Mol. Biol. Cell 15:3950.
- Wang, X.X. & K.H. Pfenninger (2005) J. Cell Sci. 119:172.
- Maile, L.A. et al. (2003) Mol. Biol. Cell 14:3519.
- Johansen, M.L. & E.J. Brown (2007) J. Biol. Chem. 282:24219.
- Takenaka, K. et al. (2007) Nat. Immunol. 8:1313.
- Ishikawa-Sekigami, T. et al. (2006) Biochem. Biophys. Res. Commun. 343:1197.
- Olsson, M. et al. (2005) Blood 105:3577.
- Ide, K. et al. (2007) Proc. Natl. Acad. Sci. USA 104:5062.
- Gardai, S.J. et al. (2003) Cell 115:13.
- Lundberg, P. et al. (2007) Biochem. Biophys. Res. Commun. 352:444.
Long Name
Alternate Names
Entrez Gene IDs
Gene Symbol
UniProt
Additional SIRP alpha/CD172a Products
Product Documents for Recombinant Mouse SIRP alpha/CD172a Fc Chimera Protein, CF
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Recombinant Mouse SIRP alpha/CD172a Fc Chimera Protein, CF
For research use only
Citations for Recombinant Mouse SIRP alpha/CD172a Fc Chimera Protein, CF
Customer Reviews for Recombinant Mouse SIRP alpha/CD172a Fc Chimera Protein, CF
There are currently no reviews for this product. Be the first to review Recombinant Mouse SIRP alpha/CD172a Fc Chimera Protein, CF and earn rewards!
Have you used Recombinant Mouse SIRP alpha/CD172a Fc Chimera Protein, CF?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
FAQs for Recombinant Mouse SIRP alpha/CD172a Fc Chimera Protein, CF
-
Q: What is the purpose of the Gly365Asp substitution in product catalog #7154-SA - Mouse SIRP alpha /CD172a?
A: The Gly365Asp substitution was not introduced into the protein sequence of Mouse SIRP alpha /CD172a by design, rather, the substitution was obtained from PCR isolation of the gene. Since we confirmed activity by the ability of the immobilized protein to support the adhesion of mouse red blood cells, we believe the substitution is not critical to the funtion of the protein.


