SL 327
Tocris Bioscience | Catalog # 1969

Key Product Details
Description
Product Description
SL 327 is a selective inhibitor of MEK1 and MEK2 (IC50 values are 0.18 and 0.22 μM respectively); blocks hippocampal LTP in vitro. Brain penetrant in vivo, blocking fear conditioning and learning in rats, and producing neuroprotection in mice, following systemic administration.
Product Specifications for SL 327
Molecular Weight
Formula
Storage
Purity
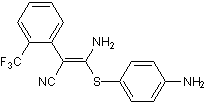
Chemical Name
CAS Number
PubChem ID
InChI Key
SMILES
The technical data provided above is for guidance only. For batch specific data refer to the Certificate of Analysis.
Solubility
| Solvent | Max Conc. mg/mL | Max Conc. mM | |
|---|---|---|---|
| Solubility | |||
| DMSO | 33.53 | 100 | |
| Ethanol | 16.77 | 50 |
Preparing Stock Solutions for SL 327
The following data is based on the product molecular weight 335.35.
Batch specific molecular weights may vary from batch to batch due to the degree of hydration, which all affect the solvent volumes required to prepare stock solutions.
| Concentration / Solvent Volume / Mass | 1 mg | 5 mg | 10 mg |
|---|---|---|---|
| 1 mM | 2.98 mL | 14.91 mL | 29.82 mL |
| 5 mM | 0.60 mL | 2.98 mL | 5.96 mL |
| 10 mM | 0.30 mL | 1.49 mL | 2.98 mL |
| 50 mM | 0.06 mL | 0.30 mL | 0.60 mL |
Calculators
Background References
References are publications that support the biological activity of the product. See our Citations tab to view 77 publications citing the usage of this product.
- Atkins The MAPK cascade is required for mammalian associative learning. Nature Neurosci. 1998 PMID: 10196568
- Scherle Regulation of cyclooxygenase-2 induction in the mouse uterus during decidualization. An event of early pregnancy. J.Biol.Chem. 2000 PMID: 10969080
- Wang Significant neuroprotection against ischemic brain injury by inhibition of the MEK1 protein kinase in mice: exploration of potential mechanism associated with apoptosis. J.Pharmacol.Exp.Ther. 2003 PMID: 12490588
- Davis The MAPK/ERK cascade targets both Elk-1 and cAMP response element-binding protein to control long-term potentiation-dependent gene expression in the dentate gyrus in vivo. J.Neurosci. 2000 PMID: 10844026
Product Documents for SL 327
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Product Specific Notices for SL 327
For research use only
Related Research Areas
Citations for SL 327
Customer Reviews for SL 327
There are currently no reviews for this product. Be the first to review SL 327 and earn rewards!
Have you used SL 327?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
