SNAP 5089
Discontinued Product
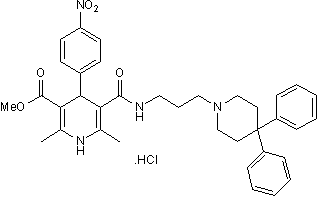
Chemical Name: 5-[[[3-(4,4-Diphenyl-1-piperidinyl)propyl]amino]carbonyl]-1,4-dihydro-2,6-dimethyl-4-(4-nitrophenyl)-3-pyridinecarboxylic acid methyl ester hydrochloride
Purity: ≥98%
Biological Activity
SNAP 5089 is a subtype-selective α1A-adrenoceptor antagonist that displays > 600-fold selectivity over other adrenoceptors (Kivalues are 0.35, 220, 370, 540, 800 and 1200 nM for α1A, α1B, α2C, α1D, α2B and α2A subtypes respectively and 540 nM for L-type Ca2+ channels). Inhibits noradrenalin-induced contractions in rabbit vascular and lower urinary tissues.Technical Data
The technical data provided above is for guidance only.
For batch specific data refer to the Certificate of Analysis.
Tocris products are intended for laboratory research use only, unless stated otherwise.
Background References
-
Discovery of α1a-adrenergic receptor antagonists based on the L-type Ca2+ channel antagonist niguldipine.
Wetzel et al.
J.Med.Chem., 1995;38:1579 -
Pharmacological characterization of the uroselective alpha-1 antagonist Rec 15-2739 (SB 216469): role of the alpha-1L adrenoceptor in tissue selectivity, Part I.
Leonardi et al.
J.Pharmacol.Exp.Ther., 1997;281:1272 -
α1L-adrenceptor mediation of smooth muscle contraction in rabbit bladder neck: a model for lower urinary tract tissues of man.
Kava et al.
Br.J.Pharmacol., 1998;123:1359
Product Datasheets
Reconstitution Calculator
Molarity Calculator
FAQs
No product specific FAQs exist for this product, however you may
View all Small Molecule FAQsReviews for SNAP 5089
There are currently no reviews for this product. Be the first to review SNAP 5089 and earn rewards!
Have you used SNAP 5089?
Submit a review and receive an Amazon gift card.
$25/€18/£15/$25CAN/¥75 Yuan/¥2500 Yen for a review with an image
$10/€7/£6/$10 CAD/¥70 Yuan/¥1110 Yen for a review without an image