The term 'mesenchymal stem cells' (MSCs) is most commonly used to describe multipotent self-renewing cells that can be differentiated in vitro to generate adipocytes, chondrocytes, and osteoblasts. However, because these biological properties and hierarchical relationships remain to be clearly demonstrated in vivo, the term 'multipotent mesenchymal stromal cells' is often used to distinguish cultured cells from their in vivo precursors. Originally discovered in mouse bone marrow, multipotent mesenchymal stromal cells cultured from a variety of species and tissue types, have been shown to differentiate into progeny of additional lineages including, cardiomyocytes, endothelial cells, hepatocytes, and neural cells. Again, the physiological relevance of these findings remains to be determined.
StemXVivo Adipogenic Supplement (100X)
R&D Systems | Catalog # CCM011

Key Product Details
Species
Product Summary for StemXVivo Adipogenic Supplement (100X)
Kit Summary
Media supplement to induce the differentiation of human MSCs into osteocytes. For use with Human/Mouse/Rat StemXVivo® Osteogenic/Adipogenic Base Media (Catalog # CCM007).
Key Benefits
- Supports induction of adipogenesis in MSCs
- Defined supplement to reduce experimental variation
- Developed and optimized using MSCs
Why Induce Adipogenesis in MSCs with a Defined Media Supplement?
Despite the well-characterized factors and protocols used to differentiate mesenchymal stem/stromal cells (MSCs) into adipocytes, differentiation efficiencies can vary depending on the quality of the MSC starting population and the reagents used to expand and differentiate MSCs.
StemXVivo® Adipogenic Supplement:
- Contains high quality differentiation factors to drive reproducible and efficient MSC adipogenesis.
- Is defined to reduce unwanted experimental variability.
- Has been developed and optimized using MSCs.
The term ‘mesenchymal stromal cells’ is commonly used to describe a heterogeneous population of cultured cells that are adherent to plastic, have a distinct morphology, and express a specific set of marker proteins. Within this heterogeneous population are cells referred to as ‘mesenchymal stem cells.’
Mesenchymal stem cells are multipotent, self-renewing cells that have the ability to differentiate into adipocytes, chondrocytes, and osteoblasts when cultured in vitro. Read More about MSC Nomenclature
StemXVivo® Adipogenic Supplement Components
This media supplement contains high quality factors to drive human, mouse, or rat MSC differentiation into adipocytes.
- This supplement requires media (not included), such as Human/Mouse/Rat StemXVivo® Osteogenic/Adipogenic Base Media (Catalog # CCM007) or equivalent.
- The quantity of adipogenic media supplement supplied is sufficient to make 350 mL of media for differentiation.
2006 Proposed Change to MSC Nomenclature
Although mesenchymal stromal cells were once referred to as ‘mesenchymal stem cells’, a change to ‘mesenchymal stromal cells’ was proposed by the International Society for Cellular Therapy in 2006.1
The change in nomenclature originates from two important factors:
- Methods used to isolate mesenchymal stem cells yield a heterogeneous population of cells with only a fraction of these cells demonstrating multipotency.
- The absence of direct evidence that mesenchymal stem cells can self-renew and differentiate in vivo.
Use of Mesenchymal Stem and Stromal Cell Terminology
Data supporting MSC self-renewal and multipotency have been obtained using in vitro conditions, which does not adequately reflect the in vivo environment. The lack of in vivo data has led some researchers to question the validity of the term ‘mesenchymal stem cell’ providing further support for the use of ‘mesenchymal stromal cells’ to describe MSCs.2 While ‘mesenchymal stromal cells’ may be the more scientifically accurate term for MSCs, the two terms are often used interchangeably in the literature. R&D Systems recognizes the use of both mesenchymal stem cells and mesenchymal stromal cells and uses ‘MSC’ to indicate mesenchymal stem/stromal cells to account for both designations.
Definitions of Mesenchymal Stromal Cells and Mesenchymal Stem Cells
- Mesenchymal Stromal Cells – A heterogeneous population of cultured cells with similar characteristics such as the ability to adhere to plastic and the expression of specific marker proteins.
- Mesenchymal Stem Cells – A subpopulation of mesenchymal stromal cells that have the capacity to self-renew and differentiate into mesodermal lineages when cultured in vitro. The capacity to self-renew and differentiate in vivo has yet to be clearly demonstrated for mesenchymal stem cells.
References
- Dominici, M. et al. (2006) Cytotherapy 8:315.
- Keating, A. (2012) Cell Stem Cell 10:709.
Scientific Data Images for StemXVivo Adipogenic Supplement (100X)
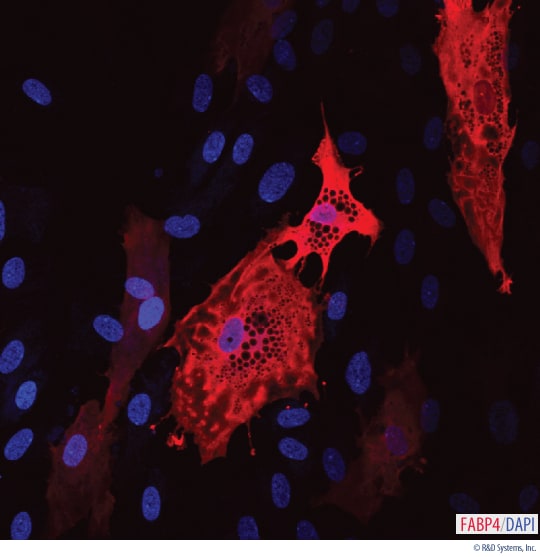
Detection of FABP4 in Human MSC-differentiated Adipocytes.
Human MSCs were differentiated for 14 days using the Human/Mouse/Rat StemXVivo®Osteogenic/Adipogenic Base Media (Catalog # CCM007) and Human/Mouse/Rat StemXVivo®Adipogenic Supplement (Catalog # CCM011). Mature differentiated adipoctyes were detected with a Goat Anti-Human FABP4 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF3150). The cells were stained with a NorthernLights™557-conjugated Donkey Anti-Goat Secondary Antibody (Catalog # NL001; red) and the nuclei were counterstained with DAPI (blue).Detection of FABP4 in Mouse MSC-differentiated Adipocytes.
Mouse MSCs were differentiated for 14 days using the Human/Mouse/Rat StemXVivo®Osteogenic/Adipogenic Base Media (Catalog # CCM007) and Human/Mouse/Rat StemXVivo®Adipogenic Supplement (Catalog # CCM011). Mature differentiated adipoctyes were detected with a Goat Anti-Mouse FABP4 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF1443). The cells were stained with a NorthernLights™557-conjugated Donkey Anti-Goat Secondary Antibody (Catalog # NL001; red) and the nuclei were counterstained with DAPI (blue).Detection of FABP4 in Rat MSC-differentiated Adipocytes.
Rat MSCs were differentiated for 14 days using the Human/Mouse/Rat StemXVivo®Osteogenic/Adipogenic Base Media (Catalog # CCM007) and Human/Mouse/Rat StemXVivo®Adipogenic Supplement (Catalog # CCM011). Mature differentiated adipoctyes were detected with a Goat Anti-Mouse FABP4 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF1443). The cells were stained with a NorthernLights™557-conjugated Donkey Anti-Goat Secondary Antibody (Catalog # NL001; red) and the nuclei were counterstained with DAPI (blue).Formulation, Preparation, and Storage
Shipping
Storage
Background: Mesenchymal Stem Cells
Alternate Names
Additional Mesenchymal Stem Cells Products
Product Documents for StemXVivo Adipogenic Supplement (100X)
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for StemXVivo Adipogenic Supplement (100X)
For research use only
Citations for StemXVivo Adipogenic Supplement (100X)
Customer Reviews for StemXVivo Adipogenic Supplement (100X)
There are currently no reviews for this product. Be the first to review StemXVivo Adipogenic Supplement (100X) and earn rewards!
Have you used StemXVivo Adipogenic Supplement (100X)?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
View specific protocols for StemXVivo Adipogenic Supplement (100X) (CCM011):
Refer to the product datasheet for complete product details.
Briefly, human, mouse, or rat MSCs are differentiated into adipocytes using the following in vitro differentiation procedure:
- Culture multipotent cells of interest
- Induce adipogenic differentiation using a media supplement
- Evaluate differentiation using a mature phenotype marker antibody and fluorescent ICC
For use with Human/Mouse/Rat StemXVivo® Osteogenic/Adipogenic Base Media (Catalog # CCM007).
View Graphic Procedure
Reagents Provided
Reagents supplied in the Human/Mouse/Rat StemXVivo® Adipogenic Supplement (Catalog # CCM011):
- Human/Mouse/Rat StemXVivo® Osteogenic/Adipogenic Base Media (Catalog # CCM007 or equivalent)
- Penicillin-Streptomycin-Glutamate (100X)
Other Supplies Required
Reagents
- StemXVivo® Osteogenic/Adipogenic Base Media (Catalog # CCM007 or equivalent)
- Penicillin-Streptomycin-Glutamate (100X)
Materials
- MSCs
- 10 cm tissue culture plates
- 15 mL centrifuge tubes
- Pipettes and pipette tips
- Serological pipettes
Equipment
- 37 °C and 5% CO2 incubator
- Centrifuge
- Hemocytometer
- Inverted microscope
- 2 °C to 8 °C refrigerator
- 37 °C water bath
Procedure Overview
This protocol has been tested using bone marrow- and/or adipose tissue-derived MSCs. If using a different tissue source or cell line, the protocol below may need to be optimized.
Adipogenic Differentiation
- Plate 2.1 x 104 MSCs/cm2 in StemXVivo® Osteogenic/Adipogenic Base Media.
- Culture cells to 100% confluency.
- Replace the medium with StemXVivo® Adipogenic Differentiation Medium to induce adipogenesis.
- Every 3-4 days, replace with fresh Adipogenic Differentiation Medium.
- After 7-21 days, adipocytes can be harvested and analyzed.
FAQs for StemXVivo Adipogenic Supplement (100X)
-
Q: In the Human Mesenchymal Stem Cell Functional Identification Kit (Catalog # SC006), are Part #'s 90415, 390416, and 390417 the same as the StemXVivo® Human Adipogenic Supplement (Catalog # CCM011), StemXVivo® Human Osteogenic Supplement (Catalog # CCM008), and StemXVivo® Human Chondrogenic Supplement (Catalog # CCM006), respectively?
A: Yes, the StemXVivo® Human Adipogenic Supplement (Catalog # CCM011), StemXVivo® Human Osteogenic Supplement (Catalog # CCM008), and StemXVivo® Human Chondrogenic Supplement (Catalog # CCM006) are the same as Part #'s 390415, 390416, and 390417, respectively, in the Human Mesenchymal Stem Cell Functional Identification Kit (Catalog # SC006).



