Phosphatase-coupled Kinase Reaction
Rate Constant Determination and Coupling Rate Calculation
R&D Systems Universal Kinase Activity Kit (Catalog # EA004) is an ADP-based phosphatase-coupled kinase assay that is used for determining enzyme activity of all kinases that produce ADP. This kit utilizes the phosphatase CD39L2/ENTPD6 to release the beta-phosphate from ADP that is produced during the kinase reaction. Thus, this assay is dependent on the efficiency with which CD39L2 is able to convert ADP to the free phosphate signal. The percentage of the ADP product that has been converted to the signal of free inorganic phosphate by the coupling reaction is called the coupling rate.
The coupling rate is used to ascertain the amount of ADP produced during the kinase reaction. This amount of ADP is needed in order to determine activity of the kinase. Coupling rates of kinase reactions are dependent upon multiple variables including the rate constant of the coupling enzyme, the volume, time and temperature of the reaction, and the pH and salt concentration of the buffer. If these conditions are different from the protocol outlined in the product datasheet, the coupling rate needs to be recalculated. This requires prior determination of the rate constant.
Determining the Rate Constant
Reagents Required
The following reagents are provided in the Universal Kinase Activity Kit (Catalog # EA004):
- ADP
- CHO cell-expressed Recombinant Mouse CD39L2/ENTPD6
- Phosphatase Buffer 4
- Malachite Green Reagents A & B
Additional reagents needed:
- Deionized or distilled water
Materials Required
- 96-well microplate (Catalog # DY990), or equivalent
- Microplate reader (needs to measure absorbance at 620 nm)
Reagent Preparation
The volume of working solution below is for 20 kinase reactions. Adjust the volume accordingly if a different number of reactions are planned. Discard any remaining preparations after the assay is complete.
1X Assay Buffer: Add 200 μL of 10X Phosphatase Assay Buffer 4 to 1.8 mL of deionized or distilled water in a tube. Mix well.
Protocol
Note: This protocol outlines the procedure for determining the rate constant for CD39L2 in a 96-well microplate.
Note: The phosphate conversion factor is determined by setting up a phosphate standard curve where the phosphate input is plotted again the corrected OD. The slope of the linear regression line is the phosphate conversion factor.
Note: Only points with less than 20% substrate consumption and linear to ADP concentration are considered to be valid.
- Add 40 μL of 10 mM ADP to 360 μL of 1X Assay Buffer in a microcentrifuge tube and mix well. Transfer 200 μL of the dilution into 200 μL of 1X Assay Buffer in a second tube. Repeat the process to prepare a 2-fold serial dilution. The eighth tube contains only 1X Assay Buffer and serves as the blank.
- Transfer 75 μL of each dilution into microplate wells in duplicate.
- Prepare 1 μL of CD39L2 diluted to 0.2-0.5 ng/μL in 1X Assay Buffer.
- Initiate the reaction by adding 75 μL of the diluted CD39L2 into each well.
- Incubate the microplate for 10 minutes at room temperature.
- Terminate the reactions by adding 30 μL of Malachite Green Reagent A to each well. Mix by gently tapping the microplate.
- Add 30 μL of Malachite Green Reagent B to each well. Mix gently by tapping the microplate.
- Incubate the microplate for 20 minutes at room temperature to stabilize the color development.
- Determine the optical density (OD) of each well using a microplate reader set to 620 nm, and adjust the OD by subtracting the negative control.
- Calculate the activity of CD39L2 in each well using a previously determined phosphate conversion factor.
- Plot CD39L2 activity versus ADP concentrations. The specific rate constant for CD39L2 will be the slope of the curve.
Data Example
Rate Constant for CD39L2
Specific activity of CD39L2 was plotted versus the ADP concentrations. The slope of the curve is the specific rate constant. The rate constant is positively related to enzyme activity.
Calculating the Coupling Rate
The coupling rate of the kinase reaction can be calculated using the following equations.
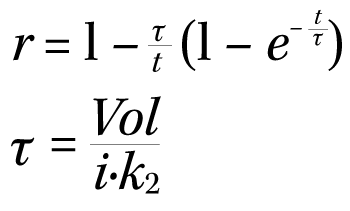
| t: | the reaction time in minutes |
| τ: | the lagging time of the coupling reaction in minutes |
| Vol: | the reaction volume in μL |
| i: | the inhibition factor caused by ATP (can be obtained from cited reference or determined experimentally) |
| k2: | the rate constant in nmol/min/mM (can be obtained by multiplying the determined specific rate constant with the amount of coupling enzyme) |
For a more detailed explanation of the equations, please refer to the cited reference.
Note: Unlike most other coupled reactions, which generally require a high coupling rate, the coupling rate for a CD39L2-coupled kinase reaction needs to be carefully controlled. CD39L2, which does prefer ADP as a substrate, will also act on ATP. As a result, increasing the amount of coupling phosphatase will increase the hydrolysis of ATP and amplify background noise. To reduce the background while maintaining a good signal, the amount of CD39L2 needs to be restricted so that the optimal coupling rate falls between 0.4 and 0.6.

