53BP1 Antibody - BSA Free
Novus Biologicals | Catalog # NB100-305

![Immunocytochemistry/ Immunofluorescence: 53BP1 Antibody - BSA Free [NB100-305] Immunocytochemistry/ Immunofluorescence: 53BP1 Antibody - BSA Free [NB100-305]](https://resources.rndsystems.com/images/products/53BP1-Antibody-Immunocytochemistry-Immunofluorescence-NB100-305-img0010.jpg)
Key Product Details
Validated by
Species Reactivity
Validated:
Cited:
Predicted:
Applications
Validated:
Cited:
Label
Antibody Source
Format
Product Specifications
Immunogen
Reactivity Notes
Marker
Clonality
Host
Isotype
Scientific Data Images for 53BP1 Antibody - BSA Free
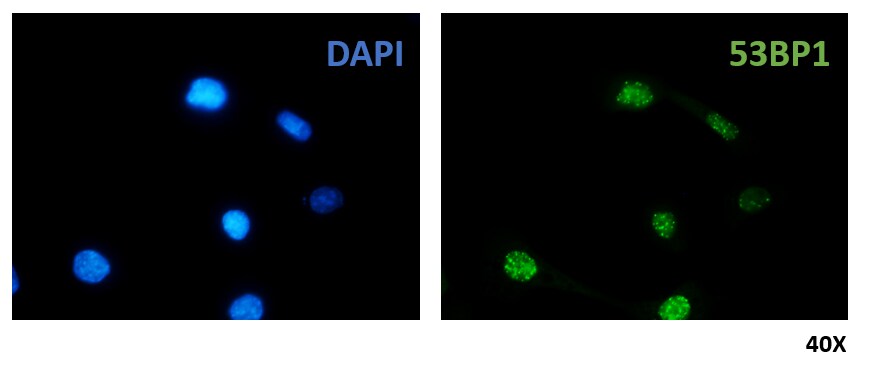
Immunocytochemistry/ Immunofluorescence: 53BP1 Antibody - BSA Free [NB100-305]
Immunocytochemistry/Immunofluorescence: 53BP1 Antibody [NB100-305] - NIH3T3 cells were fixed in 4% paraformaldehyde for 10 minutes and permeabilized in 0.5% Triton X-100 in PBS for 5 minutes. The cells were incubated with anti- NB100-305 at 2 ug/mL overnight at 4C and detected with an anti-rabbit DyLight 488 (Green) at a 1:1000 dilution for 60 minutes. Alpha tubulin (DM1A) NB100-690 was used as a co-stain at a 1:1000 dilution and detected with an anti-mouse DyLight 550 (Red) at a 1:1000 dilution. Nuclei were counterstained with DAPI (Blue). Cells were imaged using a 100X objective and digitally deconvolved.Immunohistochemistry: 53BP1 Antibody - BSA Free [NB100-305]
Immunohistochemistry: 53BP1 Antibody [NB100-305] - Immunohistochemical staining of placental villi with 53BP1 Antibody (Catalog #NB100-305) at 40X magnification.Western Blot: 53BP1 AntibodyBSA Free [NB100-305]
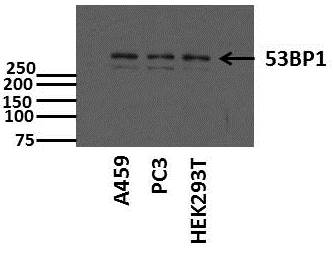
Western Blot: 53BP1 Antibody [NB100-305] - Western blot of whole cell lysate (20 ug/lane) from U2Os cells resolved on a 3 to 8 percent trisacetate gel. A band appears at approximately the theoretical molecular weight of 214 kDa.Immunocytochemistry/ Immunofluorescence: 53BP1 Antibody - BSA Free [NB100-305]
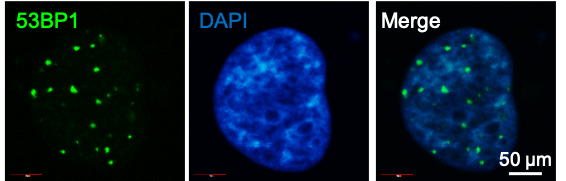
Immunocytochemistry/Immunofluorescence: 53BP1 Antibody [NB100-305] - HCC1937 cells (human) were treated with PARPi for 96 h. Nuclei are stained in blue (DAPI) and 53BP1 antibody (foci in green). ICC/IF image submitted by a verified customer review.Flow Cytometry: 53BP1 Antibody - BSA Free [NB100-305]
Flow Cytometry: 53BP1 Antibody [NB100-305] - An intracellular stain was performed on Ntera2 cells with 53BP1 Antibody NB100-305 (blue) and a matched isotype control NBP2-24891 (orange). Cells were fixed with 4% PFA and then permeabilized with 0.1% saponin. Cells were incubated in an antibody dilution of 1.0 ug/mL for 30 minutes at room temperature, followed by Rabbit IgG (H+L) Cross-Adsorbed Secondary Antibody, Dylight 550 (SA5-10033, Thermo Fisher).Immunocytochemistry/ Immunofluorescence: 53BP1 Antibody - BSA Free [NB100-305]
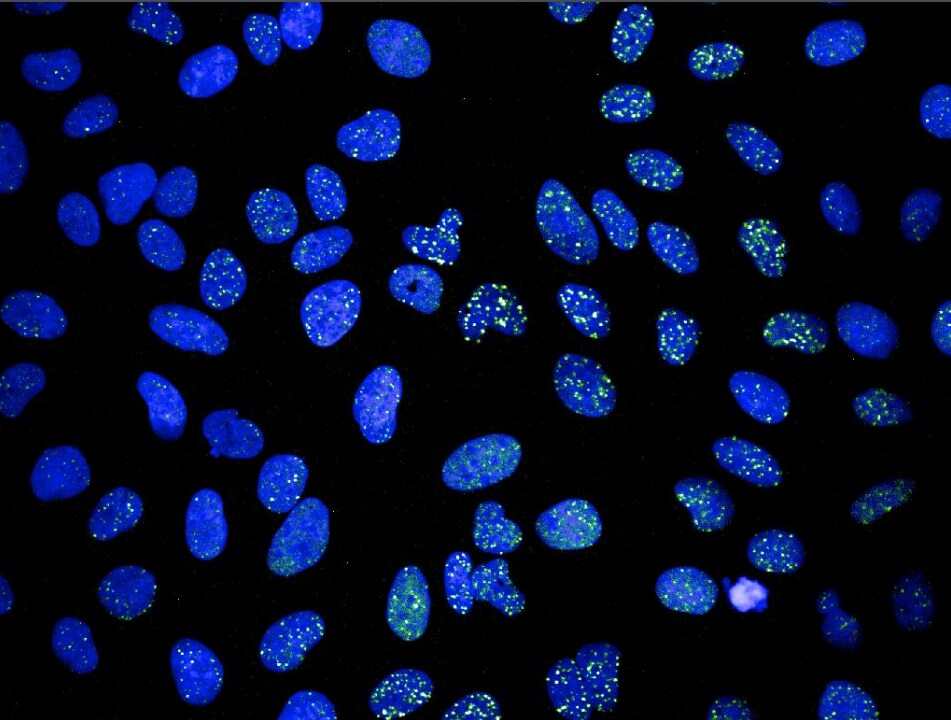
Immunocytochemistry/Immunofluorescence: 53BP1 Antibody [NB100-305] - Paraformaldehyde (4%) fixed Saos-2 cells (human osteosarcoma cell line) were stained with anti-53BP1 antibody followed by Alexa Fluor 488 secondary antibody. ICC/IF image submitted by a verified customer review.Immunocytochemistry/ Immunofluorescence: 53BP1 Antibody - BSA Free [NB100-305]
Immunocytochemistry/Immunofluorescence: 53BP1 Antibody [NB100-305] - Ntera2 cells were fixed in 4% paraformaldehyde for 10 minutes and permeabilized in 0.5% Triton X-100 in PBS for 5 minutes. The cells were incubated with anti- NB100-305 at 2 ug/mL overnight at 4C and detected with an anti-rabbit DyLight 488 (Green) at a 1:1000 dilution for 60 minutes. Alpha tubulin (DM1A) NB100-690 was used as a co-stain at a 1:1000 dilution and detected with an anti-mouse DyLight 550 (Red) at a 1:1000 dilution. Nuclei were counterstained with DAPI (Blue). Cells were imaged using a 100X objective and digitally deconvolved.Flow Cytometry: 53BP1 Antibody - BSA Free [NB100-305]
Flow Cytometry: 53BP1 Antibody [NB100-305] - An intracellular stain was performed on HeLa cells with 53BP1 Antibody (Catalog #NB100-305AF488) (blue) and a matched isotype control (orange). Cells were fixed with 4% PFA and then permeabilized with 0.1% saponin. Cells were incubated in an antibody dilution of 5 ug/mL for 30 minutes at room temperature. Both antibodies were conjugated to Alexa Fluor 488.Western Blot: 53BP1 Antibody - BSA Free [NB100-305] -
Western Blot: 53BP1 Antibody - BSA Free [NB100-305] - DNA damage & DNA damage responses upon H2O2 challenge. a, c, e, g Confocal images showing the basal region of the organ of Corti cultures treated with either culture medium alone (a, e) or containing 0.5 mM H2O2 (c, g) for 5 h before being maintained in culture medium alone for 3 days. The samples were then immunolabeled for myosin 7A (red, a, c, e, g), gamma H2AX (green, a & c) & 53BP1 (green, e & g). Scale bars: a, c, e & g = 10 μm. b, d, f, h Higher magnification images of representative OHC & IHC nuclei from all conditions tested. Scale bar = 2.5 μm. i Representative Western blot analysis using antibodies against gamma H2AX, 53BP1, DDB2, p-Chk1, p-Chk2, p53, & beta -actin in whole cochlear extracts. j, k Histograms representing the levels of gamma H2AX, 53BP1, DDB2, p-Chk1, p-Chk2, & p53 in control & in 0.4 & 0.5 mM H2O2-exposed groups (n = 6 cochleae per condition). beta -Actin served as a loading control. Data are expressed as mean ± SEM. One-way ANOVA test followed by post hoc Tukey’s test (*P ≤ 0.04, **P ≤ 0.01, ***P ≤ 0.001 vs. H2O2 0 mM). All experiments were performed in triplicate Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/30693443), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: 53BP1 Antibody - BSA Free [NB100-305] -
Western Blot: 53BP1 Antibody - BSA Free [NB100-305] - Assessment of satellite cell radiosensitivity & DNA repair pathways w/ age.(A–B) Expression of major proteins involved in homologous recombination & non-homologous end joining detected using WB. Protein nuclear extracts prepared from activated satellite cells 72 hrs post muscle injury. Lamin B used as loading control. (A) Representative images of WBs. (B) Quantification using Image J software. Data represents mean +/− SEM, n = 3 to 5 (n = a pool of muscles from 3 mice), 2-tailed unpaired Student's t-test, no significant difference. (C) Graphical representation of data from Table S1. Gene expression of old relative to young satellite cells presented as fold change w/ a 95 % confidence interval for the 12 genes w/ a statistically significant relative expression, n = 3 mice per group, two-tailed unpaired Student's t-test, *: p value<0.05. Dotted line represents young level. BER: Base Excision Repair, NER: Nucleotide-Excision Repair; MR: Mismatch Repair, DRR: DNA Repair Related. (D–E) Satellite cells freshly isolated from uninjured (D) or 72 hrs after injury (E) muscle of C57BL/6 young & old mice, & of young SCID mice. Cells plated at low density, irradiated at indicated Gray (Gy) doses, & cultured for 10 days. Myogenic colonies formed by irradiated cells quantified & represented relative to ir respective non-irradiated controls. On average, 178, 77, & 228 myogenic colonies scored per mouse for young, old, & SCID uninjured non-irradiated respectively & 67, 34, & 91 myogenic colonies scored per mouse for young, old, & SCID injured non-irradiated respectively. Data represent mean +/− SEM, n = 3 to 4 mice per group, two-tailed unpaired Student's t-test, a, b or c: p value<0.05, (a: young vs old, b: young vs SCID, c: old versus SCID). Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/23704914), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Knockdown Validated: 53BP1 Antibody - BSA Free [NB100-305] -
PARP1-dependent end joining repairs the resected DSBs in 53BP1- or RIF1-depleted G1 cellsA. HeLa cells harboring pEJ were treated with the indicated siRNAs for 24h and PARP1 was then inhibited by adding 1μM olaparib 2h before transfection with I-SceI-expressing vector. The percentages of GFP+ cells were measured after 48h as indication for end joining efficiency. PARP1-EJ was measured as the percent of suppressed end joining after PARP1 inhibition. B. Representative micrographs for gamma H2AX foci at 8h post 2Gy in A549 cells after depletion of either 53BP1 or RIF1 proteins in the presence or absence of olaparib. Nuclei were counterstained with CenpF to determine G1 (CenpF−) cells. C. Quantitation of gamma H2AX foci number at the indicated time points in CenpF− G1 cells. D. Left panel: Western blot showing an efficient siRNA-mediated knockdown of the indicated individual or combined proteins. Right panel: Asynchronous A549 cells were treated with colcemid (0.2μg/ml) for 1h pre-2Gy and the number of gamma H2AX foci was monitored in CenpF− G1 cells after the indicated treatment conditions. At least 50 nuclei were counted. (**) indicates P < 0.001. In all cases, the number of foci measured in non-irradiated cells was subtracted (relative). Shown are the mean +/-SEM for three independent experiments. CTR: control, Olap: olaparib, sc: scrambled RNA, si53: si53BP1, and siR: siRIF1. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/27494840), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Knockdown Validated: 53BP1 Antibody - BSA Free [NB100-305] -
CtIP and MRE11 but not EXO1 are required for end resection in 53BP1-depleted G1 cellsA. A549 cells were treated as in Figure 4 with the indicated siRNAs and efficient knockdown of 53BP1 and/or EXO1 was confirmed by Western blot (upper panel). Lower panel: The focus intensity of RPA was measured in 53BP1- and/or EXO1- depleted cells at 2h post-2Gy in G1 and S/G2. B. Immunoblots showing efficient siRNA-mediated knockdown of CtIP and/or 53BP1 (upper panel), MRE11 and/or 53BP1 (middle panel), or PLK3 and/or 53BP1 (lower panel). (C & D) Quantitation of the percentage of G1 cells with more than three C. RPA or D. CtIP foci after the indicated treatments. At least 50 nuclei were counted. (**) indicates P < 0.001, and (ns) means P > 0.05. Shown are the mean +/-SEM for three independent experiments. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/27494840), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunocytochemistry/ Immunofluorescence: 53BP1 Antibody - BSA Free [NB100-305] -
The production of DSBs depends on Top1 degradation in CPT-treated quiescent cells. (A–C) Serum-starved WI38 hTERT cells were co-transfected with siRNAs against cullin 3 and cullin 4B or against a control sequence and then treated with DMSO (−CPT) or 25 μM CPT (+CPT) for 1 h. (A and B) Western blotting of the indicated proteins. alpha Tubulin: loading control. (C) Number of gamma H2AX foci per nucleus from one representative experiment (246–348 nuclei were analyzed for each treatment) out of three. ***P < 0.001. (D and E) Serum-starved WI38 hTERT cells were treated with DMSO or MG132 (50 μM) for 1 h before exposure to 0.8 Gy IR. One hour post-irradiation, cells were co-stained for gamma H2AX (green) and 53BP1 (red). (D) Representative pictures. (E) Number of gamma H2AX foci per nucleus from one representative experiment (162–180 nuclei were analyzed for each treatment) out of three. Ns: not significant. (F and G) U2OS EV28 cells were treated with DMSO or MG132 (10 μM) for 1 h before the addition of ethanol (untreated) or 300 nM 4-hydroxitamoxifen (4OHT) for 4 h to express AsiSI in the nucleus (42). (F) Representative pictures of cells co-stained for gamma H2AX (green) and 53BP1 (red). (G) ChIP analysis using an anti-gamma H2AX antibody (black) or a non-immune antibody (IgG, gray). Enrichment was assessed by QPCR amplification using primers proximal to two AsiSI sites located inside two genes (Gene I: SFRS6, Gene II: CCD47) and primers distal to an AsiSI site (Control). Enrichment was normalized to the maximum recovery for each experiment (means +/- SEM, n = 3). Ns: not significant; *P < 0.05. In the microscopic images, nuclear contours, identified by DAPI staining (not shown), are indicated by dashed lines. Bars: 10 μm. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/26578593), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunocytochemistry/ Immunofluorescence: 53BP1 Antibody - BSA Free [NB100-305] -
53BP1/RIF1 work epistatically to counteract BRCA1/CtIP-mediated end resection to regulate DSB repair pathway choiceA. HeLa cells harboring pGC were treated with the indicated siRNAs before transfection with I-SceI-expressing vector to induce DSBs. After 48h, the percentage of GFP+ cells was measured as an indication for HR efficiency. B.-D. Asynchronous A549 cells were treated with the indicated siRNAs before irradiation with 2Gy and CtIP B. and RPA foci C. were monitored at 2h while RAD51 D. foci were enumerated at 4h. E. Left panel: representative photos for the colocalization between 53BP1 and RIF1 foci in A549 cells at 2h after 2Gy. Right panel: quantitation of 53BP1, RIF1 or colocalized foci in the absence (DMSO) or presence of ATM inhibitor (ATMi). F. Left panel: representative micrographs for 53BP1 or RIF1 foci in irradiated cells after depletion of either proteins. Right panel: quantitation of experiments presented in the left panel. At least 100 nuclei were counted. In all cases, the number of foci measured in non-irradiated cells was subtracted (relative). Shown are the mean +/-SEM for three independent experiments. sc: scrambled RNA, si53: si53BP1, siR: siRIF1, siB: siBRCA1, and siC: siCtIP. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/27494840), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunocytochemistry/ Immunofluorescence: 53BP1 Antibody - BSA Free [NB100-305] -
Exosomes modulate the repair of DNA DSBs in irradiated recipient cells.(A) Number of 53BP1 foci in BHY cells 1 hour after irradiation with 0 and 2 Gy and transfer of BHY exosomes isolated 24 hours after irradiation with 0 and 6 Gy [n = 5]. (B) Representative images of 53BP1 foci in BHY cells 6 hours after 2 Gy and transfer of BHY exosomes isolated 24 hours after irradiation with 0, 3, 6 or 9 Gy (53BP1 foci green, nuclei blue). (C) Number of 53BP1 foci in BHY cells 6 hours after 2 Gy and transfer of BHY exosomes isolated 24 and 48 hours after irradiation [n1 (control; EXO 0 Gy 24 h; EXO 6 Gy 24 h) = 6, n2 (EXO 0 Gy 48 h; EXO 3 Gy; EXO 6 Gy 48 h; EXO 9 Gy) = 3]. (D) Number of 53BP1 foci in FaDu cells 6 hours after 2 Gy and transfer of FaDu exosomes [n = 3]. (E) Number of 53BP1 foci in FaDu cells 6 hours after 2 Gy and transfer of BHY exosomes [n = 3]. (F) Number of 53BP1 in BHY cells after 2 Gy and transfer of destabilized BHY exosomes. Exosomes from BHY cells isolated 24 hours after irradiation with 0 and 6 Gy were treated with RNase A or a mixture of Triton and Trypsin [n1 (control; intact) = 6; n2 (RNase A 5 μg/μl) = 2; n3 (RNase A 400 μg/μl; Triton + Trypsin) = 3]. For all experiments the +/- SD was shown and p-values calculated on control were considered to be significant if * p < 0.05 and highly significant ** if p < 0.01, while ▲ p < 0.05 and ▲▲ p < 0.01 indicate significant differences to EXO 0 Gy. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/27006994), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunocytochemistry/ Immunofluorescence: 53BP1 Antibody - BSA Free [NB100-305] -
Inhibition of ACLY exacerbates radiation induced DNA damage in HNSCC cells and impairs homologous recombination. (A,B) UM-SCC-6 (A) and UT-SCC-5 (B) cells were treated with 5 uM BMS303141 for 6 h and irradiated with 2, 4, or 6 Gy. Survival was determined by colony formation assay. (C,D) Quantification of cells positive for 53BP1 foci (>5 53BP1 foci/nucleus in a single plane of view) in UM-SCC-6 (C) and UT-SCC-5 (D) cells. Shown are mean percentages of three biological replicates. Immunofluorescent representative images of examples of UM-SCC-6 or UT-SCC-5 cells, 24 h post irradiation, with or without 5 uM BMS303141 pre-treatment. Nuclei in blue, 53BP1 foci in red. (E) Homologous recombination assay. U2OS cells have been stably transfected with a construct containing a GFP gene containing a I-SceI restriction site (SceGFP) as well as a stop codon at the same site. Upon transfection with an I-SceI encoded plasmid, a double strand break is induced at the I-SceI site. Repair of this break via homologous recombination occurs through utilization of the internal GFP (iGFP) fragment downstream of the SceGFP. Successful homologous recombination results in the restoration of a functional GFP gene. (F) U2OS DRGFP cells were treated for 6 h with 5 uM BMS303141 or 5 uM BO2 (Rad51 inhibitor), and transfected with pCBASceI plasmid. 48 h post transfection, cells were harvested and the GFP positive fraction was determined using flow cytometry. Representative images from flow cytometry experiments are shown on the right, with DMSO Sce- as a negative control, and 5 uM B02 Sce+ as a positive control. In the graph, data are shown from two biological replicates. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/31817870), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunocytochemistry/ Immunofluorescence: 53BP1 Antibody - BSA Free [NB100-305] -
53BP1/RIF1 work epistatically to counteract BRCA1/CtIP-mediated end resection to regulate DSB repair pathway choiceA. HeLa cells harboring pGC were treated with the indicated siRNAs before transfection with I-SceI-expressing vector to induce DSBs. After 48h, the percentage of GFP+ cells was measured as an indication for HR efficiency. B.-D. Asynchronous A549 cells were treated with the indicated siRNAs before irradiation with 2Gy and CtIP B. and RPA foci C. were monitored at 2h while RAD51 D. foci were enumerated at 4h. E. Left panel: representative photos for the colocalization between 53BP1 and RIF1 foci in A549 cells at 2h after 2Gy. Right panel: quantitation of 53BP1, RIF1 or colocalized foci in the absence (DMSO) or presence of ATM inhibitor (ATMi). F. Left panel: representative micrographs for 53BP1 or RIF1 foci in irradiated cells after depletion of either proteins. Right panel: quantitation of experiments presented in the left panel. At least 100 nuclei were counted. In all cases, the number of foci measured in non-irradiated cells was subtracted (relative). Shown are the mean +/-SEM for three independent experiments. sc: scrambled RNA, si53: si53BP1, siR: siRIF1, siB: siBRCA1, and siC: siCtIP. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/27494840), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunocytochemistry/ Immunofluorescence: 53BP1 Antibody - BSA Free [NB100-305] -
RAP1 protects telomeres against DNA damageWestern blotting showing the expression of RAP1 and TRF2 in MRC‐5 cells of different population doublings (PD). Senescent cells (PD 72) were left in culture for at least 4 weeks before harvesting for analysis.ChIP analysis of young (PD 30) and senescent (PD 72) MRC‐5 cells performed with either anti‐RAP1, anti‐TRF2, or an IgG antibody. The immunoprecipitated and input products were loaded into a slot blot membrane and hybridized with a telomere probe, and stripped and hybridized to an Alu probe in order to determine unspecific binding. Quantification was performed by normalizing the telomere signal of the immunoprecipitated to that of the input. Data represent mean +/- SD of three biological replicates.Immunofluorescence detection of 53BP1 (red) and a FISH probe staining telomeres (green) in young (PD 26), pre‐senescent (PD 66), and senescent (PD 72 + 4 weeks) MRC‐5 fibroblasts. The upper graph shows the percentage of telomere co‐localizing with 53BP1 (TIFs). Total DNA damage was measured by immunofluorescence detection of 53BP1. The lower graph shows the total number of 53BP1 foci per nucleus. Approximately 40–50 cells were analyzed per replicate and per condition. Bars represent SEM of two biological replicates.Data information: Statistical analyses were performed using Mann–Whitney U‐test (*P < 0.05; **P < 0.001; ***P < 0.0001). Scale bar = 10 μm.Source data are available online for this figure. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/32096305), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Applications for 53BP1 Antibody - BSA Free
Chromatin Immunoprecipitation
Flow Cytometry
Immunocytochemistry/ Immunofluorescence
Immunohistochemistry
Immunohistochemistry-Frozen
Immunohistochemistry-Paraffin
Western Blot
Reviewed Applications
Read 6 reviews rated 4 using NB100-305 in the following applications:
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Formulation
Format
Preservative
Concentration
Shipping
Stability & Storage
Background: 53BP1
References
1.Henry, E., Souissi-Sahraoui, I., Deynoux, M., Lefevre, A., Barroca, V., Campalans, A.,... Arcangeli, M. L. (2019). Human hematopoietic stem/progenitor cells display ROS-dependent long-term hematopoietic defects after exposure to low dose of ionizing radiations. Haematologica. doi:10.3324/haematol.2019.226936
2.Janoshazi, A. K., Horton, J. K., Zhao, M. L., Prasad, R., Scappini, E. L., Tucker, C. J., & Wilson, S. H. (2020). Shining light on the response to repair intermediates in DNA of living cells. DNA Repair (Amst), 85, 102749. doi:10.1016/j.dnarep.2019.102749
Long Name
Alternate Names
Gene Symbol
Additional 53BP1 Products
Product Documents for 53BP1 Antibody - BSA Free
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Product Specific Notices for 53BP1 Antibody - BSA Free
This product is for research use only and is not approved for use in humans or in clinical diagnosis. Primary Antibodies are guaranteed for 1 year from date of receipt.
Related Research Areas
Citations for 53BP1 Antibody - BSA Free
Customer Reviews for 53BP1 Antibody - BSA Free (6)
Have you used 53BP1 Antibody - BSA Free?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
-
Application: ImmunocytochemistrySample Tested: human immortalized breast cancer cellsSpecies: HumanVerified Customer | Posted 06/14/2021HCC1937 cells were treated with PARPi for 96 h. Nuclei are stained in blue (DAPI) and 53BP1 foci in green.Cells were fixed in 4% PFA. 53BP1 Ig was diluted 1:200 and incubated overnight at 4ºC.
-
Application: ImmunocytochemistrySample Tested: Saos-2 human osteosarcoma cell lineSpecies: HumanVerified Customer | Posted 12/30/20194% Paraformaldehyde fixed Saos-2 cells were stained with anti-53BP1 antibody followed by Alexa488 secondary antibody.
-
Application: ImmunocytochemistrySample Tested: Cancer CellsSpecies: HumanVerified Customer | Posted 08/08/2019Cells with 53BP1 bodies after exposure to irradiation 10 Gray. Cells were fixed 2 hours following irradiation. DNA damages are present and the resulting DNA damages clusters are stain thanks to 53BP1 bodies antibodies.1/500
-
Application: Western BlotSample Tested: Whole cell lysatesSpecies: HumanVerified Customer | Posted 06/30/2015Western Blot for 53BP1
-
Application: ImmunocytochemistrySample Tested: AG1522 normal human fibroblastsSpecies: HumanVerified Customer | Posted 11/07/2013
-
Application: ImmunocytochemistrySample Tested: Mouse KeratinocytesSpecies: MouseVerified Customer | Posted 10/06/2011
There are no reviews that match your criteria.
Protocols
View specific protocols for 53BP1 Antibody - BSA Free (NB100-305):
Sample Preparation.
1. Grow cells to 60-85% confluency. Flow cytometry requires between 2 x 105 and 1 x 106 cells for optimal performance.
2. If cells are adherent, harvest gently by washing once with staining buffer and then scraping. Avoid using trypsin as this can disrupt certain epitopes of interest. If enzymatic harvest is required, use Accutase, Collagenase, or TrypLE Express for a less damaging option.
3. Reserve 100 uL for counting, then transfer cell volume into a 50 mL conical tube and centrifuge for 8 minutes at 400 RCF.
a. Count cells using a hemocytometer and a 1:1 trypan blue exclusion stain to determine cell viability before starting the flow protocol. If cells appear blue, do not proceed.
4. Re-suspend cells to a concentration of 1 x 106 cells/mL in staining buffer.
5. Aliquot out 100 uL samples in accordance with your experimental samples.
Tip: When cell surface and intracellular staining are required in the same sample, it is advisable that the cell surface staining be performed first since the fixation and permeabilization steps might reduce the availability of surface antigens.
Intracellular Staining.
Tip: When performing intracellular staining, it is important to use appropriate fixation and permeabilization reagents based upon the target and its subcellular location. Certain targets may require more gentle or transient permeabilization protocols such as the commonly employed methanol or saponin-based methods.
Protocol for Cytoplasmic Targets:
1. Fix the cells by adding 100 uL fixation solution (such as 4% PFA) to each sample for 10-15 minutes.
2. Permeabilize cells by adding 100 uL of a permeabilization buffer to every 1 x 106 cells present in the sample. Mix well and incubate at room temperature for 10 minutes.
a. For cytoplasmic targets, use a gentle permeabilization solution such as 1X PBS + 0.5% Saponin or 1X PBS + 0.5% Tween-20.
b. To maintain the permeabilized state throughout your experiment, use staining buffer + 0.1% of the permeabilization reagent (i.e. 0.1% Tween-20 or 0.1% Saponin).
3. Following the 10-minute incubation, add 2 mL of the staining buffer + 0.1% permeabilizer to each sample.
4. Centrifuge for 1 minute at 400 RCF.
5. Discard supernatant and re-suspend in 100 uL of staining buffer + 0.1% permeabilizer.
6. Add appropriate amounts of each antibody (eg. 1 test or 1 ug per sample, as experimentally determined).
7. Mix well and incubate at room temperature for 30 minutes. Gently mix samples every 10-15 minutes.
8. Following the primary/conjugate incubation, add 1-2 mL/sample of staining buffer +0.1% permeabilizer and centrifuge for 1 minute at 400 RCF.
9. Wash twice by re-suspending cells in staining buffer (2 mL for tubes or 200 uL for wells) and centrifuge at 400 RCF for 5 minutes. Discard supernatant.
10. Add appropriate amount of secondary antibody (as experimentally determined) to each sample.
11. Incubate at room temperature in dark for 20 minutes.
12. Add 1-2 mL of staining buffer and centrifuge at 400 RCF for 1 minute and discard supernatant.
13. Wash twice by re-suspending cells in staining buffer (2 mL for tubes or 200 uL for wells) and centrifuge at 400 RCF for 5 minutes. Discard supernatant.
14. Resuspend in an appropriate volume of staining buffer (usually 500 uL per sample) and proceed with analysis on your flow cytometer.
1. Remove culture medium and wash the cells briefly in PBS. Add 4% paraformaldehyde to the dish and fix at room temperature for 10 minutes.
2. Remove the paraformaldehyde and wash the cells in PBS.
3. Permeabilize the cells with 0.1% Triton X100 or other suitable detergent for 2 min.
4. Remove the permeabilization buffer and wash three times for 5 minutes each in PBS. Be sure to not let the specimen dry out.
5. To block nonspecific antibody binding, incubate in 10% normal goat serum from 1 hour to overnight at room temperature.
6. Add primary antibody at appropriate dilution and incubate overnight at 4C.
7. Remove primary antibody and replace with PBS. Wash three times for 5 minutes each.
8. Add secondary antibody at appropriate dilution. Incubate for 1 hour at room temperature.
9. Remove secondary antibody and replace with PBS. Wash three times for 5 minutes each.
10. Counter stain DNA with DAPI if required.
Bring slides to a boil in 10 mM sodium citrate buffer (pH 6.0) then maintain at a sub-boiling temperature for 10 minutes. Cool slides on bench-top for 30 minutes (keep slides in the sodium citrate buffer at all times).
Staining:
1. Wash sections in deionized water three times for 5 minutes each.
2. Wash sections in PBS for 5 minutes.
3. Block each section with 100-400 ul blocking solution (1% BSA in PBS) for 1 hour at room temperature.
4. Remove blocking solution and add 100-400 ul diluted primary antibody. Incubate overnight at 4 C.
5. Remove antibody solution and wash sections in wash buffer three times for 5 minutes each.
6. Add 100-400 ul HRP polymer conjugated secondary antibody. Incubate 30 minutes at room temperature.
7. Wash sections three times in wash buffer for 5 minutes each.
8. Add 100-400 ul DAB substrate to each section and monitor staining closely.
9. As soon as the sections develop, immerse slides in deionized water.
10. Counterstain sections in hematoxylin.
11. Wash sections in deionized water two times for 5 minutes each.
12. Dehydrate sections.
13. Mount coverslips.
2. Transfer proteins to PVDF membrane according to the instructions provided by the manufacturer of the membrane and transfer apparatus.
3. Stain the membrane with Ponceau S (or similar product) to assess transfer success, and mark molecular weight standards where appropriate.
4. Rinse the blot TBS -0.05% Tween 20 (TBST).
5. Block the membrane in 5% Non-fat milk in TBST (blocking buffer) for at least 1 hour.
6. Wash the membrane in TBST three times for 10 minutes each.
7. Dilute primary antibody in blocking buffer and incubate overnight at 4C with gentle rocking.
8. Wash the membrane in TBST three times for 10 minutes each.
9. Incubate the membrane in diluted HRP conjugated secondary antibody in blocking buffer (as per manufacturer's instructions) for 1 hour at room temperature.
10. Wash the blot in TBST three times for 10 minutes each (this step can be repeated as required to reduce background).
11. Apply the detection reagent of choice in accordance with the manufacturer's instructions.
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- ChIP Protocol Video
- Chromatin Immunoprecipitation (ChIP) Protocol
- Chromatin Immunoprecipitation Protocol
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Liperfluo
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
FAQs for 53BP1 Antibody - BSA Free
-
Q: Hello I want to ask you for an advice in case of antibodies. In our research we focus on DNA repair foci and use your Novus antibodies for phosphorylated histone H2AX (monoclonal mouse) and protein 53BP1(polyclonal rabbit). Now we want to use antobody for phosphorylated 53BP1 protein to increase specifity and I want to ask you which from your NOVUS antibodies will be the best for us.
A:
We currently stock just one antibody to phosphorylated 53BP1, which you can see at the following link: NB100-1803. This is guaranteed for detection of 53BP1 (pSer25) in human and mouse samples by WB, FLOW, ICC/IF,IHC-P, IP and PLA. As this antibody is a rabbit polyclonal, you would be unable to stain for both 53BP1 and its phosphorylated isoform in the same sample, unless you used an antibody from a different host for detection of total 53BP1 or used directly conjugated primaries. Our mouse monoclonal to 53BP1, with catalogue number NBP2-25028, is validated for detection of the human protein by WB, FLOW and IHC. Unfortunately we do not currently stock any other antibodies to phosphorylated 53BP1.
-
Q: We're looking for anti53BP1 antibody covalently labeled with a fluorofor preferabley Cy3. Do you make it?
A:
We sell 53BP1 antibodies available conjugated to DyLight 488, 550 (very similar in spectrum to Cy3) and 650. You may also purchase the unlabeled antibodies and conjugate them to the fluorophores of your choice. The catalog numbers that you may be interested in are: NB100-904R, NB100-304R, NB100-305R.
-
Q: Hello I want to ask you for an advice in case of antibodies. In our research we focus on DNA repair foci and use your Novus antibodies for phosphorylated histone H2AX (monoclonal mouse) and protein 53BP1(polyclonal rabbit). Now we want to use antobody for phosphorylated 53BP1 protein to increase specifity and I want to ask you which from your NOVUS antibodies will be the best for us.
A:
We currently stock just one antibody to phosphorylated 53BP1, which you can see at the following link: NB100-1803. This is guaranteed for detection of 53BP1 (pSer25) in human and mouse samples by WB, FLOW, ICC/IF,IHC-P, IP and PLA. As this antibody is a rabbit polyclonal, you would be unable to stain for both 53BP1 and its phosphorylated isoform in the same sample, unless you used an antibody from a different host for detection of total 53BP1 or used directly conjugated primaries. Our mouse monoclonal to 53BP1, with catalogue number NBP2-25028, is validated for detection of the human protein by WB, FLOW and IHC. Unfortunately we do not currently stock any other antibodies to phosphorylated 53BP1.
-
Q: We're looking for anti53BP1 antibody covalently labeled with a fluorofor preferabley Cy3. Do you make it?
A:
We sell 53BP1 antibodies available conjugated to DyLight 488, 550 (very similar in spectrum to Cy3) and 650. You may also purchase the unlabeled antibodies and conjugate them to the fluorophores of your choice. The catalog numbers that you may be interested in are: NB100-904R, NB100-304R, NB100-305R.

![Western Blot: 53BP1 Antibody - BSA Free [NB100-305] Knockdown Validated: 53BP1 Antibody - BSA Free [NB100-305]](https://resources.rndsystems.com/images/products/53BP1-Antibody-Knockdown-Validated-NB100-305-img0013.jpg)
![Immunocytochemistry/ Immunofluorescence: 53BP1 Antibody - BSA Free [NB100-305] Immunocytochemistry/ Immunofluorescence: 53BP1 Antibody - BSA Free [NB100-305]](https://resources.rndsystems.com/images/products/53BP1-Antibody-Immunocytochemistry-Immunofluorescence-NB100-305-img0008.jpg)
![Immunohistochemistry: 53BP1 Antibody - BSA Free [NB100-305] Immunohistochemistry: 53BP1 Antibody - BSA Free [NB100-305]](https://resources.rndsystems.com/images/products/53BP1-Antibody-Immunohistochemistry-NB100-305-img0005.jpg)
![Western Blot: 53BP1 AntibodyBSA Free [NB100-305] Western Blot: 53BP1 AntibodyBSA Free [NB100-305]](https://resources.rndsystems.com/images/products/53BP1-Antibody-Western-Blot-NB100-305-img0004.jpg)
![Immunocytochemistry/ Immunofluorescence: 53BP1 Antibody - BSA Free [NB100-305] Immunocytochemistry/ Immunofluorescence: 53BP1 Antibody - BSA Free [NB100-305]](https://resources.rndsystems.com/images/products/53BP1-Antibody-Immunocytochemistry-Immunofluorescence-NB100-305-img0012.jpg)
![Flow Cytometry: 53BP1 Antibody - BSA Free [NB100-305] Flow Cytometry: 53BP1 Antibody - BSA Free [NB100-305]](https://resources.rndsystems.com/images/products/53BP1-Antibody-Flow-Cytometry-NB100-305-img0011.jpg)
![Immunocytochemistry/ Immunofluorescence: 53BP1 Antibody - BSA Free [NB100-305] Immunocytochemistry/ Immunofluorescence: 53BP1 Antibody - BSA Free [NB100-305]](https://resources.rndsystems.com/images/products/53BP1-Antibody-Immunocytochemistry-Immunofluorescence-NB100-305-img0007.jpg)
![Immunocytochemistry/ Immunofluorescence: 53BP1 Antibody - BSA Free [NB100-305] Immunocytochemistry/ Immunofluorescence: 53BP1 Antibody - BSA Free [NB100-305]](https://resources.rndsystems.com/images/products/53BP1-Antibody-Immunocytochemistry-Immunofluorescence-NB100-305-img0009.jpg)
![Flow Cytometry: 53BP1 Antibody - BSA Free [NB100-305] Flow Cytometry: 53BP1 Antibody - BSA Free [NB100-305]](https://resources.rndsystems.com/images/products/53BP1-Antibody-Flow-Cytometry-NB100-305-img0006.jpg)
![Western Blot: 53BP1 Antibody - BSA Free [NB100-305] - 53BP1 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb100-305_rabbit-polyclonal-53bp1-antibody-310202415532062.jpg)
![Western Blot: 53BP1 Antibody - BSA Free [NB100-305] - 53BP1 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb100-305_rabbit-polyclonal-53bp1-antibody-31020241553204.jpg)
![Knockdown Validated: 53BP1 Antibody - BSA Free [NB100-305] - 53BP1 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb100-305_rabbit-polyclonal-53bp1-antibody-knockdown-validated-132202617561219.jpg)
![Knockdown Validated: 53BP1 Antibody - BSA Free [NB100-305] - 53BP1 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb100-305_rabbit-polyclonal-53bp1-antibody-knockdown-validated-13220261758417.jpg)
![Immunocytochemistry/ Immunofluorescence: 53BP1 Antibody - BSA Free [NB100-305] - 53BP1 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb100-305_rabbit-polyclonal-53bp1-antibody-immunocytochemistry-immunofluorescence-132202619213321.jpg)
![Immunocytochemistry/ Immunofluorescence: 53BP1 Antibody - BSA Free [NB100-305] - 53BP1 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb100-305_rabbit-polyclonal-53bp1-antibody-immunocytochemistry-immunofluorescence-132202619354813.jpg)
![Immunocytochemistry/ Immunofluorescence: 53BP1 Antibody - BSA Free [NB100-305] - 53BP1 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb100-305_rabbit-polyclonal-53bp1-antibody-immunocytochemistry-immunofluorescence-13220261941180.jpg)
![Immunocytochemistry/ Immunofluorescence: 53BP1 Antibody - BSA Free [NB100-305] - 53BP1 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb100-305_rabbit-polyclonal-53bp1-antibody-immunocytochemistry-immunofluorescence-132202619213313.jpg)
![Immunocytochemistry/ Immunofluorescence: 53BP1 Antibody - BSA Free [NB100-305] - 53BP1 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb100-305_rabbit-polyclonal-53bp1-antibody-immunocytochemistry-immunofluorescence-132202619445811.jpg)
![Immunocytochemistry/ Immunofluorescence: 53BP1 Antibody - BSA Free [NB100-305] - 53BP1 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb100-305_rabbit-polyclonal-53bp1-antibody-immunocytochemistry-immunofluorescence-13220261926172.jpg)