CHD7 Antibody - BSA Free
Novus Biologicals | Catalog # NBP1-77393

![Immunocytochemistry/ Immunofluorescence: CHD7 Antibody - BSA Free [NBP1-77393] Immunocytochemistry/ Immunofluorescence: CHD7 Antibody - BSA Free [NBP1-77393]](https://resources.rndsystems.com/images/products/Chd7-Antibody-Immunocytochemistry-Immunofluorescence-NBP1-77393-img0003.jpg)
Key Product Details
Species Reactivity
Validated:
Cited:
Applications
Validated:
Cited:
Label
Antibody Source
Format
Product Specifications
Immunogen
Reactivity Notes
Localization
Clonality
Host
Isotype
Scientific Data Images for CHD7 Antibody - BSA Free
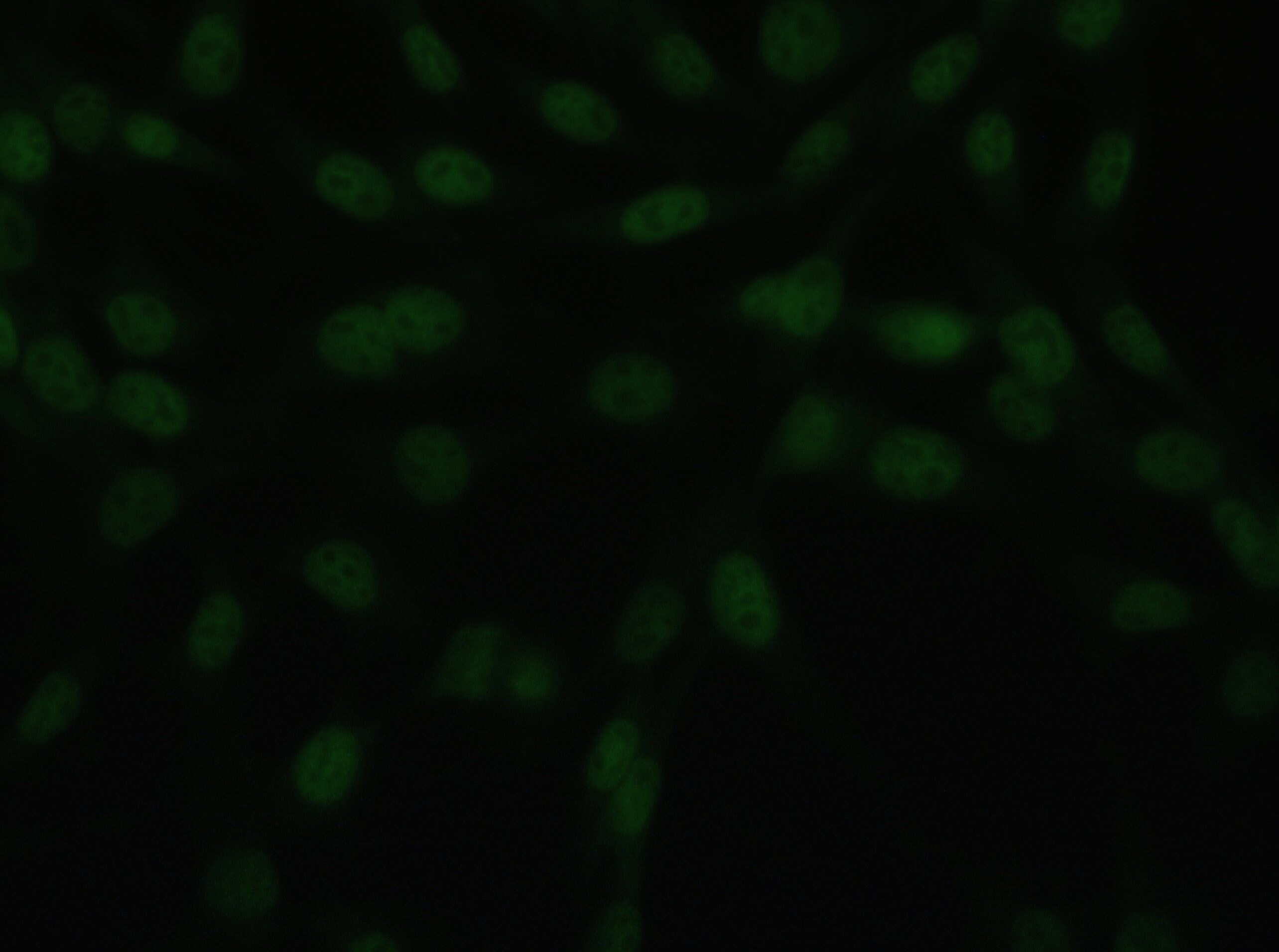
Immunocytochemistry/ Immunofluorescence: CHD7 Antibody - BSA Free [NBP1-77393]
Immunocytochemistry/Immunofluorescence: Chd7 Antibody [NBP1-77393] - Antibody was tested in NIH/3T3 cells with FITC (green). Nuclei and actin were counterstained with Dapi (blue) and Phalloidin (red).Immunohistochemistry: CHD7 Antibody - BSA Free [NBP1-77393]
Immunohistochemistry: Chd7 Antibody [NBP1-77393] - IHC analysis of CHD7 in mouse intestine using DAB with hematoxylin counterstain.Applications for CHD7 Antibody - BSA Free
Immunocytochemistry/ Immunofluorescence
Immunohistochemistry
Immunohistochemistry-Paraffin
Western Blot
Reviewed Applications
Read 1 review rated 5 using NBP1-77393 in the following applications:
Formulation, Preparation, and Storage
Purification
Formulation
Format
Preservative
Concentration
Shipping
Stability & Storage
Background: CHD7
Long Name
Alternate Names
Gene Symbol
UniProt
Additional CHD7 Products
Product Documents for CHD7 Antibody - BSA Free
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Product Specific Notices for CHD7 Antibody - BSA Free
This product is for research use only and is not approved for use in humans or in clinical diagnosis. Primary Antibodies are guaranteed for 1 year from date of receipt.
Citations for CHD7 Antibody - BSA Free
Customer Reviews for CHD7 Antibody - BSA Free (1)
Have you used CHD7 Antibody - BSA Free?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
-
Application: ImmunocytochemistrySample Tested: GN11 cell lineSpecies: MouseVerified Customer | Posted 09/28/2017Fixation: 4% paraformaldehyde in PBS pH 7.4 for 15 minutes at room temperature Permeabilization: PBS containing 0.2% Triton X-100 for 10 minutes Blocking: PBST (PBS+ 0.1% Tween 20) 10% goat serum for 60 minutes Immunostaining: primary Ab 1:100 in PBST in a humidified chamber overnight at 4°C
There are no reviews that match your criteria.
Protocols
View specific protocols for CHD7 Antibody - BSA Free (NBP1-77393):
Immunocytochemistry Protocol
Culture cells to appropriate density in 35 mm culture dishes or 6-well plates.
1. Remove culture medium and add 10% formalin to the dish. Fix at room temperature for 30 minutes.
2. Remove the formalin and add ice cold methanol. Incubate for 5-10 minutes.
3. Remove methanol and add washing solution (i.e. PBS). Be sure to not let the specimen dry out. Wash three times for 10 minutes.
4. To block nonspecific antibody binding incubate in 10% normal goat serum from 1 hour to overnight at room temperature.
5. Add primary antibody at appropriate dilution and incubate at room temperature from 2 hours to overnight at room temperature.
6. Remove primary antibody and replace with washing solution. Wash three times for 10 minutes.
7. Add secondary antibody at appropriate dilution. Incubate for 1 hour at room temperature.
8. Remove antibody and replace with wash solution, then wash for 10 minutes. Add Hoechst 33258 to wash solution at 1:25,0000 and incubate for 10 minutes. Wash a third time for 10 minutes.
9. Cells can be viewed directly after washing. The plates can also be stored in PBS containing Azide covered in Parafilm (TM). Cells can also be cover-slipped using Fluoromount, with appropriate sealing.
Immunohistochemistry-Paraffin Embedded Sections
Antigen Unmasking:
Bring slides to a boil in 10 mM sodium citrate buffer (pH 6.0) then maintain at a sub-boiling temperature for 10 minutes. Cool slides on bench-top for 30 minutes.
Staining:
1. Wash sections in deionized water three times for 5 minutes each.
2. Wash sections in wash buffer for 5 minutes.
3. Block each section with 100-400 ul blocking solution for 1 hour at room temperature.
4. Remove blocking solution and add 100-400 ul diluted primary antibody. Incubate overnight at 4C.
5. Remove antibody solution and wash sections in wash buffer three times for 5 minutes each.
6. Add 100-400 ul biotinylated diluted secondary antibody. Incubate 30 minutes at room temperature.
7. Remove secondary antibody solution and wash sections three times with wash buffer for 5 minutes each.
8. Add 100-400 ul Streptavidin-HRP reagent to each section and incubate for 30 minutes at room temperature.
9. Wash sections three times in wash buffer for 5 minutes each.
10. Add 100-400 ul DAB substrate to each section and monitor staining closely.
11. As soon as the sections develop, immerse slides in deionized water.
12. Counterstain sections in hematoxylin.
13. Wash sections in deionized water two times for 5 minutes each.
14. Dehydrate sections.
15. Mount coverslips.
Western Blot Protocol
1. Perform SDS-PAGE on samples to be analyzed, loading 40 ug of total protein per lane.
2. Transfer proteins to membrane according to the instructions provided by the manufacturer of the membrane and transfer apparatus.
3. Stain according to standard Ponceau S procedure (or similar product) to assess transfer success, and mark molecular weight standards where appropriate.
4. Rinse the blot.
5. Block the membrane using standard blocking buffer for at least 1 hour.
6. Wash the membrane in wash buffer three times for 10 minutes each.
7. Dilute primary antibody in blocking buffer and incubate 1 hour at room temperature.
8. Wash the membrane in wash buffer three times for 10 minutes each.
9. Apply the diluted HRP conjugated secondary antibody in blocking buffer (as per manufacturers instructions) and incubate 1 hour at room temperature.
10. Wash the blot in wash buffer three times for 10 minutes each (this step can be repeated as required to reduce background).
11. Apply the detection reagent of choice in accordance with the manufacturers instructions.
*Note: Tween-20 can be added to the blocking or antibody dilution buffer at a final concentration of 0.05-0.2%.
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
FAQs for CHD7 Antibody - BSA Free
-
Q: I am working with Chd7 and have just visited your website. I would like to have some advices or protocols to use the NBP1-77393 Ab, especially western blot protocol/extraction buffers. My concern is that Chd7 is a high molecular weight protein and it seems very difficult to extract it and to transfer it on a membrane I was just wondering if you had any tips regarding this matter
A: It was a 5% gel ran 100V for 60 min stressing that it was done in cold conditions with ice packs. It should be ok to do overnight at a lower voltage also in cold conditions. We had the best success on NIH3T3 cells, but also tested on Jurkat. In regards to your inquiry about the Western Blot protocol specific for NBP1-77393, we do have one available on our website. You can find this under the protocol tab. In case you have trouble locating this I will provide it to you in this email. These are just suggestions and some additional optimization may need to be done to achieve the best results based on sample preparation and protein expression. Western Blot Protocol: 1. Perform SDS-PAGE on samples to be analyzed, loading 40 ug of total protein per lane. 2. Transfer proteins to membrane according to the instructions provided by the manufacturer of the membrane and transfer apparatus. 3. Stain according to standard Ponceau S procedure (or similar product) to assess transfer success, and mark molecular weight standards where appropriate. 4. Rinse the blot. 5. Block the membrane using standard blocking buffer for at least 1 hour. 6. Wash the membrane in wash buffer three times for 10 minutes each. 7. Dilute primary antibody in blocking buffer and incubate 1 hour at room temperature. 8. Wash the membrane in wash buffer three times for 10 minutes each. 9. Apply the diluted HRP conjugated secondary antibody in blocking buffer (as per manufacturers instructions) and incubate 1 hour at room temperature. 10. Wash the blot in wash buffer three times for 10 minutes each (this step can be repeated as required to reduce background). 11. Apply the detection reagent of choice in accordance with the manufacturers instructions. Note: Tween-20 can be added to the blocking or antibody dilution buffer at a final concentration of 0.05-0.2%.

![Immunohistochemistry: CHD7 Antibody - BSA Free [NBP1-77393] Immunohistochemistry: CHD7 Antibody - BSA Free [NBP1-77393]](https://resources.rndsystems.com/images/products/Chd7-Antibody-Immunohistochemistry-NBP1-77393-img0004.jpg)