GPR56 is a member of the LN-TM7 family of adhesion-type 7-transmembrane (TM) G-protein coupled receptors (GPCR) with long extracellular N-termini (1‑3). The 693 amino acid (aa) human GPR56 contains a 25 aa signal sequence, a 377 aa N-terminal extracellular domain (ECD) and seven TM regions separated by short intracellular and extracellular regions. Like other LN-TM7 members, the ECD contains a highly glycosylated mucin-like stalk followed by a GPCR proteolytic cleavage site (GPS) (1, 4). Cleavage of the 60 kDa N-terminus from the 80 kDa full length form is needed for efficient cell surface expression (5, 6). While the cleaved portion may remain non-covalently associated, it has also been found in conditioned medium of cultured cells (5). Human GPR56 shares 71%, 72%, 80%, 80% and 79% aa identity with mouse, rat, canine, equine, and bovine GPR56 within the cleaved ECD. A functional splice variant lacking the GPS site and a non-functional splice variant lacking portions of the TM domains have also been described (4). A human brain developmental disorder, bilateral frontoparietal polymicrogyria, is associated with GPR56 mutations that also show impaired GPS cleavage, intracellular trafficking, and expression at the cell surface (5). GPR56 is widely distributed, with highest mRNA or expressed sequence tag expression in brain, thyroid, skin and female reproductive system (3, 4). GPR56 expression is upregulated during cell transformation and is high in melanomas, glioblastomas and astrocytomas, but downregulated in melanomas with high metastatic potential (2, 6‑8). Although the function of GPR56 is not completely known, it is clearly an adhesion protein (6‑8). Tissue transglutaminase (TG2) is one reported ligand, binding of which inhibits melanoma growth and metastasis (6). Association of GPR56 with the tetraspanin CD81 stabilizes its complex with Gaq/11 for cell signaling (9).

Key Product Details
Species Reactivity
Validated:
Human
Cited:
Human, Insect
Applications
Validated:
Western Blot, Flow Cytometry, Simple Western, CyTOF-ready
Cited:
Western Blot, Immunocytochemistry
Label
Unconjugated
Antibody Source
Polyclonal Sheep IgG
Loading...
Product Specifications
Immunogen
Chinese hamster ovary cell line CHO-derived recombinant human GPR56
Arg26-Val342
Accession # AAP35975
Arg26-Val342
Accession # AAP35975
Specificity
Detects human GPR56 in direct ELISAs and Western blots. In direct ELISAs, less than 1% cross-reactivity with recombinant human (rh) GPR30, rhGPR114, rhGPR115, rhGPR124, and rhGPR125 is observed.
Clonality
Polyclonal
Host
Sheep
Isotype
IgG
Scientific Data Images for Human GPR56 Antibody
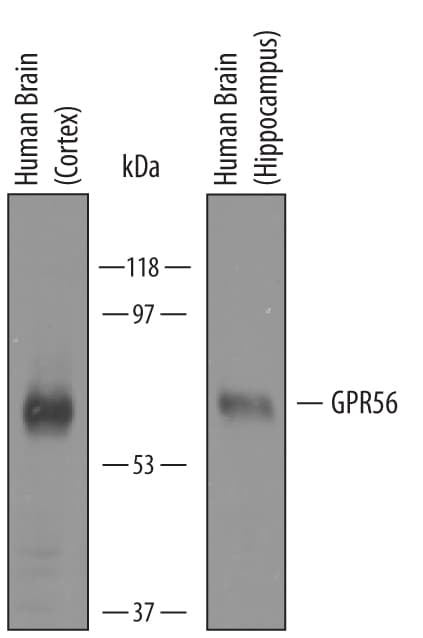
Detection of Human GPR56 by Western Blot.
Western blot shows lysates of human brain (cortex) tissue and human brain (hippocampus) tissue. PVDF membrane was probed with 1 µg/mL of Sheep Anti-Human GPR56 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF4634) followed by HRP-conjugated Anti-Sheep IgG Secondary Antibody (Catalog # HAF016). A specific band was detected for GPR56 at approximately 65 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 8.Detection of GPR56 in Human Blood Lymphocytes by Flow Cytometry.
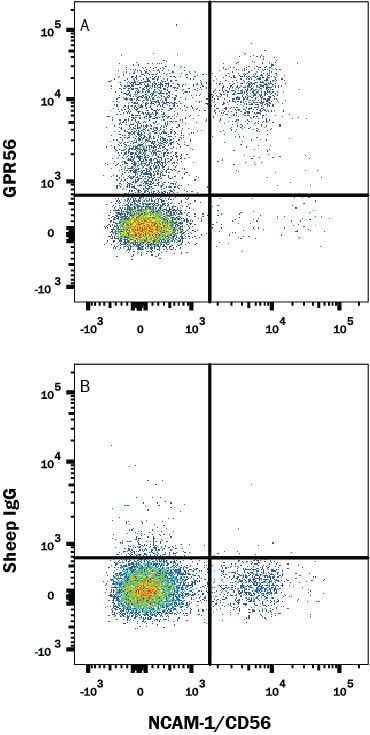
Human peripheral blood lymphocytes were stained with (A) Sheep Anti-Human GPR56 Affinity-purified Polyclonal Antibody (Catalog # AF4634) or (B) control antibody (Catalog # 5-001-A), followed by Phycoerythrin-conjugated Anti-Sheep IgG Secondary Antibody (Catalog # F0126) and Mouse anti-Human CD56 APC-conjugated monoclonal antibody (Catalog # FAB2408A). View our protocol for Staining Membrane-associated Proteins.Detection of Human GPR56 by Simple WesternTM.
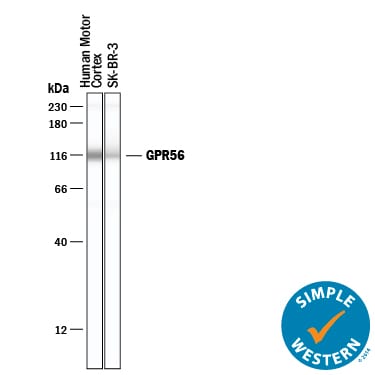
Simple Western lane view shows lysates of human brain (motor cortex) tissue and SK-BR-3 human breast cancer cell line, loaded at 0.2 mg/mL. A specific band was detected for GPR56 at approximately 116 kDa (as indicated) using 10 µg/mL of Sheep Anti-Human GPR56 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF4634) followed by 1:50 dilution of HRP-conjugated Anti-Sheep IgG Secondary Antibody (Catalog # HAF016). This experiment was conducted under reducing conditions and using the 12-230 kDa separation system. Non-specific interaction with the 230 kDa Simple Western standard may be seen with this antibody.Detection of GPR56 by Immunocytochemistry/ Immunofluorescence
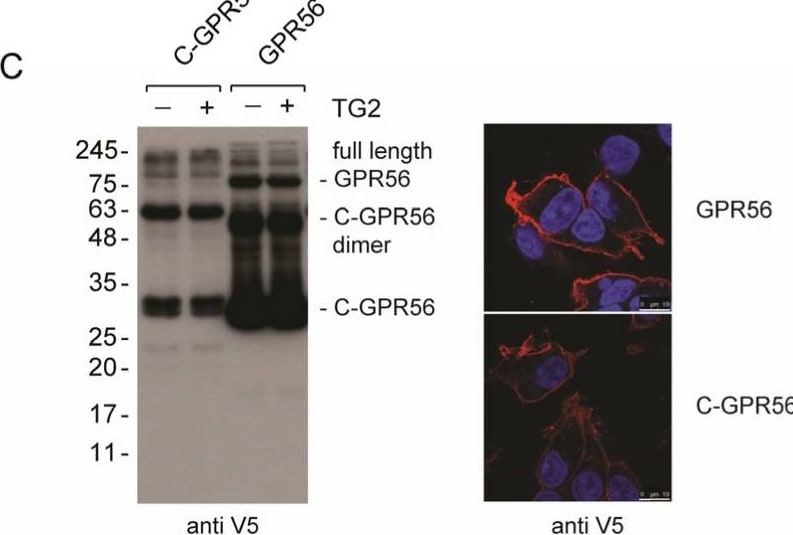
GPR56 activation by TG2 and anti-N-GPR56 antibody requires full length GPR56. (A) Comparison of C230-A TG2 (20 µg/mL), TG2 (20 µg/mL) and anti-N-GPR56 antibody (5 µg/mL) for their ability to activate full-length GPR56-dependent AP-AR shedding. (B) GPR56-dependent activation of shedding by TG2 requires intact GPR56. C-GPR56 is auto-active and not activated by TG2 (n = 3). (C) Western blot panel: Detection of the C-terminal GPR56 fragment in GPR56 and C-GPR56 expressing cells using the V5-epitope antibody. Confocal microscopy panel: Cell surface expression of GPR56 and C-GPR56 using anti-V5 antibody in red. Scale bar 10µm. (D) Luciferase reporter assay to assess TG2 dependent signalling in N-GPR56, GPR56, and C-GPR56-expressing cells. To determine Rho activation, cells were co-transfected with N-GPR56, GPR56, or C-GPR56 plasmid and SRF-RE luciferase reporter. After 48 h post-transfection, cells were stimulated with 20 µg/mL C230A TG2 for 6 h, and luciferase activity was determined in lysates. TG2 activated GPR56 dependent SRF-RE luciferase reporter activity, while C-GPR56 was not stimulated (n = 3). (E) N-GPR56 antibody (5 µg/mL) activates GPR56 dependent SRF-RE luciferase reporter activity. Neither N-GPR56 nor C-GPR56 was stimulated by N-GPR56 antibody treatment. Controls were treated with control sheep IgG (n = 3). (F) Type III collagen (83 nM) did not activate GPR56. N-GPR56 antibody was used as a positive control (n = 3). p values of <0.001 equals (***). Image collected and cropped by CiteAb from the following open publication (https://www.mdpi.com/1422-0067/25/4/2329), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of GPR56 by Immunocytochemistry/ Immunofluorescence
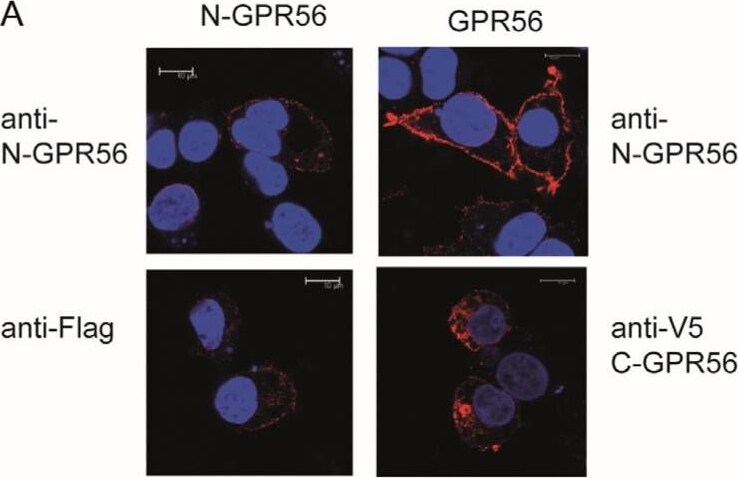
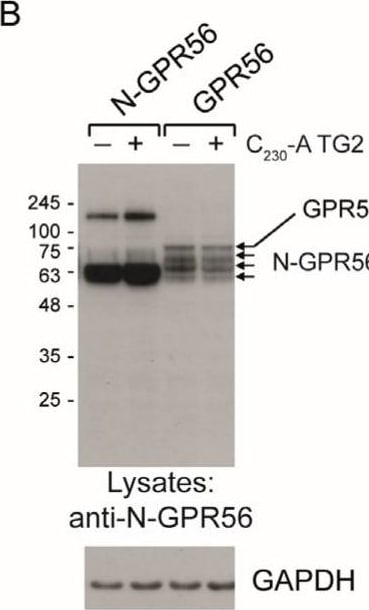
TG2 treatment of GPR56 or N-GPR56-expressing cells does not result in detectable intermolecular crosslinking of the N-terminal GPR56 domain. (A) Optical section acquired by Confocal microscopy showing N-GPR56 and GPR56 expression in transfected cells under non permeabilising conditions (Top panel: anti N-GPR56 antibody with GPR56 signals in red) and permeabilising conditions (Bottom panel: anti Flag antibody for N-GPR56-expressing cells and anti-V5 antibody for GPR56-expressing cells shown in red). Scale bar 10 µm. (B,C) Western blot analysis of cell lysate from AP-AR shedding experiments represented in Figure 3D stained for the N-terminal GPR56 domain and a GAPDH loading control (B) or the V5 epitope tag recognizing C-terminal GPR56 (C). Note that N-GPR56 is not recognized by the V5 antibody. (D) Detection of C-terminal GPR56 fragments required that samples were incubated in SDS sample buffer without heat treatment to avoid loss through aggregation/precipitation. (E,F) Western blot analysis of conditioned medium from AP-AR shedding experiment represented in Figure 3D stained with anti-N-GPR56 antibody or CUB7402 anti-TG2 antibody. Non-specific bands in medium are indicated by asterisks. Image collected and cropped by CiteAb from the following open publication (https://www.mdpi.com/1422-0067/25/4/2329), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of GPR56 by Immunocytochemistry/ Immunofluorescence
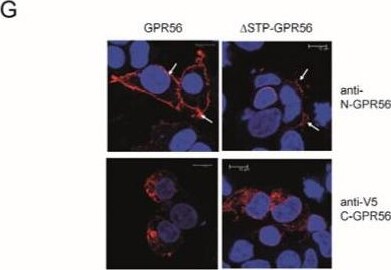
GPR56 activation requires N-terminal GPR56, C-terminal TG2, and is independent of C-terminal GPR56 phosphorylation sites. (A,B) Real-time measurements were conducted whereby the enzymatic reaction was initiated by Ca2+ injection (n = 3). Fluorescence in the absence of Ca2+ activation was subtracted from the fluorescence obtained with activated enzyme to correct for sample bleaching over time. Only the linear part of the fluorescence increase was used, and rates were determined by linear regression. We compared transamidation and isopeptidase activities of wild type TG2 (20 µg/mL) and N-TG2 lacking the beta -barrel domains (20 µg/mL), showing that N-TG2 is catalytically functional. (C) N-TG2 does not activate GPR56- or C-GPR56-dependent Rho activity using SRF-RE reporter assays (n = 3). (D) delta STP-GPR56 lacking the TG2 binding site cannot be activated by TG2 using the AP-AR shedding assay (n = 3). (E,F) Western blot analysis of lysates from delta STP-GPR56 cells shows that it is only partially processed at the GPS-site (large arrow), and N-terminal as well as C-terminal domain aggregates are apparent (small arrows). A diagram of delta STP-GPR56 lacking the TG2 binding site is shown for clarity. (G) delta STP-GPR56 shows little cell surface expression when compared to GPR56 by confocal microscopy. Red stain corresponds to GPR56 staining using either anti-N-GPR56 or anti-V5 antibody. Blue nuclear stain. Scale bar 10µm. (H) GPR56 mutants lacking C-terminal Serine or Threonine tail phosphorylation sites are activated by TG2 using shedding assays (n = 3). p values of <0.01 equals (**), p values of <0.001 equals (***). Image collected and cropped by CiteAb from the following open publication (https://www.mdpi.com/1422-0067/25/4/2329), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of GPR56 by Western Blot
TG2 treatment of GPR56 or N-GPR56-expressing cells does not result in detectable intermolecular crosslinking of the N-terminal GPR56 domain. (A) Optical section acquired by Confocal microscopy showing N-GPR56 and GPR56 expression in transfected cells under non permeabilising conditions (Top panel: anti N-GPR56 antibody with GPR56 signals in red) and permeabilising conditions (Bottom panel: anti Flag antibody for N-GPR56-expressing cells and anti-V5 antibody for GPR56-expressing cells shown in red). Scale bar 10 µm. (B,C) Western blot analysis of cell lysate from AP-AR shedding experiments represented in Figure 3D stained for the N-terminal GPR56 domain and a GAPDH loading control (B) or the V5 epitope tag recognizing C-terminal GPR56 (C). Note that N-GPR56 is not recognized by the V5 antibody. (D) Detection of C-terminal GPR56 fragments required that samples were incubated in SDS sample buffer without heat treatment to avoid loss through aggregation/precipitation. (E,F) Western blot analysis of conditioned medium from AP-AR shedding experiment represented in Figure 3D stained with anti-N-GPR56 antibody or CUB7402 anti-TG2 antibody. Non-specific bands in medium are indicated by asterisks. Image collected and cropped by CiteAb from the following open publication (https://www.mdpi.com/1422-0067/25/4/2329), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of GPR56 by Western Blot
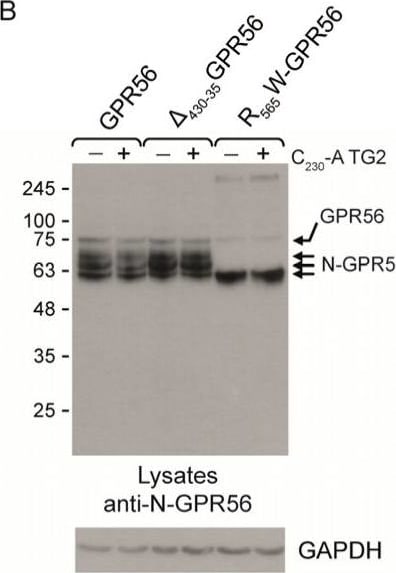
Comparison of the relative signalling activities of wild type GPR56, delta 430-435GPR56, and R565W-GPR56. (A) Comparison of the TG2 signalling response in GPR56, delta 430-435GPR56, and R565W-GPR56-expressing cells measuring SRF-RE activity, and thus RhoA activation. delta 430-435GPR56 is activated, but not R565W-GPR56 (n = 3). (B,C) Western blot analysis of GPR56, delta 430-435GPR56, and R565W-GPR56 using N-GPR56 and V5 epitope antibody in cell lysates, showing correct processing and glycosylation for GPR56 and delta 430-435GPR56, but not R565W-GPR56, which shows a reduced molecular mass for the N-GPR56 fragment. (D) Confocal analysis of delta 430-435GPR56 and R565W-GPR56 cell surface expression levels using N-GPR56 (top panel) and V5 epitope antibodies (bottom panel), demonstrating significant loss of cell surface localisation for R565W-GPR56. Scale bar 10µm. Red GPR56 staining using either anti-N-GPR56 or anti-V5 antibodies. Surface staining for delta 430-435GPR56 is indicated using white arrows. Blue nuclear stain. (E) Comparison of MAPK activation using SRE-RE luciferase activity in response to TG2 stimulation of wild type GPR56 and delta 430-435GPR56 expressing cells (n = 3). p values below 0.05 (*) were considered significant. p values of <0.001 equals (***). Image collected and cropped by CiteAb from the following open publication (https://www.mdpi.com/1422-0067/25/4/2329), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human GPR56 Antibody
Application
Recommended Usage
CyTOF-ready
Ready to be labeled using established conjugation methods. No BSA or other carrier proteins that could interfere with conjugation.
Flow Cytometry
0.25 µg/106 cells
Sample:
Sample:
| Human peripheral blood lymphocytes |
Simple Western
10 µg/mL
Sample: Human brain (motor cortex) tissue and SK‑BR‑3 human breast cancer cell line
Sample: Human brain (motor cortex) tissue and SK‑BR‑3 human breast cancer cell line
Western Blot
1 µg/mL
Sample: Human brain (cortex) tissue and human brain (hippocampus) tissue
Sample: Human brain (cortex) tissue and human brain (hippocampus) tissue
Reviewed Applications
Read 1 review rated 4 using AF4634 in the following applications:
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: GPR56
References
- Fredriksson, R. et al. (2002) FEBS Lett. 531:407.
- Zendman, A.J.W. et al. (1999) FEBS Lett. 446:292.
- Liu, M. et al. (1999) Genomics 55:296.
- Bjarnadottir, T.K. et al. (2007) Gene 387:38.
- Jin, Z. et al. (2007) Hum. Mol. Genet. 16:1972.
- Xu, L. et al. (2006) Proc. Natl. Acad. Sci. USA 103:9023.
- Shashidhar, S. et al. (2005) Oncogene 24:1673.
- Ke, N. et al. (2007) Mol. Cancer Ther. 6:1840.
- Little, K.D. et al. (2004) Mol. Biol. Cell 15:2375.
Long Name
G Protein-coupled Receptor 56
Alternate Names
BFPP, EGF-TM7-like, TM7LN4, TM7XN1
Gene Symbol
ADGRG1
UniProt
Additional GPR56 Products
Product Documents for Human GPR56 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human GPR56 Antibody
For research use only
Related Research Areas
Citations for Human GPR56 Antibody
Customer Reviews for Human GPR56 Antibody (1)
4 out of 5
1 Customer Rating
Have you used Human GPR56 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Showing
1
-
1 of
1 review
Showing All
Filter By:
-
Application: Western BlotSample Tested: Cancer cell lysatesSpecies: HumanVerified Customer | Posted 04/28/2017
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Cellular Response to Hypoxia Protocols
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Liperfluo
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- R&D Systems Quality Control Western Blot Protocol
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...