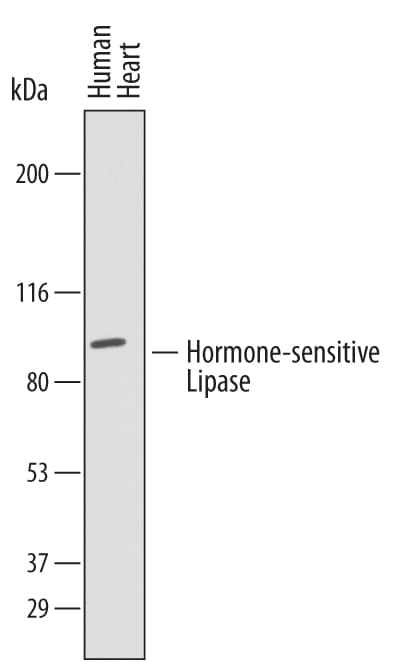
LIPE (also known as hormone sensitive lipase/HSL) is a 88-90 kDa member of the GDXG lipolytic enzyme family of molecules. It is expressed in multiple cell types, including skeletal muscle, adrenal gland and adipocytes, and is regulated by a series of phosphorylations. Catecholamines activate the enzyme via PKA through the phosphorylation of Ser853, Ser855, and Ser951, while insulin depresses its activity via PDE3B. LIPE acts on triglycerides to release free fatty acids, and serves as a retinyl ester hydrolase. Human LIPE is 1076 amino acids (aa) in length, and contains one HSL domain (aa 302-616). This restricted 116 kDa isoform is found in testis (spermatids). There are additional isoform variants. One is 88-90 kDa in size in SDS-Page and possesses an alternative start site to the long form at Met302. This is considered the standard and most common LIPE isoform. A second is approximately 93 kDa in size and contains a 39 aa substitution for aa 1-294. A third isoform is 80 kDa in size and shows a 76 aa deletion in the center of the molecule, reducing its enzymatic activity. It is suggested that LIPE may act as both a monomer and homodimer. Over aa 302-425, human HSL shares 92-93% aa identity with both mouse and rat LIPE.