Human IL-8/CXCL8 Quantikine ELISA Kit
R&D Systems | Catalog # D8000C


Key Product Details
Assay Length
Sample Type & Volume Required Per Well
Sensitivity
Assay Range
Product Summary for Human IL-8/CXCL8 Quantikine ELISA Kit
Product Specifications
Assay Type
Format
Measurement
Detection Method
Conjugate
Species
Specificity
Cross-reactivity
Interference
Sample Values
| Condition | Day 1 (pg/mL) | Day 5 (pg/mL) |
| Unstimulated | 27,000 | 33.000 |
| Stimulated | 73,000 | 102,000 |
Precision
Intra-Assay Precision (Precision within an assay) Three samples of known concentration were tested on one plate to assess intra-assay precision.
Inter-Assay Precision (Precision between assays) hree samples of known concentration were tested in separate assays to assess inter-assay precision.
Cell Culture Supernates
| Intra-Assay Precision | Inter-Assay Precision | |||||
|---|---|---|---|---|---|---|
| Sample | 1 | 2 | 3 | 1 | 2 | 3 |
| n | 20 | 20 | 20 | 20 | 20 | 20 |
| Mean (pg/mL) | 115 | 386 | 802 | 132 | 410 | 817 |
| Standard Deviation | 5.3 | 17.0 | 37.9 | 10.7 | 28.0 | 42.4 |
| CV% | 4.6 | 4.4 | 4.7 | 8.1 | 6.8 | 5.2 |
Citrate Plasma, EDTA Plasma, Heparin Plasma, Serum
| Intra-Assay Precision | Inter-Assay Precision | |||||
|---|---|---|---|---|---|---|
| Sample | 1 | 2 | 3 | 1 | 2 | 3 |
| n | 20 | 20 | 20 | 20 | 20 | 20 |
| Mean (pg/mL) | 168 | 526 | 1093 | 196 | 581 | 1160 |
| Standard Deviation | 9.4 | 28.6 | 70.7 | 14.5 | 56.6 | 70.2 |
| CV% | 5.6 | 5.4 | 6.5 | 7.4 | 9.7 | 6.1 |
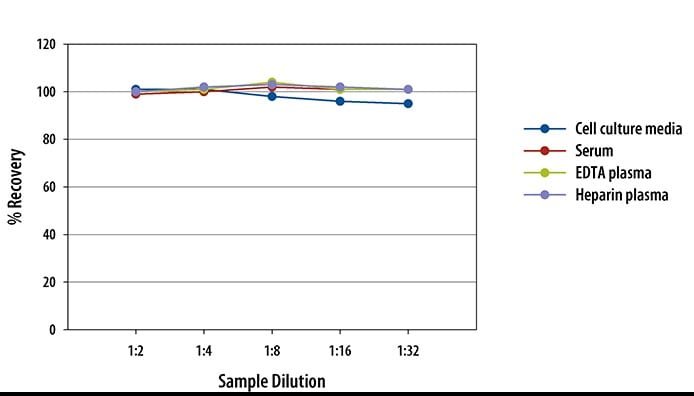
Recovery for Human IL-8/CXCL8 Quantikine ELISA Kit
The recovery of IL-8 spiked to three different levels throughout the range of the assay in various matrices was evaluated.
| Sample Type | Average % Recovery | Range % |
|---|---|---|
| Cell Culture Media (n=5) | 98 | 85-114 |
| Citrate Plasma (n=5) | 105 | 95-114 |
| EDTA Plasma (n=5) | 103 | 97-111 |
| Heparin Plasma (n=5) | 102 | 92-107 |
| Serum (n=5) | 98 | 88-106 |
Linearity
To assess the linearity of the assay, samples were spiked with high concentrations of IL-8 in various matrices and diluted with the appropriate Calibrator Diluent to produce samples with values within the dynamic range of the assay.
Scientific Data Images for Human IL-8/CXCL8 Quantikine ELISA Kit
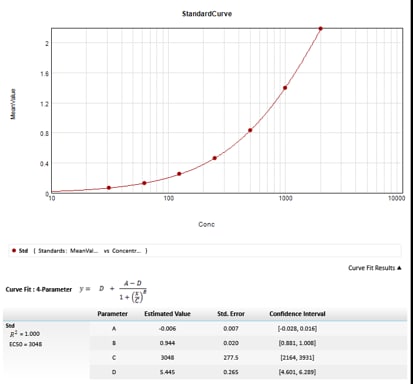
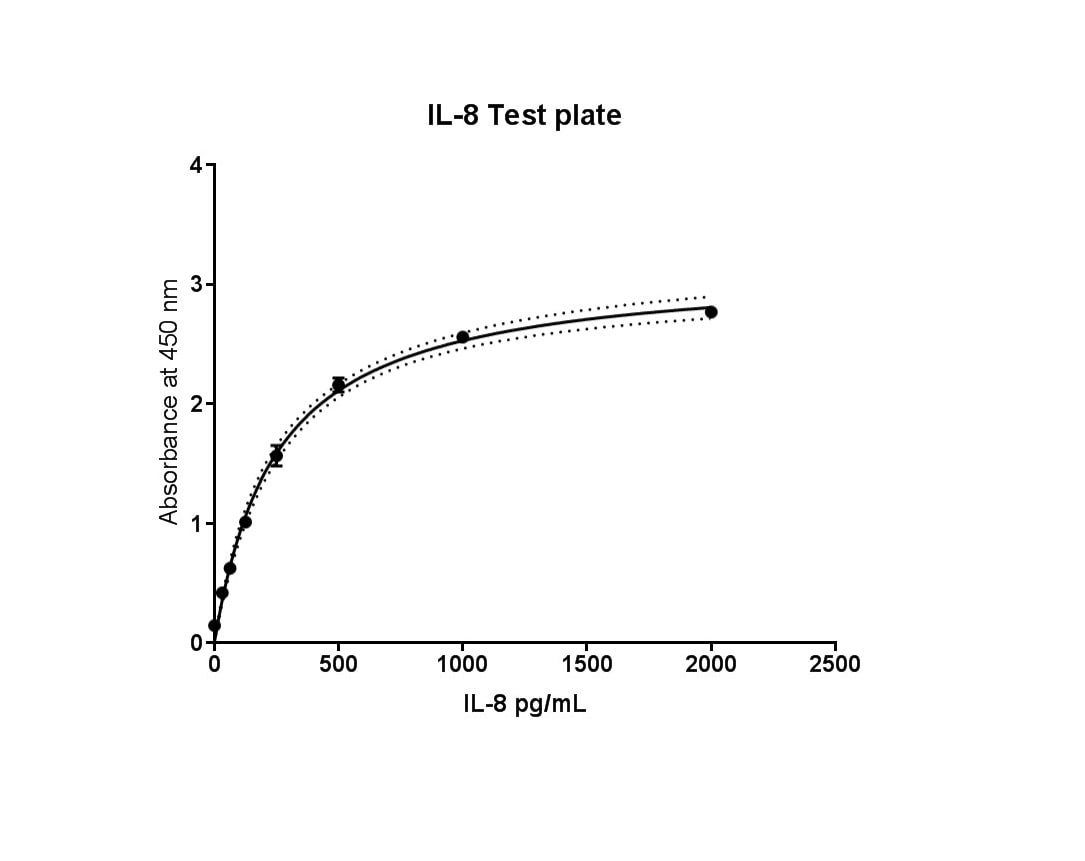
Human CXCL8 ELISA/IL-8 ELISA Cell Culture Supernate Standard Curve
Human CXCL8 ELISA/IL-8 ELISA Serum/Plasma Standard Curve
Human IL-8/CXCL8 Quantikine ELISA Kit
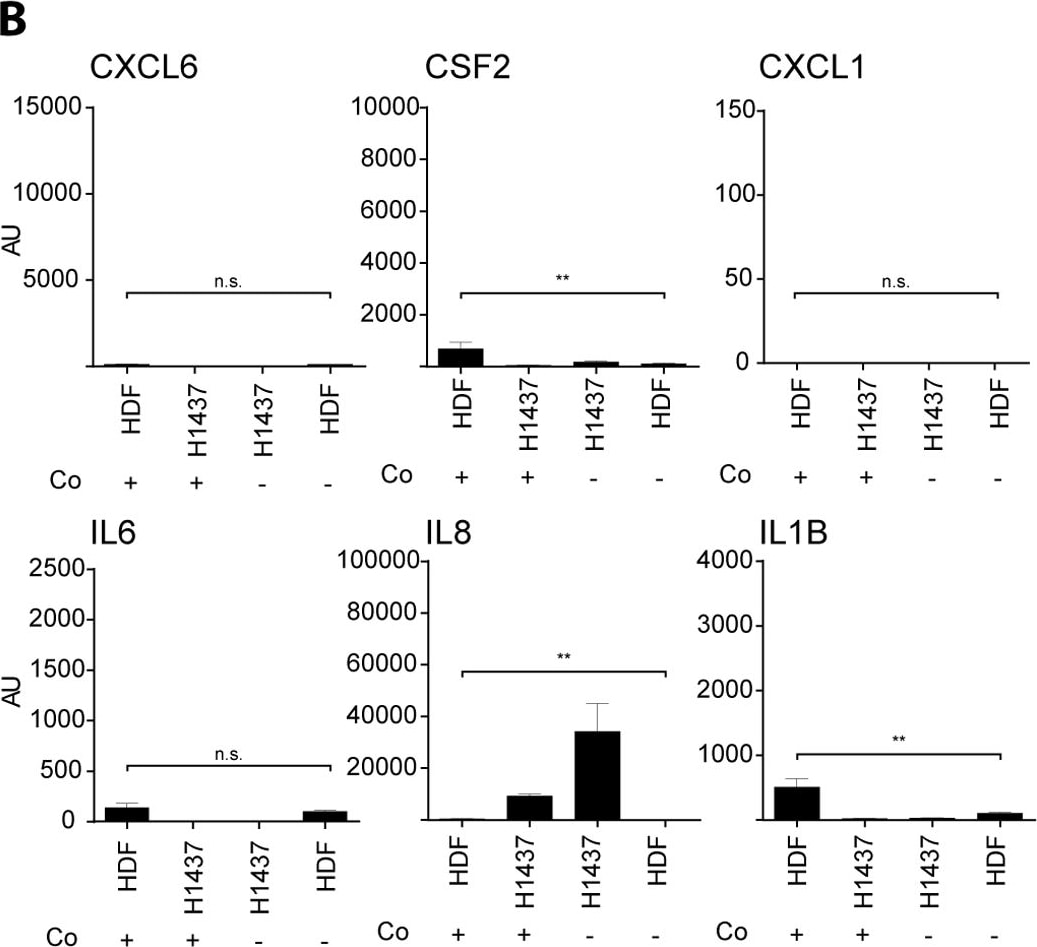
RT-qPCR for a set of cytokines (CSF2, IL6, IL8 and IL1B) and chemokines (CXCL1 and CXCL6) of total RNA samples derived from mono- and co-cultures in a transwell assay.The respective co- (+) or mono- (-) culture is indicated on the X-axis. The respective analyzed cytokine or chemokine is indicated in the header of each graph. Expression values are shown in arbitrary units (AU) and have been normalized to beta-2 microglobulin (B2M) mRNA copies. Experiments were performed in triplicates (n = 3). Statistical analysis was performed on the mean values by unpaired comparison of mono-cultured HDF and co-cultured HDF RNA samples by using Student’s t-test (**p<0.01, ***p<0.001; n.s.: not significant). Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/25919140), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human Human IL-8/CXCL8 Quantikine ELISA Kit by ELISA
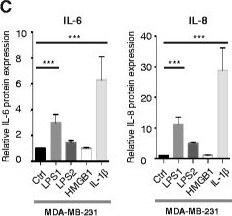
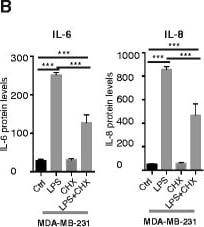
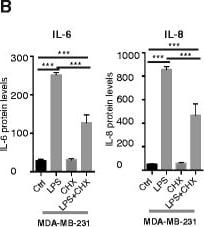
Pathogen-associated molecular patterns (PAMPs) and damage-associated molecular pattern (DAMPs) induce cytokine expression in human breast cancer cells in vitro. a Relative expression of IL-6 and IL-8 mRNAs after 6 h stimulation with lipopolysaccharide 1 (LPS1) and LPS2, using quantitative real-time PCR (QPCR) on mRNA from the cell lines indicated; n = 6. Error bars standard error of the mean (SEM); ***P <0.001 (analysis of variance (ANOVA)). b Release of IL-6 and IL-8 after stimulation with LPS1 and cycloheximide for 6 h, using ELISA on supernatants from stimulated MDA-MB-231 cells; n = 3. c-e Relative release of IL-6 and IL-8 after stimulation with LPS1, LPS2, the DAMP HMGB1 or IL-1 beta for 6 h, using ELISA on supernatants from stimulated MDA-MB-231 cells (c), SUM-149 cells (d) and SUM-159 cells (e); n = 10. Error bars SEM; *P <0.05, ***P <0.001 (ANOVA). f Dual luciferase reporter assays of MDA-MB-231 cells transfected with an NF kappa B reporter. TK-Renilla was co-transfected as control (Ctrl). LPS1, LPS2, HMGB1 or IL-1 beta was added to stimulate NF kappa B activity as described in “Methods”; n = 14. Error bars SEM; ***P <0.001 (ANOVA). g The relative release of IL-6 and IL-8 after stimulation with the DAMP S100A9 for 20 h, using ELISA on supernatants from stimulated MDA-MB-231, SUM-149 and SUM-159 cells; n = 6. Error bars SEM. *P <0.05, ***P <0.001 (ANOVA). h Transient transfection of pDUO-MD2/hTLR4 for 72 h in MCF-7 cells induces release of both IL-6 and IL-8 measured by ELISA; n = 6. Error bars SEM; *P <0.05, ***P <0.001 (Students t test) Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/26392082), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human Human IL-8/CXCL8 Quantikine ELISA Kit by ELISA
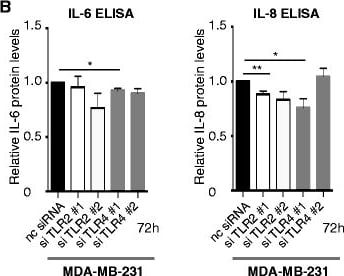
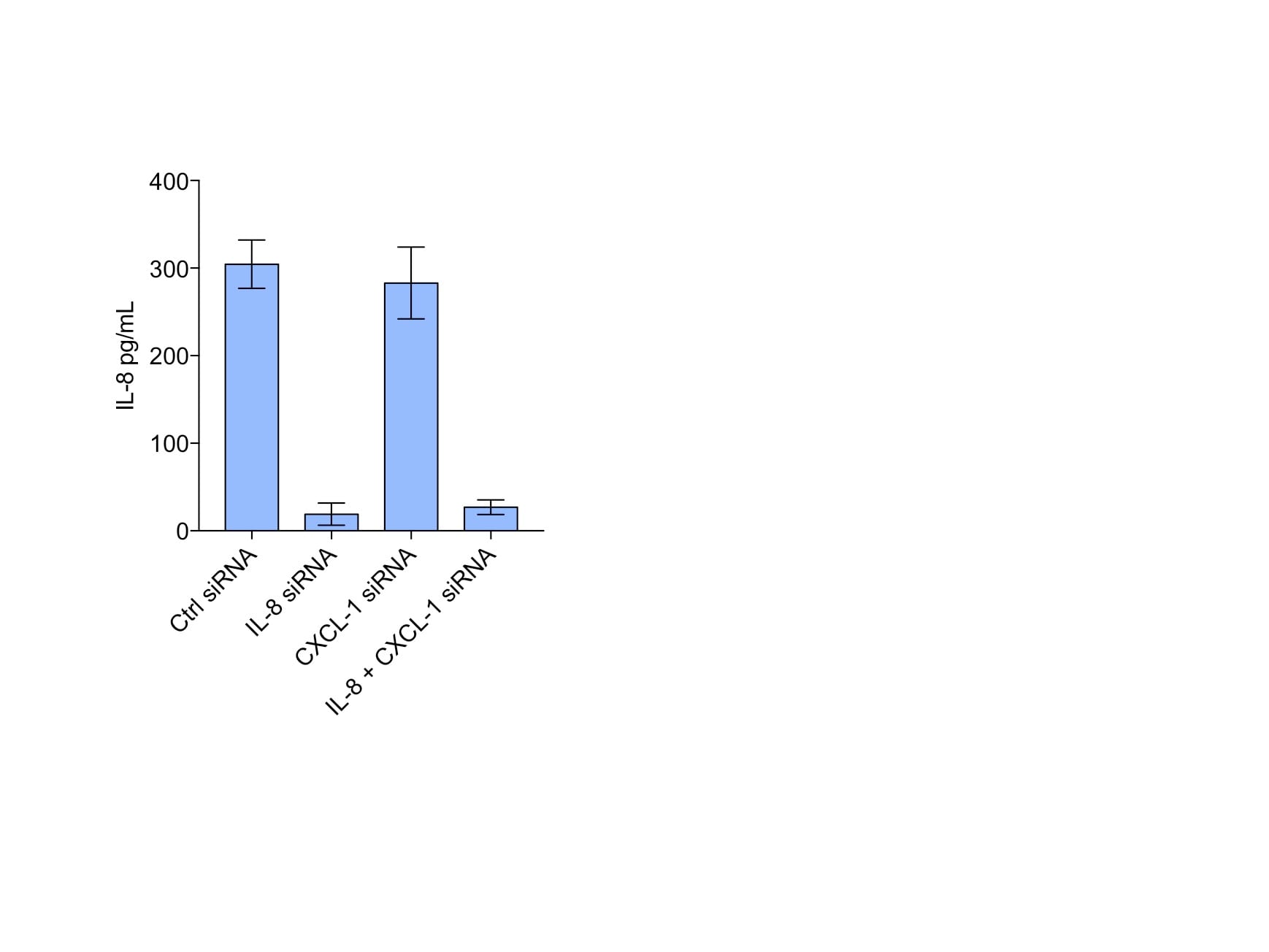
Toll-like receptor 4 (TLR4) silencing decreases endogenous levels of pro-inflammatory cytokines. a Effect of TLR2/4 silencing in breast cancer cells transfected with negative control (nc) siRNA, or siRNA directed against TLR2 mRNA (si#1 and si#2) or TLR4 mRNA (si#1 and si#2) was analyzed using quantitative real-time PCR; ***P <0.001 (analysis of variance (ANOVA)). b IL-6 (left) and IL-8 (right) ELISA on supernatants from MDA-MB-231 breast cancer cells transfected with nc siRNA, or siRNA directed against TLR2 mRNA (si#1 and si#2) or TLR4 mRNA (si#1 and si#2); n = 4. Error bars standard error of the mean (SEM); *P <0.05, **P <0.01, ***P <0.001 (ANOVA). c Boyden chamber migration assays. Migration of primary human myeloid cells towards supernatants from different cell lines indicated. Human primary peripheral blood mononuclear cells were isolated as previously described [48] and allowed to migrate through a Costar Transwell® Permeable Support 8.0-μm 24-well plate (Corning) to the supernatants of breast cancer supernatants cultured under serum-free conditions. Percentage of migrated CD11b+ cells was analyzed using a flow cytometer and CD11b-APC antibodies (BD Sciences); n = 4. Error bars SEM; *P <0.05, **P <0.01, ***P <0.001 (ANOVA). d Matrigel invasion assays. Invasion of lipopolysaccharide (LPS)-stimulated/un-stimulated MDA-MB-231 cells into matrigel invasion chambers (BD Sciences) as indicated: 25 × 103 MDA-MB-231 cells were stimulated or not with LPS and allowed to invade from 72 h. Amount of invaded cells was analyzed using crystal violet staining and manual counting in four separate experiments; n = 4. Error bars SEM; *P <0.05, **P <0.01, ***P <0.001 (Student’s t test) Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/26392082), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human Human IL-8/CXCL8 Quantikine ELISA Kit by ELISA
Pathogen-associated molecular patterns (PAMPs) and damage-associated molecular pattern (DAMPs) induce cytokine expression in human breast cancer cells in vitro. a Relative expression of IL-6 and IL-8 mRNAs after 6 h stimulation with lipopolysaccharide 1 (LPS1) and LPS2, using quantitative real-time PCR (QPCR) on mRNA from the cell lines indicated; n = 6. Error bars standard error of the mean (SEM); ***P <0.001 (analysis of variance (ANOVA)). b Release of IL-6 and IL-8 after stimulation with LPS1 and cycloheximide for 6 h, using ELISA on supernatants from stimulated MDA-MB-231 cells; n = 3. c-e Relative release of IL-6 and IL-8 after stimulation with LPS1, LPS2, the DAMP HMGB1 or IL-1 beta for 6 h, using ELISA on supernatants from stimulated MDA-MB-231 cells (c), SUM-149 cells (d) and SUM-159 cells (e); n = 10. Error bars SEM; *P <0.05, ***P <0.001 (ANOVA). f Dual luciferase reporter assays of MDA-MB-231 cells transfected with an NF kappa B reporter. TK-Renilla was co-transfected as control (Ctrl). LPS1, LPS2, HMGB1 or IL-1 beta was added to stimulate NF kappa B activity as described in “Methods”; n = 14. Error bars SEM; ***P <0.001 (ANOVA). g The relative release of IL-6 and IL-8 after stimulation with the DAMP S100A9 for 20 h, using ELISA on supernatants from stimulated MDA-MB-231, SUM-149 and SUM-159 cells; n = 6. Error bars SEM. *P <0.05, ***P <0.001 (ANOVA). h Transient transfection of pDUO-MD2/hTLR4 for 72 h in MCF-7 cells induces release of both IL-6 and IL-8 measured by ELISA; n = 6. Error bars SEM; *P <0.05, ***P <0.001 (Students t test) Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/26392082), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human Human IL-8/CXCL8 Quantikine ELISA Kit by ELISA
Pathogen-associated molecular patterns (PAMPs) and damage-associated molecular pattern (DAMPs) induce cytokine expression in human breast cancer cells in vitro. a Relative expression of IL-6 and IL-8 mRNAs after 6 h stimulation with lipopolysaccharide 1 (LPS1) and LPS2, using quantitative real-time PCR (QPCR) on mRNA from the cell lines indicated; n = 6. Error bars standard error of the mean (SEM); ***P <0.001 (analysis of variance (ANOVA)). b Release of IL-6 and IL-8 after stimulation with LPS1 and cycloheximide for 6 h, using ELISA on supernatants from stimulated MDA-MB-231 cells; n = 3. c-e Relative release of IL-6 and IL-8 after stimulation with LPS1, LPS2, the DAMP HMGB1 or IL-1 beta for 6 h, using ELISA on supernatants from stimulated MDA-MB-231 cells (c), SUM-149 cells (d) and SUM-159 cells (e); n = 10. Error bars SEM; *P <0.05, ***P <0.001 (ANOVA). f Dual luciferase reporter assays of MDA-MB-231 cells transfected with an NF kappa B reporter. TK-Renilla was co-transfected as control (Ctrl). LPS1, LPS2, HMGB1 or IL-1 beta was added to stimulate NF kappa B activity as described in “Methods”; n = 14. Error bars SEM; ***P <0.001 (ANOVA). g The relative release of IL-6 and IL-8 after stimulation with the DAMP S100A9 for 20 h, using ELISA on supernatants from stimulated MDA-MB-231, SUM-149 and SUM-159 cells; n = 6. Error bars SEM. *P <0.05, ***P <0.001 (ANOVA). h Transient transfection of pDUO-MD2/hTLR4 for 72 h in MCF-7 cells induces release of both IL-6 and IL-8 measured by ELISA; n = 6. Error bars SEM; *P <0.05, ***P <0.001 (Students t test) Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/26392082), licensed under a CC-BY license. Not internally tested by R&D Systems.Preparation and Storage
Shipping
Stability & Storage
Background: IL-8/CXCL8
Long Name
Alternate Names
Gene Symbol
Additional IL-8/CXCL8 Products
Product Documents for Human IL-8/CXCL8 Quantikine ELISA Kit
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human IL-8/CXCL8 Quantikine ELISA Kit
For research use only
⚠ WARNING: This product can expose you to chemicals including N,N-Dimethylforamide, which is known to the State of California to cause cancer. For more information, go to www.P65Warnings.ca.gov.Citations for Human IL-8/CXCL8 Quantikine ELISA Kit
Customer Reviews for Human IL-8/CXCL8 Quantikine ELISA Kit (11)
Have you used Human IL-8/CXCL8 Quantikine ELISA Kit?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
-
Sample Tested: Caco-2 human colorectal adenocarcinoma cell lineVerified Customer | Posted 05/06/2023This product works very well
-
Sample Tested: Cell culture supernatantVerified Customer | Posted 04/24/2023
-
Sample Tested: Urine and PlasmaVerified Customer | Posted 11/07/2022Can detect in normal urine at low levels. Urine was measured neat, 4-fold, and 16-fold. Could not detect in plasma run neat.
-
Sample Tested: human aortic endothelial cellsVerified Customer | Posted 12/06/2019
-
Sample Tested: Human cell conditioned mediumVerified Customer | Posted 01/17/2019Great product! Very easy to use
-
Sample Tested: Cell Culture MediaVerified Customer | Posted 05/02/2018
-
Sample Tested: CHO Chinese hamster ovary cell lineVerified Customer | Posted 04/24/2018
-
Sample Tested: SerumVerified Customer | Posted 04/01/2018
-
Sample Tested: EDTA PlasmaVerified Customer | Posted 01/18/2018
-
Sample Tested: Primary human lung fibroblastsVerified Customer | Posted 12/04/2017This result has been published in PlosONE journal: 10.1371/journal.pone.0058258
-
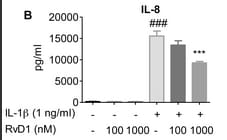
Sample Tested: A375 human melanoma cell lineVerified Customer | Posted 04/26/2017Supernatents of A375-MA2 treated with siCtrl or siIL-8 were assessed using IL-8 ELISA (R&D).
There are no reviews that match your criteria.
Protocols
View specific protocols for Human IL-8/CXCL8 Quantikine ELISA Kit (D8000C):
- Prepare all reagents, standard dilutions, and samples as directed in the product insert.
- Remove excess microplate strips from the plate frame, return them to the foil pouch containing the desiccant pack, and reseal.
- Add 100 µL of Assay Diluent to each well.
- Add 50 µL of Standard, control, or sample to each well. Cover with a plate sealer, and incubate at room temperature for 2 hours.
- Aspirate each well and wash, repeating the process 3 times for a total of 4 washes.
- Add 100 µL of Conjugate to each well. Cover with a new plate sealer, and incubate at room temperature for 1 hour.
- Aspirate and wash 4 times.
- Add 200 µL Substrate Solution to each well. Incubate at room temperature for 30 minutes. PROTECT FROM LIGHT.
- Add 50 µL of Stop Solution to each well. Read at 450 nm within 30 minutes. Set wavelength correction to 540 nm or 570 nm.




Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- ELISA Sample Preparation & Collection Guide
- ELISA Troubleshooting Guide
- How to Run an R&D Systems DuoSet ELISA
- How to Run an R&D Systems Quantikine ELISA
- How to Run an R&D Systems Quantikine™ QuicKit™ ELISA
- Quantikine HS ELISA Kit Assay Principle, Alkaline Phosphatase
- Quantikine HS ELISA Kit Principle, Streptavidin-HRP Polymer
- Sandwich ELISA (Colorimetric) – Biotin/Streptavidin Detection Protocol
- Sandwich ELISA (Colorimetric) – Direct Detection Protocol
- Troubleshooting Guide: ELISA
- View all Protocols, Troubleshooting, Illustrated assays and Webinars