Human/Mouse FGF‑8 Antibody
R&D Systems | Catalog # MAB323

Key Product Details
Species Reactivity
Validated:
Human, Mouse
Cited:
Human, Mouse, Avian - Chicken
Applications
Validated:
Immunohistochemistry, Western Blot, Neutralization
Cited:
Immunohistochemistry, Immunohistochemistry-Paraffin, Western Blot, Immunocytochemistry, In ovo assay
Label
Unconjugated
Antibody Source
Monoclonal Mouse IgG1 Clone # 47109
Loading...
Product Specifications
Immunogen
E. coli-derived recombinant mouse FGF-8b
Gln23-Arg215
Accession # NP_001159834
Gln23-Arg215
Accession # NP_001159834
Specificity
Detects human and mouse FGF-8 in direct ELISAs and Western blots. In direct ELISAs and Western blots, less than 2% cross-reactivity with recombinant human (rh) FGF-5, rhFGF-7, and rhFGF-9 is observed.
Clonality
Monoclonal
Host
Mouse
Isotype
IgG1
Endotoxin Level
<0.10 EU per 1 μg of the antibody by the LAL method.
Scientific Data Images for Human/Mouse FGF‑8 Antibody
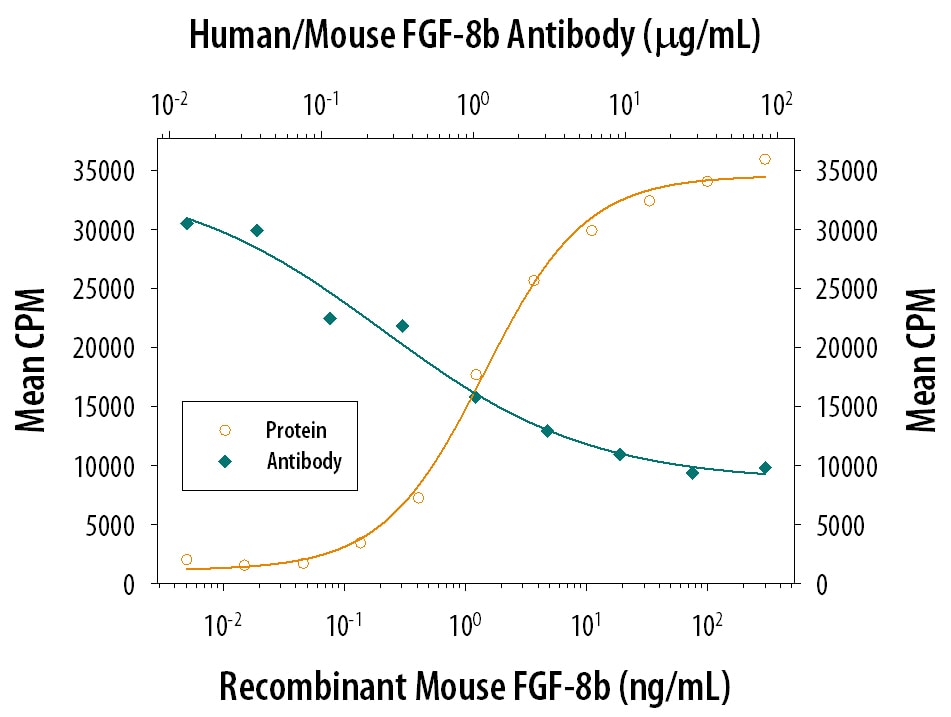
Cell Proliferation Induced by FGF‑8 and Neutralization by Human and Mouse FGF‑8 Antibody.
Recombinant Mouse FGF-8 c Isoform (Catalog # 424-FC) stimulates proliferation in the the NR6R-3T3 mouse fibroblast cell line in a dose-dependent manner (orange line). Proliferation elicited by Recombinant Mouse FGF-8 c Isoform (125 ng/mL) is neutralized (green line) by increasing concentrations of Mouse Anti-Human/Mouse FGF-8 Monoclonal Antibody (Catalog # MAB323). The ND50 is typically 0.25-0.75 µg/mL in the presence of heparin (0.1 µg/mL).FGF‑8 in Human Prostate.
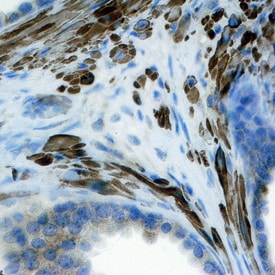
FGF-8 was detected in immersion fixed paraffin-embedded sections of human prostate using Mouse Anti-Human/Mouse FGF-8 Monoclonal Antibody (Catalog # MAB323) at 25 µg/mL overnight at 4 °C. Tissue was stained using the Anti-Mouse HRP-DAB Cell & Tissue Staining Kit (brown; Catalog # CTS002) and counterstained with hematoxylin (blue). Specific staining was localized to stromal cell cytoplasm. View our protocol for Chromogenic IHC Staining of Paraffin-embedded Tissue Sections.Detection of Human/Mouse FGF-8 by Immunohistochemistry
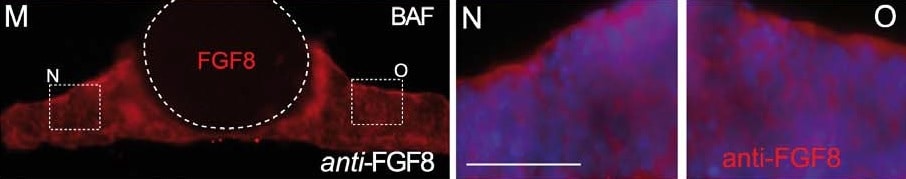
Bafilomycin A1 (BAF) treatment demonstrates the polarization of ERK activity by FGF8 signal activity along the neuraltube. A-C) BAF amplifies ectopic ERK1/2 activity-related FGF8 induction revealing a clear polarized distribution of ERK1/2 activity around FGF8soaked beads depending of the implanted bead; rostral to IsO (C,D,G), or caudal to IsO (C,E). Nonetheles, isthmic organizer morphogenetic activityseems unaffected for Fgf8 (A) and negative regulator Sprouty 2 (B) expressions. Note that PBS bead implantation in control side (blue asterisk in C andF) did not show any ectopic induction. Two hours after bead implantation a clear amplified and almost non-homogeneous ERK1/2 activity wasdetected rostrally in the mesencephalon (rostral to the IsO), which was detected caudally when bead was placed in hindbrain (caudal to the IsO)territories (E). In telencephalic vesicles, caudal to the anr (H) the polarity of ERK activation was reversed. This polarized dpERK detection around thebead is lost at the zli (zona limitans intrathalamica) region (I). Similar symmetric ERK-related FGF8 signal found in zli was seen when placing a FGF8bead in the midbrain of Fgf8 hypomorphic mice (J,K). Importantly FGF8b protein distribution (M) was observed apparently in equal intensity andrange at rostral (N) and caudal (O) sides of the bead (for comparison with PBS bead in panel L). Scale bars are 0,5 mm in A, B, C, H, I, 200 mm in D, E, J,100 mm in F, G, K, L, M, and 50 mm in N, O. Image collected and cropped by CiteAb from the following publication (https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3391221/), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human/Mouse FGF-8 by Immunohistochemistry
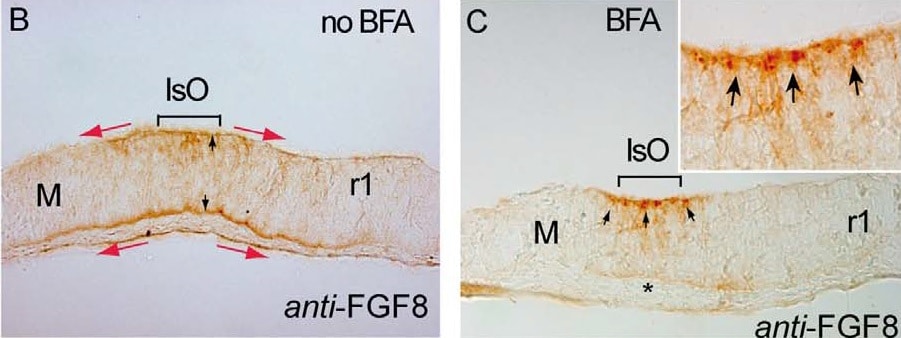
Brefeldin A (BFA) treatment inhibits ERK1/2 activity & modulates differentially Fgf8 negative feed-back regulators. A, B & C) are 12 mM cryostat transversal sections of mouse ONTCs to the isthmic constriction. A & B) are control example before BFA administration to the culture medium showing an ISH for Fgf8 (A) & an Immunostaining for anti-FGF8 (B). Note different domains of expression of the transcript (solid line & arrows) & of the protein FGF8 (red arrows). FGF8 immunodetection is detected both at basal & ventricular sides of the ONTCs (black arrows in B). After 4 hours of BFA incubation (C-J) mRNA of Fgf8 was maintained at IsO (D) while the FGF8 protein profile changed dramatically being accumulated only at the ventricular side (D) as small vesicle-like, (arrows in fig. C & the magnified insert). Moreover, ERK1/2 activity disappears in Isthmic cells & nearby cells (E). Inside this negative gap, FGF8b beads still exerts polarizing ERK1/2 effects (F). Also inside this gap, genes such as Mkp3 (G) & Sef (H) disappear in mesencephalon while Sprouty family genes are maintained (I,J). K) represents experiments & model by which FGF8 planar induction activity coming from mouse FGF8-related secondary organizers (IsO & anr) exerts a different tissue preferential signaling effects (based on the activation of ERK1/2). The direction of polarized ERK1/2 activity depends on location of FGF8-related secondary organizers & the establishment of positional information signaling is dependent mainly on FGF8 negative modulator system, particularly Sprouty2 (blue gradient). FGF8 morphogenetic planar instruction signals coming from rostral (anr) & caudal (IsO) diminish & lose their polarization effect at the diencephalic region (zli) resulting in an equilibrium state. Scale bars is 100 mm. Image collected & cropped by CiteAb from the following publication (https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3391221/), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse Human/Mouse FGF-8 Antibody by Immunohistochemistry
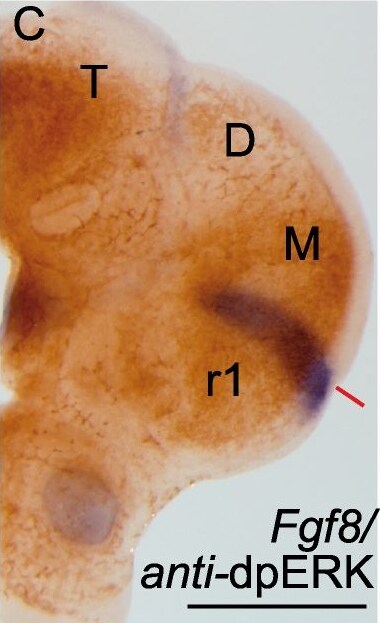
Phosphorylation patterns of ERK1/2 (dpERK) enzymes in the anterior neural tube.Whole mount in situ hybridization (ISH) of E9.5 mouse embryo (A,B,B’) and corresponding organotypic neural tissue cultures of mouse E9.5 anterior neural tube (A’,B”,C-E”; ONTCs; [42]) where it shows the maintenance of gene expression profiles such us Fgf8, Tcf4 (A,A’) (used to delimit main brain subdivisions such the diencephalon (D; Tcf4 positive) and the mesencephalic anlage (M; negative staining), Pax6 D-D” (delimiting di-mesencephalic boundary and rhombomere 1–2 limits) and En2 E-E”. In B-B”) are photomicrographs of E9.5 mouse embryo with anti-dpERK Immunohistochemistry (IHC) taken from lateral (B) and caudal (B’) sides and the corresponding IHC in ONTCs. (C) double staining procedure: ISH (in blue) for Fgf8 and IHC for dpERK (dark brown) to localize inside the dpERK domain the position of the IsO, marked by the solid red line. (D-E”) photomicrographs of same ONTCs in which first a whole mount ISH for Pax6 (D) or Tcf4/En2 (E) were made and afterwards IHC against dpERK. (D’,E’ respectively). Dashed lines mark the main transversal (in black) and longitudinal (in red) brain subdivisions. These ONTCs were cut into transversal sections to the isthmic constriction (D”,E”) to proof that indeed dpERK expression reaches diencephalic anlage (D”; see asterisk; rostral is left) and has a wider expansion than En2 expression (E”; see asterisk; rostral is left). anr is anterior neural ridge secondary organizer; ba is branchial arch; IsO is isthmic organizer; os is optic stalk; ov is otic vesicle; r is rhombomere; T is telencephalon, D, diencephalon, M, mesencephalon. Scale bars are 0,5 mm except in D”, E” they are 100 µm. Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/22792203), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human/Mouse FGF‑8 Antibody
Application
Recommended Usage
Immunohistochemistry
8-25 µg/mL
Sample: Immersion fixed paraffin-embedded sections of human prostate cancer tissue and normal human prostate
Sample: Immersion fixed paraffin-embedded sections of human prostate cancer tissue and normal human prostate
Western Blot
1 µg/mL
Sample: Recombinant Human FGF-8a (Catalog # 4745-F8)
Recombinant Mouse FG-8b (Catalog # 423-F8)
Recombinant Mouse FGF-8c (Catalog # 424-FC)
Recombinant Human FGF-8e (Catalog # 4746-F8)
Recombinant Human FGF-8f (Catalog # 5027-FF)
Sample: Recombinant Human FGF-8a (Catalog # 4745-F8)
Recombinant Mouse FG-8b (Catalog # 423-F8)
Recombinant Mouse FGF-8c (Catalog # 424-FC)
Recombinant Human FGF-8e (Catalog # 4746-F8)
Recombinant Human FGF-8f (Catalog # 5027-FF)
Neutralization
Measured by its ability to neutralize FGF‑8-induced proliferation in the NR6R‑3T3 mouse fibroblast cell line. Rizzino, A. et al. (1988) Cancer Res. 48:4266. The Neutralization Dose (ND50) is typically 0.25-0.75 µg/mL in the presence of 125 ng/mL Recombinant Mouse FGF‑8 c Isoform and 0.1 µg/mL heparin.
Formulation, Preparation, and Storage
Purification
Protein A or G purified from ascites
Reconstitution
Reconstitute at 0.5 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: FGF-8
References
- Mattila, M.M. and P.L. Harkonen (2007) Cytokine Growth Factor Rev. 18:257.
- Reuss, B. and O. von Bohlen und Halbach (2003) Cell Tiss. Res. 313:139.
- Tanaka, A. et al. (1992) Proc. Natl. Acad. Sci. USA 89:8928.
- Gemel, J. et al. (1996) Genomics 35:253.
- Olsen, S.K. et al. (2006) Genes Dev. 20:185.
- Crossley, P.H. et al. (1996) Cell, 84:127.
- Heikinheimo, M. et al. (1994) Mech. Dev. 48:129.
- Sun, X. et al. (1999) Genes Dev. 13:1834.
- Ghosh, A.K. et al. (1996) Cell Growth Differ. 7:1425.
- Mattila, M.M. et al. (2001) Oncogene 20:2791.
- Valve, E. et al. (2000) Int. J. Cancer 88:718.
- Valve, E.M. et al. (2001) Lab. Invest. 81:815.
- Nezu, M. et al. (2005) Biochem. Biophys. Res. Commun. 335:843.
Long Name
Fibroblast Growth Factor 8
Alternate Names
AIGF, FGF8, HBGF-8
Gene Symbol
FGF8
UniProt
Additional FGF-8 Products
Product Documents for Human/Mouse FGF‑8 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human/Mouse FGF‑8 Antibody
For research use only
Related Research Areas
Citations for Human/Mouse FGF‑8 Antibody
Customer Reviews for Human/Mouse FGF‑8 Antibody
There are currently no reviews for this product. Be the first to review Human/Mouse FGF‑8 Antibody and earn rewards!
Have you used Human/Mouse FGF‑8 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars