ACAC-A (Acetyl-CoA carboxylase alpha/1; also ACC-1 and biotin carboxylase) is a 260-265 kDa cytoplasmic, phosphorylated biotinyl-enzyme. It is widely expressed, and found to be concentrated in hepatocytes, adipocytes and lactating mammary epithelium. It is one of two gene products (ACAC-B/beta being the other) that catalyze the formation of malonyl-CoA from acetyl-CoA. The formation of malonyl-CoA by ACAC-A is a rate-limiting step in fatty acid synthesis; malonyl-CoA formed by ACAC-B acts as a regulator of CPT-1 during fatty acidoxidation. Human ACAC-A is 2346 amino acids (aa ) in length. It contains an N-terminal acetylated Met, one ATP-Grasp domain (aa 275-466) with an embedded biotin carboxylation domain (aa 117-618), a biotinyl-binding region (aa 752-818), and a carboxyltransferase domain (aa 1698-2194). There are at least 17 utilized phosphorylation sites, and two acetylated Lys. ACAC-A exists as either a dimer or higher-order oligomer. Multiple splice variants exist. One possesses an alternative start site at Met79, a second utilizes an alternative start site 37 aa upstream of the standard site, and a third (called PIII) shows a 17 aa substitution for aa 1-75. Over aa 1185-1352, human ACAC-A shares 95% aa identity with mouse ACAC-A, and 97% aa identity with both ovine and bovine ACAC-A.
Human/Mouse/Rat Acetyl-CoA
Carboxylase alpha /ACACA Antibody
R&D Systems | Catalog # AF6898

Key Product Details
Species Reactivity
Human, Mouse, Rat
Applications
Western Blot, Simple Western
Label
Unconjugated
Antibody Source
Polyclonal Sheep IgG
Loading...
Product Specifications
Immunogen
E. coli-derived recombinant human Acetyl-CoA Carboxylase alpha /ACACA
Pro1185-Phe1352
Accession # Q13085
Pro1185-Phe1352
Accession # Q13085
Specificity
Detects human Acetyl-CoA Carboxylase alpha /ACACA in direct ELISAs and Western blots.
Clonality
Polyclonal
Host
Sheep
Isotype
IgG
Scientific Data Images for Human/Mouse/Rat Acetyl-CoA
Carboxylase alpha /ACACA Antibody
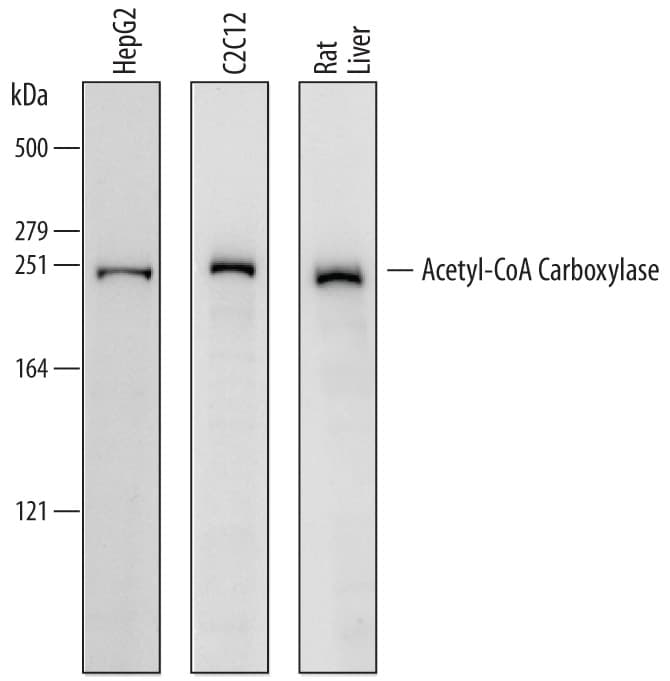
Detection of Human, Mouse, and Rat Acetyl-CoA Carboxylase alpha /ACACA by Western Blot.
Western blot shows lysates of HepG2 human hepatocellular carcinoma cell line, C2C12 mouse myoblast cell line, and rat liver tissue. PVDF membrane was probed with 0.5 µg/mL of Sheep Anti-Human/Mouse/Rat Acetyl-CoA Carboxylase a/ACACA Antigen Affinity-purified Polyclonal Antibody (Catalog # AF6898) followed by HRP-conjugated Anti-Sheep IgG Secondary Antibody (HAF016). A specific band was detected for Acetyl-CoA Carboxylase a/ACACA at approximately 250 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.Detection of Human/Mouse/Rat Acetyl‑CoA Carboxylase alpha /ACACA by Simple WesternTM.
Simple Western lane view shows lysates of A431 human epithelial carcinoma cells and C2C12 mouse myoblast cells, loaded at 0.2 mg/mL. A specific band was detected for Acetyl‑CoA Carboxylase alpha /ACACA at approximately 380-400 kDa (as indicated) using 20 µg/mL of Sheep Anti-Human/Mouse/Rat Acetyl‑CoA Carboxylase alpha /ACACA Antigen Affinity-purified Polyclonal Antibody (Catalog # AF6898) followed by 1:50 dilution of HRP-conjugated Anti-Sheep IgG Secondary Antibody (Catalog # HAF016). This experiment was conducted under reducing conditions and using the 66‑440 kDa separation system.Applications for Human/Mouse/Rat Acetyl-CoACarboxylase alpha /ACACA Antibody
Application
Recommended Usage
Simple Western
20 µg/mL
Sample: A431 human epithelial carcinoma cells and C2C12 mouse myoblast cells
Sample: A431 human epithelial carcinoma cells and C2C12 mouse myoblast cells
Western Blot
0.5 µg/mL
Sample: HepG2 human hepatocellular carcinoma cell line, C2C12 mouse myoblast cell line, and rat liver tissue
Sample: HepG2 human hepatocellular carcinoma cell line, C2C12 mouse myoblast cell line, and rat liver tissue
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Sterile PBS to a final concentration of 0.2 mg/mL. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: Acetyl-CoA Carboxylase alpha/ACACA
Long Name
Acetyl-Coenzyme A Carboxylase alpha
Alternate Names
ACACA, ACC1, ACCA, AcetylCoA Carboxylase alpha
Gene Symbol
ACACA
UniProt
Additional Acetyl-CoA Carboxylase alpha/ACACA Products
Product Documents for Human/Mouse/Rat Acetyl-CoACarboxylase alpha /ACACA Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human/Mouse/Rat Acetyl-CoACarboxylase alpha /ACACA Antibody
For research use only
Citations for Human/Mouse/Rat Acetyl-CoA
Carboxylase alpha /ACACA Antibody
Customer Reviews
There are currently no reviews for this product. Be the first to review Human/Mouse/Rat Acetyl-CoACarboxylase alpha /ACACA Antibody and earn rewards!
Have you used Human/Mouse/Rat Acetyl-CoACarboxylase alpha /ACACA Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Cellular Response to Hypoxia Protocols
- R&D Systems Quality Control Western Blot Protocol
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...

