RGM-C, also known as hemojuvelin, is a member of the repulsive guidance molecule (RGM) family of GPI-linked neuronal and muscle membrane glycoproteins (1, 2). RGM-C is expressed in striated muscle and periportal hepatocytes (3-5). The protein undergoes partial cleavage intracellularly, resulting in a disulfide-linked dimer of the 14 kDa N-terminal and 33 kDa C-terminal portions (4, 6, 7). The N-terminal fragment contains an RGD motif, while the C-terminal fragment carries the GPI attachment site (4, 7). Two alternatively spliced isoforms lack either approximately half or the entire N-terminal fragment. Full length RGM-C can also be released from the cell and circulates in the blood (6, 8). RGM-C is disrupted in type 2A juvenile hemochromatosis, a hereditary iron homeostasis disorder characterized by excessive iron accumulation (5). In mouse, loss of RGM-C function results in decreased expression of the iron regulatory hormone hepicidin and increased iron deposition in liver, pancreas, and heart (5, 9). Membrane associated RGM-C upregulates hepicidin while soluble RGM-C downregulates hepicidin expression (8). This appears to be an iron-responsive regulatory system, as high blood iron levels reduce the amount of soluble RGM-C produced (8). RGM-C, similar to RGM-A, associates with neogenin (7). Disease-related point mutations can prevent internal RGM-C cleavage or its ability to interact with neogenin (6, 7). Experimental inflammatory conditions result in decreased RGM-C expression and increased hepicidin expression, although the two effects occur independently (5, 10). RGM-C also functions as a BMP coreceptor and enhances BMP-2 and BMP-4 signaling (11). In this context, RGM-C enhances the BMP-2 upregulation of hepatic hepicidin (11). Mature human RGM-C shares 89% amino acid (aa) sequence identity with mouse and rat RGM-C. It shares 49% and 44% aa sequence identity with human RGM-A and RGM-B, respectively.
Human/Mouse RGM‑C/Hemojuvelin Antibody
R&D Systems | Catalog # AF3720

Key Product Details
Species Reactivity
Validated:
Cited:
Applications
Validated:
Cited:
Label
Antibody Source
Product Specifications
Immunogen
Gln36-Asp400
Accession # Q6ZVN8
Specificity
Clonality
Host
Isotype
Scientific Data Images for Human/Mouse RGM‑C/Hemojuvelin Antibody
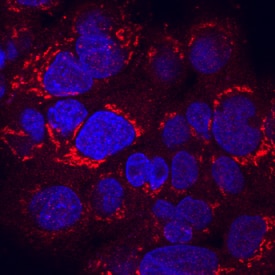
RGM‑C/Hemojuvelin in HepG2 Human Cell Line.
RGM-C/Hemojuvelin was detected in immersion fixed HepG2 human hepatocellular carcinoma cell line using Goat Anti-Human/Mouse RGM-C/Hemojuvelin Antigen Affinity-purified Polyclonal Antibody (Catalog # AF3720) at 10 µg/mL for 3 hours at room temperature. Cells were stained using the NorthernLights™ 557-conjugated Anti-Goat IgG Secondary Antibody (red; Catalog # NL001) and counterstained with DAPI (blue). Specific staining was localized to cytoplasm. View our protocol for Fluorescent ICC Staining of Cells on Coverslips.Applications for Human/Mouse RGM‑C/Hemojuvelin Antibody
Immunocytochemistry
Sample: Immersion fixed HepG2 human hepatocellular carcinoma cell line
Western Blot
Sample:
Recombinant Human RGM-C (Catalog # 3720-RG)
Recombinant Mouse RGM-C (Catalog # 3634-RG)
Formulation, Preparation, and Storage
Purification
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Formulation
Shipping
Stability & Storage
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: RGM-C/Hemojuvelin
References
- Papanikolaou, G. et al. (2004) Nat. Genet. 36:77.
- Schmidtmer, J. and D. Engelkamp (2004) Gene Exp. Patterns 4:105.
- Oldekamp, J. et al. (2004) Gene Exp. Patterns 4:283.
- Niederkofler, V. et al. (2004) J. Neurosci. 24:808.
- Niederkofler, V. et al. (2005) J. Clin. Invest. 115:2180.
- Kuninger, D. et al. (2006) J. Cell Sci. 119:3273.
- Zhang, A.S. et al. (2005) J. Biol. Chem. 280:33885.
- Lin, L. et al. (2005) Blood 106:2884.
- Huang, F.W. et al. (2005) J. Clin. Invest. 115:2187.
- Krijt, J. et al. (2004) Blood 104:4308.
- Babitt, J.L. et al. (2006) Nat. Genet. 38:531.
Long Name
Alternate Names
Gene Symbol
UniProt
Additional RGM-C/Hemojuvelin Products
Product Documents for Human/Mouse RGM‑C/Hemojuvelin Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human/Mouse RGM‑C/Hemojuvelin Antibody
For research use only
Related Research Areas
Citations for Human/Mouse RGM‑C/Hemojuvelin Antibody
Customer Reviews for Human/Mouse RGM‑C/Hemojuvelin Antibody
There are currently no reviews for this product. Be the first to review Human/Mouse RGM‑C/Hemojuvelin Antibody and earn rewards!
Have you used Human/Mouse RGM‑C/Hemojuvelin Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
