Human Placental Lactogen (abbreviated PL or hPL), also called chorionic somatomammotropin hormone 1 (abbreviated CSH1), is a member of the prolactin/growth hormone (PRL/GH) family (1). It is found in a cluster of growth hormones on chromosome 17 that appear to have a common ancestry. Identical 191 amino acid (aa) mature hPL proteins may be formed from one of two genes (2). PL contains a pair of C-terminal cysteines that may form either intra- or interchain disulfides. Human PL shares 98% aa identity with chimpanzee PL and >85% aa sequence identity with other human growth hormones, but only ~25% aa identity with mouse, ovine or bovine PL. PL is mainly expressed by cells in the syncytiotrophoblast layer of the placenta, which produce increasing amounts of PL as pregnancy proceeds. The major portion enters the maternal circulation, where it joins GH2 (placenta-specific GH) in replacing the functions of pituitary GH during pregnancy. A smaller amount of PL circulates in the fetus. Primate PL shows high affinity for the PRL receptor and low affinity for the GH receptor (1). Reduced stimulation of PL by angiotensin 2 correlates with intrauterine growth restriction (3). There is some evidence that mature angiogenic PL may be cleaved to form an anti-angiogenic N-terminal fragment (4). Although PL promotes pancreatic beta cell survival, it does not appear to be altered in gestational diabetes. It helps prepare mammaries for lactation, but probably does not influence lactation itself. PL may be a ligand of stabilin-1, which has been proposed to regulate PL internalization and degradation or re-expression (6).
Human Placental Lactogen/CSH1 Antibody
R&D Systems | Catalog # MAB5757

Key Product Details
Species Reactivity
Validated:
Human
Cited:
Human
Applications
Validated:
Immunohistochemistry, Western Blot
Cited:
Immunohistochemistry, Western Blot, Immunocytochemistry
Label
Unconjugated
Antibody Source
Monoclonal Mouse IgG1 Clone # 658230
Loading...
Product Specifications
Immunogen
Chinese hamster ovary cell line CHO-derived recombinant human Placental Lactogen/CSH1
Val27-Phe217
Accession # P01243
Val27-Phe217
Accession # P01243
Specificity
Detects human Placental Lactogen/CSH1 in direct ELISAs and Western blot. In direct ELISAs, no cross-reactivity with recombinant
human Growth Hormone is observed.
Clonality
Monoclonal
Host
Mouse
Isotype
IgG1
Scientific Data Images for Human Placental Lactogen/CSH1 Antibody
Detection of Human Placental Lactogen/CSH1 by Western Blot.
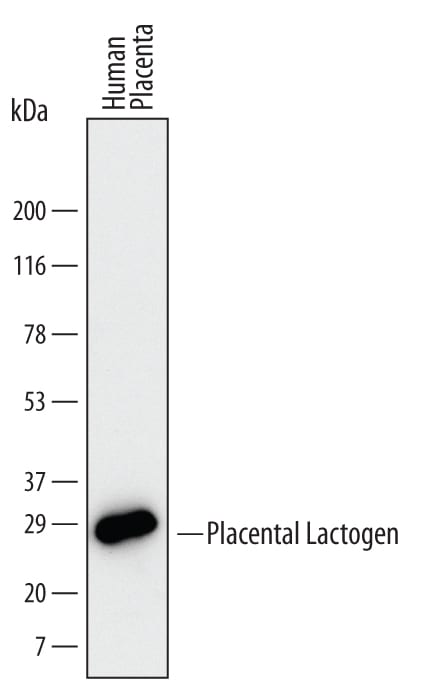
Western blot shows lysates of human placenta tissue. PVDF Membrane was probed with 2 µg/mL of Human Placental Lactogen/CSH1 Monoclonal Antibody (Catalog # MAB5757) followed by HRP-conjugated Anti-Mouse IgG Secondary Antibody (Catalog # HAF007). A specific band was detected for Placental Lactogen/CSH1 at approximately 25 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.Placental Lactogen/CSH1 in Human Placenta.
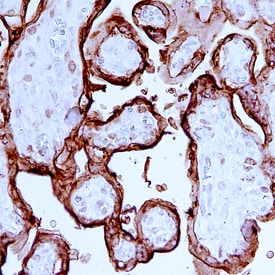
Placental Lactogen/CSH1 was detected in immersion fixed paraffin-embedded sections of human placenta using Human Placental Lactogen/CSH1 Monoclonal Antibody (Catalog # MAB5757) at 15 µg/mL overnight at 4 °C. Before incubation with the primary antibody, tissue was subjected to heat-induced epitope retrieval using Antigen Retrieval Reagent-Basic (Catalog # CTS013). Tissue was stained using the Anti-Mouse HRP-DAB Cell & Tissue Staining Kit (brown; Catalog # CTS002) and counterstained with hematoxylin (blue). Specific staining was localized to cytoplasm in syncytiotrophoblast cells. View our protocol for Chromogenic IHC Staining of Paraffin-embedded Tissue Sections.Applications for Human Placental Lactogen/CSH1 Antibody
Application
Recommended Usage
Immunohistochemistry
8-25 µg/mL
Sample: Immersion fixed paraffin-embedded sections of human placenta
Sample: Immersion fixed paraffin-embedded sections of human placenta
Western Blot
2 µg/mL
Sample: Human placenta tissue
Sample: Human placenta tissue
Formulation, Preparation, and Storage
Purification
Protein A or G purified from hybridoma culture supernatant
Reconstitution
Sterile PBS to a final concentration of 0.5 mg/mL. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: Placental Lactogen/CSH1
References
- Handwerger, S. and M. Freemark (2000) J. Ped. Endocrinol. Metab. 13:343.
- Selby, M.J. et al. (1984) J. Biol. Chem. 259:13131.
- Szukiewicz, D. et al. (2008) Int. Immunopharmacol. 8:177.
- Struman, I. et al. (1999) Proc. Natl. Acad. Sci. USA 96:1246.
- Fujinaka, Y. et al. (2007) J. Biol. Chem. 282:30707.
- Kzhyshkowska, J. et al. (2008) J. Immunol. 180:3028.
Alternate Names
Choriomammotropin, CSH1, CSMT, Lactogen, PL
Entrez Gene IDs
1442 (Human)
Gene Symbol
CSH1
UniProt
Additional Placental Lactogen/CSH1 Products
Product Documents for Human Placental Lactogen/CSH1 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human Placental Lactogen/CSH1 Antibody
For research use only
Citations for Human Placental Lactogen/CSH1 Antibody
Customer Reviews for Human Placental Lactogen/CSH1 Antibody
There are currently no reviews for this product. Be the first to review Human Placental Lactogen/CSH1 Antibody and earn rewards!
Have you used Human Placental Lactogen/CSH1 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...