Mouse GDF‑8/Myostatin Propeptide Antibody
R&D Systems | Catalog # AF1539

Key Product Details
Species Reactivity
Validated:
Mouse
Cited:
Human, Mouse, Transgenic Mouse
Applications
Validated:
Western Blot, Neutralization
Cited:
Western Blot, Immunoprecipitation
Label
Unconjugated
Antibody Source
Polyclonal Sheep IgG
Loading...
Product Specifications
Immunogen
Mouse myeloma cell line NS0-derived recombinant mouse GDF‑8/Myostatin Propeptide
Asn25-Ser265
Accession # O08689
Asn25-Ser265
Accession # O08689
Specificity
Detects GDF‑8/Myostatin Propeptide in direct ELISAs and Western blots. In direct ELISAs, less than 10% cross‑reactivity with mature recombinant mouse (rm) GDF-8 is observed and less than 5% cross-reactivity with mature rmGDF-1, -3, -5, -6, -7, -9, and recombinant human GDF-11 is observed.
Clonality
Polyclonal
Host
Sheep
Isotype
IgG
Endotoxin Level
<0.10 EU per 1 μg of the antibody by the LAL method.
Scientific Data Images for Mouse GDF‑8/Myostatin Propeptide Antibody
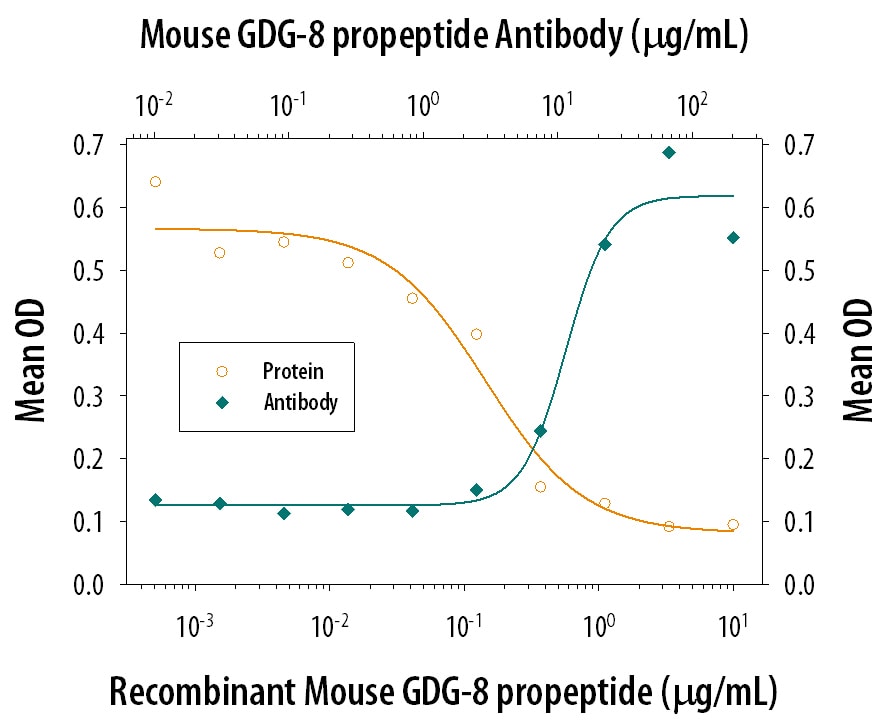
GDF‑8 Propeptide Inhibition of GDF‑8/Myostatin-induced Hemoglobin Expression and Neutralization by Mouse GDF‑8 Propeptide Antibody.
Recombinant Mouse GDF-8 Propeptide (Catalog # 1539-PG) inhibits Recombinant Mouse GDF-8/Myostatin (Catalog # 788-G8) induced hemoglobin expression in the K562 human chronic myelogenous leukemia cell line in a dose-dependent manner (orange line), as measured by the psuedoperoxidase assay. Inhibition of Recombinant Mouse GDF-8/Myostatin (40 ng/mL) activity elicited by Recombinant Mouse GDF-8 Propeptide (800 ng/mL) is neutralized (green line) by increasing concentrations of Sheep Anti-Mouse GDF-8/Myostatin Propeptide Antigen Affinity-purified Polyclonal Antibody (Catalog # AF1539). The ND50 is typically 10-30 µg/mL.Detection of Mouse GDF-8/Myostatin by Western Blot
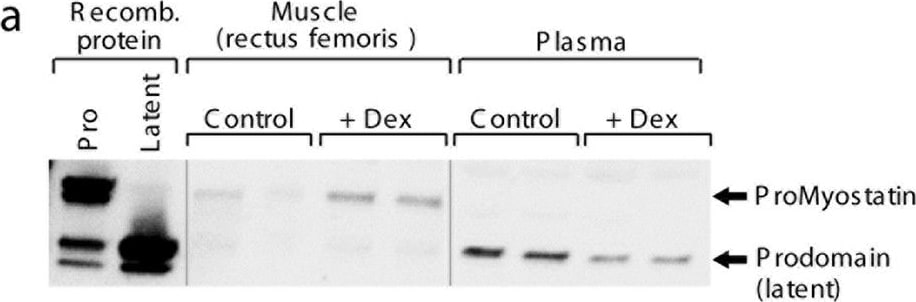
Myostatin precursors redistribute during atrophy. (a) Western blot using a polyclonal antibody raised to the prodomain of myostatin. Recombinant protein controls (proMyostatin and latent myostatin) are used to visualize the migration of proMyostatin and myostatin prodomain bands. In muscle from a pilot experiment in which mice were administered dexamethasone for 15 days, proMyostatin levels increase in muscle, while the levels of latent myostatin in plasma (inferred from the prodomain band) decrease. (b) In a follow-on experiment, animals were administered either vehicle (No Dex) or dexamethasone in their drinking water for two weeks, and given a single 20 mg/kg dose of test antibodies (SRK-015 or IgG control) at day 1. Differences in gastrocnemius muscle weights (expressed as percent difference from the mean of the IgG (no Dex) control group) are shown for days 4, 6, 8 and 15. Individual data points (n = 8–10 animals) along with means +/− standard deviations are shown. Group means were compared by one-way ANOVA followed by a Holm-Sidak test. (c–e) Quantitation of proMyostatin and latent myostatin levels in murine muscle and serum at 4, 6, 8, and 15 days following either dexamethasone or vehicle administration. For all data presented, a minimum of three biological replicates were measured to generate the presented average values, and error bars on all graphs represent standard deviations. Statistical significance was determined by t test (two-tailed, homoscedastic). Image collected and cropped by CiteAb from the following publication (https://www.nature.com/articles/s41598-018-20524-9), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Mouse GDF‑8/Myostatin Propeptide Antibody
Application
Recommended Usage
Western Blot
0.1 µg/mL
Sample: Recombinant Mouse GDF‑8/Myostatin Propeptide (Catalog # 1539-PG)
Sample: Recombinant Mouse GDF‑8/Myostatin Propeptide (Catalog # 1539-PG)
Neutralization
Measured by its ability to neutralize GDF‑8 Propeptide inhibition of GDF‑8/Myostatin-dependent hemoglobin expression in the K562 human chronic myelogenous leukemia cell line. The Neutralization Dose (ND50) is typically 10-30 µg/mL in the presence of 800 ng/mL Recombinant Mouse GDF‑8 Propeptide and 40 ng/mL Recombinant Mouse GDF‑8/Myostatin.
Reviewed Applications
Read 1 review rated 5 using AF1539 in the following applications:
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: GDF-8/Myostatin
References
- McPherron, A.C. (2010) Immunol. Endocr. Metab. Agents Med. Chem. 10:217.
- McPherron, A.C. et al. (1997) Nature 387:83.
- Zimmers, T.A. et al. (2002) Science 296:1486.
- Wolfman, N.M. et al. (2003) Proc. Natl. Acad. Sci. 100:15842.
- McFarlane, C. et al. (2005) Dev. Biol. 283:58.
- Lee, S.J. et al. (2001) Proc. Natl. Acad. Sci. 98:9306.
- Thies, R.S. et al. (2001) Growth Factors 18:251.
- Amthor, H. et al. (2004) Dev. Biol. 270:19.
- Hill, J.J. et al. (2002) J. Biol. Chem. 277:40735.
- Miura, T. et al. (2006) Biochem. Biophys. Res. Commun. 340:675.
- Hill, J.J. et al. (2003) Molecular Endocrinology 17:1144.
- Anderson, S.B. et al. (2008) J. Biol. Chem. 283:7027.
- Rebbapragada, A. et al. (2003) Mol. Cell. Biol. 23:7230.
- Guo, W. et al. (2008) J. Biol. Chem. 283:9136.
- Matsakas, A. et al. (2009) Neuromuscul. Disord. 19:489.
- Guo, T. et al. (2009) PloS ONE 4:e4937.
Long Name
Growth Differentiation Factor 8
Alternate Names
GDF8, MSLHP, MSTN, Myostatin
Gene Symbol
MSTN
UniProt
Additional GDF-8/Myostatin Products
Product Documents for Mouse GDF‑8/Myostatin Propeptide Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Mouse GDF‑8/Myostatin Propeptide Antibody
For research use only
Related Research Areas
Citations for Mouse GDF‑8/Myostatin Propeptide Antibody
Customer Reviews for Mouse GDF‑8/Myostatin Propeptide Antibody (1)
5 out of 5
1 Customer Rating
Have you used Mouse GDF‑8/Myostatin Propeptide Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Showing
1
-
1 of
1 review
Showing All
Filter By:
-
Application: Western BlotSample Tested: See PMID 23829672Species: HumanVerified Customer | Posted 01/05/2015
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Cellular Response to Hypoxia Protocols
- R&D Systems Quality Control Western Blot Protocol
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...