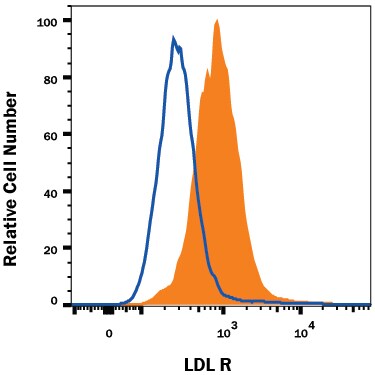
The low density lipoprotein receptor (LDL R) is the founding member of the LDL R family of scavenger receptors (1, 2, 3, 4). This family contains type I transmembrane molecules that are characterized by the presence of EGF repeats, complement-like repeats, and YWTD motifs that form beta -propellers. Although members of the family were originally thought to be endocytic receptors, it is now clear that some members interact with adjacent cell-surface molecules, expanding their range of activities (2, 4). Mouse LDL R is synthesized as a 864 amino acid (aa) precursor that contains a 21 aa signal sequence, a 769 aa extracellular region, a 22 aa transmembrane segment and a 52 aa cytoplasmic tail (5). The extracellular region is complex. It consists of seven N-terminal complement-like cysteine-rich repeats (class A LDL domains) that bind LDL. Cysteines in this region participate in intrachain disulfide bonds. This region is followed by two EGF-like domains and six class B LDL repeats that generate a beta -propeller whose blades each contain a YWTD motif. This area is likely responsible for ligand dissociation (6). Finally, there is a 50 aa membrane proximal Ser/Thr-rich region that shows extensive O-linked glycosylation, generating a native molecular weight for LDL R of 135 kDa (5). Within the 52 aa cytoplasmic region, there is an NPxY motif that links the receptor to clathrin pits and binds to select adaptor proteins (1, 7, 8). The extracellular region of mouse LDL R shares 78% and 87% aa identity with the extracellular region of human and rat LDL R, respectively. LDL R is constitutively expressed and binds apoB of LDL and apoE of VLDL (9). It is responsible for clearing 70% of plasma LDL in liver (9).