Mre11 Antibody - BSA Free
Novus Biologicals | Catalog # NB100-142

![Simple Western: Mre11 Antibody [NB100-142] Simple Western: Mre11 Antibody [NB100-142]](https://resources.rndsystems.com/images/products/Mre11-Antibody-Simple-Western-NB100-142-img0010.jpg)
Key Product Details
Validated by
Species Reactivity
Validated:
Cited:
Predicted:
Applications
Validated:
Cited:
Label
Antibody Source
Format
Product Specifications
Immunogen
Reactivity Notes
Clonality
Host
Isotype
Theoretical MW
Disclaimer note: The observed molecular weight of the protein may vary from the listed predicted molecular weight due to post translational modifications, post translation cleavages, relative charges, and other experimental factors.
Scientific Data Images for Mre11 Antibody - BSA Free
Simple Western: Mre11 Antibody [NB100-142]
Simple Western: Mre11 Antibody [NB100-142] - Image shows a specific band for Mre11 in 0.5 mg/mL of HeLa lysate. A band appears at the theoretical molecular weight of 81 kDa for this product. This experiment was performed under reducing conditions using the 12-230 kDa separation system.Immunocytochemistry/ Immunofluorescence: Mre11 Antibody [NB100-142]
Immunocytochemistry/Immunofluorescence: Mre11 Antibody [NB100-142] - IF analysis of Mre11 on HeLa. Image from verified customer review.Immunohistochemistry: Mre11 Antibody [NB100-142]
Immunohistochemistry: Mre11 Antibody [NB100-142] - Immunohistochemical staining of MRE11 in a human epidermis cross section.Western Blot: Mre11 Antibody [NB100-142]
Western Blot: Mre11 Antibody [NB100-142] - Western blot analysis of Mre11 on 50 ug of HeLa and MEF lysates, displaying bands at the molecular weight of 81 kDa.Immunocytochemistry/ Immunofluorescence: Mre11 Antibody [NB100-142]
Immunocytochemistry/Immunofluorescence: Mre11 Antibody [NB100-142] - HeLa cells were fixed for 10 minutes using 10% formalin and then permeabilized for 5 minutes using 1X TBS + 0.5% Triton X-100. The cells were incubated with anti-Mre11 [NB100-142] at a 1:500 dilution overnight at 4C and detected with an anti-rabbit DyLight 488 (Green) at a 1:500 dilution. Actin was detected with Phalloidin 568 (Red) at a 1:200 dilution. Nuclei were counterstained with DAPI (Blue). Cells were imaged using a 40X objective.Western Blot: Mre11 Antibody [NB100-142] -
Western Blot: Mre11 Antibody [NB100-142] - MRE11 associates with the DS region of OriP.A) DNA affinity purification assays were performed with Raji nuclear extracts & DNA templates derived from DS wt, DS nm-, or delta DS. Input & template bound proteins were analyzed by Western blot with antibodies to TRF2, MRE11, EBNA1, PCNA, & PARP1, as indicated. B) Schematic of template DNA used for DNA affinity purification in A, & their respective protein binding sites. C) ChIP assays to monitor EBNA1, TRF2, & MRE11 binding at OriP wt or OriP nm- in transfected 293 cells. Immunoprecipitated DNA was quantified by real time PCR relative to total input DNA recovered for each transfected cell. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/18040525), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: Mre11 Antibody [NB100-142] -
Western Blot: Mre11 Antibody [NB100-142] - Immunoblotting analysis of IR-induced AKT (Ser473) activation. (A) The levels of AKT activation were measured by AKT Ser473 phosphorylation in 293T, 293T/eIF3f & 293T/eIF3f-hMSH4 cells treated with 10 Gy IR in comparison to untreated controls. Levels of p53 Ser15 phosphorylation & the total protein levels of AKT, hMRE11 & p53 were also analyzed. alpha -tubulin was used as a loading control. (B) The levels of Chk2 activation (Chk2 Thr68 phosphorylation) in 293T, 293T/eIF3f & 293T/eIF3f-hMSH4 cells in response to 10 Gy IR. Cell lysates were prepared at 1 hr post IR treatment. Untreated cells were analyzed as controls, while alpha -tubulin was used as a loading control. Image collected & cropped by CiteAb from the following publication (https://molecular-cancer.biomedcentral.com/articles/10.1186/1476-4598-1…), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: Mre11 Antibody [NB100-142] -
Western Blot: Mre11 Antibody [NB100-142] - The DNA damage response at telomeres uncapped through TRF2 depletion does not require BRCA1 or CtIPImmortalized Brca1F/− MEFs were infected with retroviruses expressing the indicated shRNAs and/or Cre recombinase, followed by selection with puromycin for 72 h. Cell extracts were prepared 48 h later & analysed by Western blotting as indicated. SMC1 & tubulin were used as loading controls. *non-specific band.Cells treated as in (A) were fixed 48 h after selection & stained with an anti-53BP1 antibody (green). Telomeres were visualized with a Cy3-conjugated (CCCTAA)3-PNA probe (red). Yellow arrowheads point to 53BP1 foci that co-localize with telomeres.Quantification of TIFs in cells treated as in (B). A minimum of 200 nuclei were scored for each sample. Error bars represent SD of two independent experiments. P-values were calculated using an unpaired two-tailed t-test. *P ≤ 0.05; NS, P > 0.05. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/25582120), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: Mre11 Antibody [NB100-142] -
Western Blot: Mre11 Antibody [NB100-142] - MRE11 & NBS1 contribute to OriP replication & EBV episome stability.A) D98/HR1 cells were transfected with siRNA for MRE11 or NBS1, or Luciferase control & assayed by immuno-blot (IB) with antibodies specific for MRE11 (left pane) or NBS1 (right panel), or PCNA (lower panel) as a loading control. B) Transient DNA replication assays of OriP-containing plasmids were analyzed by Southern blot. D98/HR1 cells were transfected with OriP plasmid & siRNA for either MRE11, NBS1, or luciferase control (as indicated above each lane). Hirt extracted plasmid DNA was digested with DpnI plus BamHI (top panel) or BamHI only (lower panel) & probed with OriP specific probes. DNA was quantified by PhosphorImager analysis & the replication value was determined as the ratio of DpnI/BamHI to BamHI recovered products. Quantification shown below is a summary of at least four independent replication assays (data not shown). C)) EBV episomes from latently infected D98/HR1 cells were analyzed by pulse field electrophoresis & Southern blotting after transfection of siRNA for control (siLuc), MRE11, or NBS1. Raji cells were used as a control for EBV episome size & abundance. The percentage of EBV episomes relative to the EBV DNA retained in the well is calculated below. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/18040525), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: Mre11 Antibody [NB100-142] -
Western Blot: Mre11 Antibody [NB100-142] - Evidence that MRE11 & NBS1 are required for EBV episome maintenance.A) OriP plasmid maintenance assays were performed in MRE11 mutant (ATLD3) & reconstituted (ATLD3/wtMRE11) cell lines (left panel) or NBS1 mutant (GM7166) & reconstituted (GM7166/wtNBS1) cell lines (right panel). Plasmids containing OriP & EBNA1 were monitored by Southern blotting of Hirt lysates at 1 (lower panel) & 7 (upper panel) days post-transfection. Phosphorimager quantification of four independent experiments as shown in panal A where maintenance is measured as the ratio of day 7 to day 1 for OriP plasmid detection is shown below. B) EBV transformed B-lymphocytes were analyzed by PFG & Southern blotting for the presence of episomal forms of the EBV genome. Raji, Namalwa, NBS1 (GM15808), NBS1 (GM07078), LCL3472, & LCLAW7 DNA was loaded at equal concentrations & analyzed by ethidium staining of PFG (lower panel) or by Southern blot (upper panel) with EBV specific probe. C) Western blot of the same cell lines used for PFG analysis shown in panel B was probed with antibodies specific for NBS1 (antibody 143 & antibody 277), MRE11, or loading control Actin. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/18040525), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: Mre11 Antibody [NB100-142] -
Western Blot: Mre11 Antibody [NB100-142] - Involvement of CBP & p300 in the transcription of the BRCA1 & RAD51 genes.(A) Down-regulation of BRCA1 & RAD51 proteins upon depletion of CBP and/or p300. H1299 cells were transfected for 48 hr with non-targeting (siNT), CBP (siCBP), p300 (sip300), or CBP+p300 (siC+p) siRNAs. The cells were harvested & whole cell extracts were subjected to immunoblotting. (B, C) Reduction of BRCA1 & RAD51 transcripts in CBP- & p300-depleted cells. H1299 cells were transfected for 48 hr with non-targeting (siNT), CBP (siCBP), p300 (sip300), or CBP+p300 (siC+p) siRNAs. Cells were harvested & subjected to quantitative real-time PCR for the detection of BRCA1 (B) & RAD51 (C) mRNAs. Expression levels were normalized against the levels of GAPDH mRNA. Data represent the mean ± SD. (D) H1299 cells were transfected for 48 hr with non-targeting (siNT), CBP (siCBP), p300 (sip300), or CBP+p300 (siC+p) siRNAs, & stained with propidium iodide (PI). The percentage of cells in each cell cycle phase was determined by FACS. Percentages of cells in G1, S, & G2/M are shown. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/23285190), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunocytochemistry/ Immunofluorescence: Mre11 Antibody - BSA Free [NB100-142] -
Normal brain and the cerebellum of Mre11-PC delta mice. (A) Abdominal view of wild type (Ctr) and Mre11-PC delta mice showed normal limbs stretching. (B) Bodyweight of control (Ctr) and Mre11-PC delta mice at 18 months of age. The number of mice analyzed is indicated within the graph bars. Error bars indicate the value mean +/- SEM; n.s., not significant; data comparison was performed through Student’s t-test. (C) A dorsal view of the whole brain of control (Ctr) and Mre11-PC delta mice. (D) Brain weight of 12-month-old Ctr and Mre11-PC delta mice. The number of mice analyzed is indicated within the graph bars. Error bars indicate the value mean +/- SEM; data comparison was performed by Student’s t-test. n.s., not significant. (E) Sagittal cryosections of 18-month-old Ctr and Mre11-PC delta mice after staining with anti-MRE11 antibody and DAPI. Note a reduction of MRE11 signal in mutant Purkinje cells. The Purkinje cell layer in the cerebellum region is shown. Dotted lines highlight the nucleus of Purkinje cells. (F,G)Mre11-PC delta mice have normal density and morphology of Purkinje cells. Sagittal sections of control (Ctr) and Mre11-PC delta mice were stained with Calbindin-D28K (D28K) antibody (green) and DAPI (blue) to label Purkinje cells and nuclei, respectively (F). The upper panels show the whole cerebellum, and the lower panels show the magnified view of red frames in the respective upper panels. ML, molecular layer; PCL, Purkinje cell layer. (G) Quantification of total Purkinje cell number in each individual cerebellar lobule from panel (F). Three mice of each genotype were analyzed. Error bars indicate the value mean +/- SEM; data comparison was performed through Student’s t-test. n.s., not significant. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/35153719), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunocytochemistry/ Immunofluorescence: Mre11 Antibody - BSA Free [NB100-142] -
Deletion of Nbs1 is compatible with mouse survival and cerebellar development. (A) Bodyweight of 20∼24 months old mice from the indicated group. (B) Brain weight of the animal for panel (A), Error bars indicate the value mean +/- SEM; n.s., not significant. Data comparison was performed through Student’s t-test. (C,D) Deletion of Nbs1 in Purkinje cells leads to dislocation of MRE11 and RAD50. Sagittal cryo-sections of control (Ctr) and Nbs1-PC delta mice at the indicated age were stained with anti-MRE11 (C) and anti-RAD50 (D) antibodies together with DAPI to label nuclei. The Purkinje cell layer in the cerebellum region is shown. Scale bars: 20 μm. Representative images of sections from two mice are shown. Arrows point to Purkinje cells. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/35153719), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Applications for Mre11 Antibody - BSA Free
ELISA
Immunoblotting
Immunocytochemistry/ Immunofluorescence
Immunohistochemistry
Immunohistochemistry-Frozen
Immunohistochemistry-Paraffin
Immunoprecipitation
Knockout Validated
Proximity Ligation Assay
Simple Western
Western Blot
See Simple Western Antibody Database for Simple Western validation: Tested in HeLa lysate 0.5 mg/mL, separated by Size, antibody dilution of 1:250, apparent MW was 102 kDa. Separated by Size-Wes, Sally Sue/Peggy Sue.
Reviewed Applications
Read 4 reviews rated 4.5 using NB100-142 in the following applications:
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Formulation
Format
Preservative
Concentration
Shipping
Stability & Storage
Background: Mre11
The MRN assembles as a hetero-hexamer consisting of 2 RAD50 subunits, 2 MRE11 nucleases, and 2 NBS1 subunits, specific to eukaryotes. Mre11 is highly conserved with the N-terminal region of the human protein sharing approximately 50% amino acid sequence identity with the yeast ortholog. Human Mre11 has 3 known isoforms with the predominant isoform having a theoretical molecular weight of 80.6kDa. This nuclear localized protein has high expression in proliferating tissues. Defects in Mre11 can lead to genomic instability, telomere shortening, aberrant meiosis, hypersensitivity to DNA damage, and ataxia telangiectasia-like disorder (ATLD). MRN expression has been associated with the prognosis of cancer and MRN mutants have been linked to cancer susceptibility in ovarian cancer and glioma (4, 5).
References
1. Paull TT. (2018) 20 Years of Mre11 Biology: No End in Sight. Mol Cell. 71(3):419-427. PMID: 30057197
2. Syed A, Tainer JA. (2018) The MRE11-RAD50-NBS1 Complex Conducts the Orchestration of Damage Signaling and Outcomes to Stress in DNA Replication and Repair. Annu Rev Biochem. 87:263-294. PMID: 29709199
3. Wang Y, Cortez D, Yazdi P, Neff N, Elledge SJ, Qin J. (2000) BASC, a super complex of BRCA1-associated proteins involved in the recognition and repair of aberrant DNA structures. Genes Dev. 14(8):927-39. PMID: 10783165
4. Kim JH, Grosbart M, Anand R, Wyman C, Cejka P, Petrini JHJ. (2017) The Mre11-Nbs1 Interface Is Essential for Viability and Tumor Suppression. Cell Rep. 18(2):496-507. PMID: 28076792
5. Bian L, Meng Y, Zhang M, Li D. (2019) MRE11-RAD50-NBS1 complex alterations and DNA damage response: implications for cancer treatment. Mol Cancer. 18(1):169. PMID: 31767017
Long Name
Alternate Names
Gene Symbol
UniProt
Additional Mre11 Products
Product Documents for Mre11 Antibody - BSA Free
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Product Specific Notices for Mre11 Antibody - BSA Free
This product is for research use only and is not approved for use in humans or in clinical diagnosis. Primary Antibodies are guaranteed for 1 year from date of receipt.
Citations for Mre11 Antibody - BSA Free
Customer Reviews for Mre11 Antibody - BSA Free (4)
Have you used Mre11 Antibody - BSA Free?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
-
Application: ImmunofluorescenceSample Tested: testesSpecies: MouseVerified Customer | Posted 12/12/2014
-
Application: ImmunofluorescenceSample Tested: MammalianSpecies: OtherVerified Customer | Posted 10/08/2014
-
Application: ImmunofluorescenceSample Tested: HeLa cellsSpecies: HumanVerified Customer | Posted 07/31/2014IF
-
Application: Western BlotSample Tested: TIG-1 human primary fibroblasts, whole cell lysate (30 ug)Species: HumanVerified Customer | Posted 04/10/2014Mre11 antibody (NB100-142)
There are no reviews that match your criteria.
Protocols
View specific protocols for Mre11 Antibody - BSA Free (NB100-142):
A 5beta in situ extraction method [10mM Pipes, pH 6.8 / 0.2% Triton X-100 / 100mM MgCl2 / 100mM sucrose/ 10mM EGTA Beta on ice] followed by 4% paraformaldehyde fixation of tissues works well for immunofluorescence of anti-hMre11 (NB 100-142).
Please see reference: Franchitto, A., Pichierri, P., Blooms syndrome protein is required for correct relocalization of RAD50/Mre11/nbs1 complex after replication fork arrest. J. of Cell Biology, DOI: 10 (2002)
Tissue Preparation: Formalin fixation and embedding in paraffin wax
The tissue needs to be fixed overnight in formalin at room temperature (approx. 16 hours).
There needs to be at least a 10:1 ratio of formalin volume to volume of the tissue.
The tissue can be no more than 4-5 mm thick. If it is thicker than this, it will need to be bivalved (bisected so as to make two similar separate parts) before being added to the formalin due to the diffusion coefficient of formalin.
The working area of the tissue cassettes is roughly 2.5 cm by 3.0 cm. So the cut tissue sample can be no larger than this.
Formalin refers to 10% neutral buffered formalin.
After formalin fixation overnight tissues can be processed according to the following protocol:
Tissue Sectioning: Make 4-um sections and place on pre-cleaned and charged microscope slides.
Heat in a tissue-dryingoven for 45 minutes at 60 degrees C.
Deparaffinization: Wash dry slides in 3 changes of xylene for 5 minutes each @ RT
Rehydration: Wash slides in 3 changes of 100% alcohol for 3 minutes each @ RT
Wash slides in 2 changes of 95% alcohol for 3 minutes each @ RT
Wash slides in 1 change of 80% alcohol for 3 minutes @ RT
Rinse slides in gentle running distilled water for 5 minutes @ RT
Antigen retrieval: Steam slides in 0.01 M sodium citrate buffer, pH 6.0 at 99-100 degrees C - 20 minutes
Remove from heat and let stand at room temperature in buffer - 20 minutes
Rinse in 1X TBS with Tween (TBST) at 1 minute @ RT
Immunostaining: (Do not allow tissues to dry at any time during the staining procedure)
Apply a universal protein block for 20 minutes @ RT
Drain protein block from slides, apply diluted primary antibody for 45 minutes @ RT
Rinse slides in 1X TBST - 1 minute @ RT
Apply a biotinylated anti-rabbit IgG (H+L) secondary for 30 minutes @ RT
Rinse slides in 1X TBST - 1 minute @ RT
Apply alkaline phosphatase streptavidin for 30 minutes @ RT
Rinse slides in 1X TBST - 1 minute @ RT
Apply alkaline phosphatase chromogen substrate for 30 minutes @ RT
Wash slides in distilled water for 1 minute @ RT
Dehydrate: (This method should only be used if the chromogen substrate is alcohol insoluble (e.g. Vector Red, DAB)
Wash slides in 2 changes of 80% alcohol for 1 minute each @ RT
Wash slides in 2 changes of 95% alcohol for 1 minute each @ RT
Wash slides in 3 changes of 100% alcohol for 1 minute each @ RT
Wash slides in 3 changes of xylene for 1 minute each @ RT
Apply coverslip
1. For IP reactions, start with extract (whole cell or nuclear) from around 3 million cells prepared in 0.5-1 ml lysis buffer (100 mM NaCl, 10 mM Tris HCl, 5 mM EDTA, 0.5% nonidet p40).
2. Cells are resuspended in lysis buffer, then incubated with rotation about 15 min at 4 degrees C.
3. The lysate is then centrifuged 5 min at 14000g to remove insoluble material.
4. To cleared lysate, add 1-3 ul of antiserum and incubate on ice for 30 min.
5. Collect immune complexes on Protein A Sepharose by adding 25 ul of a 50% slurry, and incubate with rotation for 1 hour at 4 degrees C.
6. The complexes are pelleted gently (5000g for 5-10 sec.) then washed with 1 ml lysis buffer.
7. Repeat the wash 2 more times.
8. Analyze the immunoprecipitates by SDS PAGE. This antibody works well for IP reactions from both human and mouse cells. The intact complex is stable and can be immunoprecipitated in many common lysis buffers (up to 0.5 M NaCl).
Western Blot Procedure
1. Run 50 ug of protein on a 4-20% Tris-glycine mini-gel at 125V for 90 minutes.
2. Equilibrate gel, nitrocellulose membrane, Whatman paper, and blotting pads in transfer buffer for 15 minutes.
3. Transfer protein to the membrane at 25V for 90 minutes.
4. Allow membrane to air-dry.
5. Block membrane with 1XPBS/3% BSA for 1 hour at room temperature (23-27 degrees C).
6. Wash membrane twice, for 5 minutes each, with 1XPBS/0.05% Tween-20 (PBST).
7. Incubate membrane with 1:5000 dilution of NB100-142 (anti-hMre11), diluted in 1XPBS/1% BSA, for 1 hour at room temperature.
8. Wash membrane once for 15 minutes, then four times for 5 minutes each, with PBST.
9. Incubate membrane with goat anti-rabbit IgG-HRP, diluted in 1XPBS/1% BSA, for 1 hour at room temperature.
10. Wash membrane once for 15 minutes, then four times for 5 minutes each, with PBST.
11. Detect cross-reacting proteins using Renaissance Chemiluminescence Reagent Plus kit from NEN Life Sciences.
NOTE: HeLa whole cell extracts (NB800-PC1) were used as a positive control for this antibody.
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- ChIP Protocol Video
- Chromatin Immunoprecipitation (ChIP) Protocol
- Chromatin Immunoprecipitation Protocol
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- ELISA Sample Preparation & Collection Guide
- ELISA Troubleshooting Guide
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- How to Run an R&D Systems DuoSet ELISA
- How to Run an R&D Systems Quantikine ELISA
- How to Run an R&D Systems Quantikine™ QuicKit™ ELISA
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Immunoprecipitation Protocol
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Liperfluo
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- Quantikine HS ELISA Kit Assay Principle, Alkaline Phosphatase
- Quantikine HS ELISA Kit Principle, Streptavidin-HRP Polymer
- R&D Systems Quality Control Western Blot Protocol
- Sandwich ELISA (Colorimetric) – Biotin/Streptavidin Detection Protocol
- Sandwich ELISA (Colorimetric) – Direct Detection Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: ELISA
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
FAQs for Mre11 Antibody - BSA Free
-
Q: Could you recommend an optimal Mre11 positive control for Western blot among your products?
A: NBL1-13219 would be the best product since it is intended for this purpose.
-
Q: I would like to know if these antibodies are suitable with IHC techniques using HRP conjugated antibodies and DAB as chromogen? Cat# NB200-175, NB100-904 and NB100-142
A: Yes, all of these antibodies are useful for IHC-P with DAB based detection. Please note that the PTEN Antibody (28H6) NB200-175 has been discontinued, however.
-
Q: Could you recommend an optimal Mre11 positive control for Western blot among your products?
A: NBL1-13219 would be the best product since it is intended for this purpose.
-
Q: I would like to know if these antibodies are suitable with IHC techniques using HRP conjugated antibodies and DAB as chromogen? Cat# NB200-175, NB100-904 and NB100-142
A: Yes, all of these antibodies are useful for IHC-P with DAB based detection. Please note that the PTEN Antibody (28H6) NB200-175 has been discontinued, however.

![Immunocytochemistry/ Immunofluorescence: Mre11 Antibody [NB100-142] Immunocytochemistry/ Immunofluorescence: Mre11 Antibody [NB100-142]](https://resources.rndsystems.com/images/products/Mre11-Antibody-Immunocytochemistry-Immunofluorescence-NB100-142-img0009.jpg)
![Western Blot: Mre11 Antibody [NB100-142] Knockdown Validated: Mre11 Antibody [NB100-142]](https://resources.rndsystems.com/images/products/Mre11-Antibody-Knockdown-Validated-NB100-142-img0012.jpg)
![Immunohistochemistry: Mre11 Antibody [NB100-142] Immunohistochemistry: Mre11 Antibody [NB100-142]](https://resources.rndsystems.com/images/products/Mre11-Antibody-Immunohistochemistry-NB100-142-img0004.jpg)
![Western Blot: Mre11 Antibody [NB100-142] Western Blot: Mre11 Antibody [NB100-142]](https://resources.rndsystems.com/images/products/Mre11-Antibody-Western-Blot-NB100-142-img0005.jpg)
![Immunocytochemistry/ Immunofluorescence: Mre11 Antibody [NB100-142] Immunocytochemistry/ Immunofluorescence: Mre11 Antibody [NB100-142]](https://resources.rndsystems.com/images/products/Mre11-Antibody-Immunocytochemistry-Immunofluorescence-NB100-142-img0011.jpg)
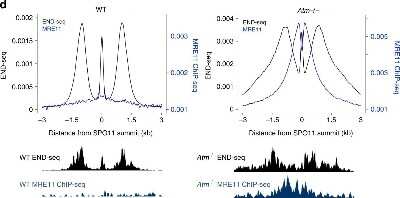
![Western Blot: Mre11 Antibody [NB100-142] Knockdown Validated: Mre11 Antibody [NB100-142]](https://resources.rndsystems.com/images/products/Mre11-Antibody-Knockdown-Validated-NB100-142-img0008.jpg)
![Western Blot: Mre11 Antibody [NB100-142] - Mre11 Antibody](https://resources.rndsystems.com/images/products/nb100-142_rabbit-polyclonal-mre11-antibody-310202415384114.jpg)
![Western Blot: Mre11 Antibody [NB100-142] - Mre11 Antibody](https://resources.rndsystems.com/images/products/nb100-142_rabbit-polyclonal-mre11-antibody-310202415293334.jpg)
![Western Blot: Mre11 Antibody [NB100-142] - Mre11 Antibody](https://resources.rndsystems.com/images/products/nb100-142_rabbit-polyclonal-mre11-antibody-31020241554548.jpg)
![Western Blot: Mre11 Antibody [NB100-142] - Mre11 Antibody](https://resources.rndsystems.com/images/products/nb100-142_rabbit-polyclonal-mre11-antibody-310202416574.jpg)
![Western Blot: Mre11 Antibody [NB100-142] - Mre11 Antibody](https://resources.rndsystems.com/images/products/nb100-142_rabbit-polyclonal-mre11-antibody-31020241612675.jpg)
![Western Blot: Mre11 Antibody [NB100-142] - Mre11 Antibody](https://resources.rndsystems.com/images/products/nb100-142_rabbit-polyclonal-mre11-antibody-310202415525210.jpg)
![Immunocytochemistry/ Immunofluorescence: Mre11 Antibody - BSA Free [NB100-142] - Mre11 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb100-142_rabbit-polyclonal-mre11-antibody-immunocytochemistry-immunofluorescence-132202619442311.jpg)
![Immunocytochemistry/ Immunofluorescence: Mre11 Antibody - BSA Free [NB100-142] - Mre11 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb100-142_rabbit-polyclonal-mre11-antibody-immunocytochemistry-immunofluorescence-132202619434118.jpg)

-(005-ml)_NB100-142_9111.jpg)
-(005-ml)_NB100-142_6806.jpg)