NCOA2 Antibody - BSA Free
Novus Biologicals | Catalog # NB100-388

![Western Blot: NCOA2 Antibody [NB100-388] Western Blot: NCOA2 Antibody [NB100-388]](https://resources.rndsystems.com/images/products/NCOA2-Antibody-Western-Blot-NB100-388-img0002.jpg)
Key Product Details
Validated by
Independent Antibodies
Species Reactivity
Validated:
Human, Mouse, Rat
Cited:
Rat
Applications
Validated:
Western Blot, Immunocytochemistry/ Immunofluorescence, Immunoprecipitation
Cited:
Immunocytochemistry/ Immunofluorescence
Label
Unconjugated
Antibody Source
Polyclonal Rabbit IgG
Format
BSA Free
Loading...
Product Specifications
Immunogen
The immunogen recognized by this antibody maps to a region between residues 700 and 750 of human Nuclear Receptor Coactivator 2 using the numbering given in entry NP_006531.1 (GeneID 10499).
Reactivity Notes
Rat reactivity reported in scientific literature (PMID: 28552353).
Specificity
Antibody was affinity purified using an epitope specific to SRC2 immobilized on solid support.
Clonality
Polyclonal
Host
Rabbit
Isotype
IgG
Scientific Data Images for NCOA2 Antibody - BSA Free
Western Blot: NCOA2 Antibody [NB100-388]
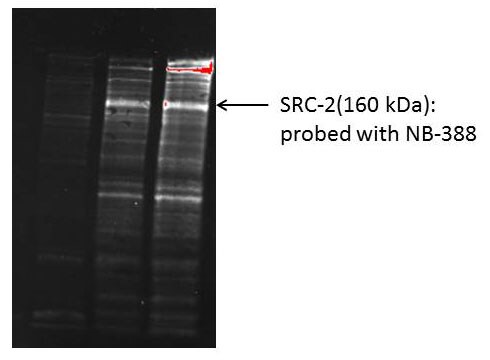
Western Blot: NCOA2 Antibody [NB100-388] - Detection of Mouse NCOA2/ SRC2 by Western Blot. Samples: Whole cell lysate (50 ug) from TCMK-1, 4T1, and CT26.WT cells. Antibodies: Affinity purified rabbit anti-NCOA2/SRC2 antibody NB100-388 used for WB at 1 ug/ml. Detection: Chemiluminescence with an exposure time of 3 minutes.Immunoprecipitation: NCOA2 Antibody [NB100-388]
Immunoprecipitation: NCOA2 Antibody [NB100-388] - Detection of human NCOA2/ SRC2 by western blot and immunoprecipitation. Samples: Whole cell lysate from HeLa(15 and 50 ug for WB; 1 mg for IP, 20% of IP loaded), HEK293T (T; 50 ug) and Jurkat (J; 50 ug) cells. Antibodies: Affinity purified rabbit anti-NCOA2/SRC2 antibody NB100-388 (lot 2) used for WB at 0.1 ug/ml (A) and 1 ug/ml (B) and used for IP at 6 ug/mg lysate. SRC2 was also immunoprecipitated by a previous lot (lot 1) and by rabbit anti-NCOA1/SRC2 antibody NBP2-22162, which recognizes a downstream epitope. Detection: Chemiluminescence with exposure times of 3 minutes (A) and 10 seconds (B).Applications for NCOA2 Antibody - BSA Free
Application
Recommended Usage
Immunoprecipitation
1-4 ug/mg lysate
Western Blot
1:1000-1:10000
Application Notes
NB 100-388 may be used for Western Blot, where a band at ~160 kDa is seen, representing SRC2 protein. Though this antibody will recognize cytosolic protein in Western blot, it does much better with nuclear extracts. It may also be used for immunoprecipitation on HeLa nuclear extracts. Suggested working dilutions: * Western Blot - 1:1,000-1:10,000 Immunocytochemistry - Not Determined Immunoprecipitation - 1-4 ug/mg *The investigator should determine the optimal working dilution for a specific application. Use in Immunocytochemistry/immunofluorescence reported in scientific literature (PMID: 28552353).
Reviewed Applications
Read 1 review rated 4 using NB100-388 in the following applications:
Formulation, Preparation, and Storage
Purification
Immunogen affinity purified
Formulation
Tris-Citrate/Phosphate (pH 7.0 - 8.0)
Format
BSA Free
Preservative
0.09% Sodium Azide
Concentration
1.0 mg/ml
Shipping
The product is shipped with polar packs. Upon receipt, store it immediately at the temperature recommended below.
Stability & Storage
Store at 4C. Do not freeze.
Background: NCOA2
Long Name
Nuclear Receptor Co-activator 2
Alternate Names
bHLHe75, HTIF2, KAT13C, TIF2
Entrez Gene IDs
10499 (Human)
Gene Symbol
NCOA2
UniProt
Additional NCOA2 Products
Product Documents for NCOA2 Antibody - BSA Free
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Product Specific Notices for NCOA2 Antibody - BSA Free
This product is for research use only and is not approved for use in humans or in clinical diagnosis. Primary Antibodies are guaranteed for 1 year from date of receipt.
Related Research Areas
Citations for NCOA2 Antibody - BSA Free
Customer Reviews for NCOA2 Antibody - BSA Free (1)
4 out of 5
1 Customer Rating
Have you used NCOA2 Antibody - BSA Free?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
Showing
1
-
1 of
1 review
Showing All
Filter By:
-
Application: Western BlotSample Tested: Brain (hypothalamus) tissueSpecies: MouseVerified Customer | Posted 10/10/2016western blot was done on proteins from adult female mouse hypothalamus to probe for SRC-2 fraction that was associated with Progestin receptors.
There are no reviews that match your criteria.
Protocols
View specific protocols for NCOA2 Antibody - BSA Free (NB100-388):
Nuclear Extract and Cytoplasmic Fraction Preparation protocol for NCOA2 Antibody (NB100-388):
Nuclear Extract and Cytoplasmic Fraction Preparation
1. Nuclear extracts (NE) and cytoplasmic fractions (S100) were prepared by Dignam's method (Dignam, Lebovitz, and Roeder, Nucleic Acids Res. 11: 1475-1489. 1983).
2. 100 liters of HeLa cell culture were harvested and washed 3 times with cold PBS.
3. The packed-cell volume (PCV) was measured, and the cell pellet was gently resuspended with 5 PCVs of hypotonic buffer (10 mM HEPES-KOH [pH 8], 10 mM KCl, 1.5 mM MgCl2, 1 mM DTT, 0.2 mM PMSF).
4. Cells were incubated on ice for 10 minutes and then pelleted by centrifugation at 1,800xg for 10 minutes.
5. Hypotonic buffer was added to 2 PCVs, and cells were resuspended and then homogenized with 15 strokes using a pestle B in a Dounce glass homogenizer until the cells were more than 90% lysed, as determined by a light microscope.
6. The lysate was centrifuged at 20,000xg for 30 minutes at 4 degrees Celcius.
7. The supernatant was saved for S100 fraction, and the pellet was saved to measure the packed nuclear volume (PNV).
8. 0.4 ml of extraction buffer (20 mM HEPES-KOH [pH 8], 0.6 M KCl, 1.5 mM MgCl2, 0.2 mM EDTA, 25% [vol/vol] glycerol, 1 mM DTT, 0.2 mM PMSF) per ml of PNV was added.
9. Cell nuclei were homogenized with 10 strokes of pestle A in the homogenizer.
10. Suspension was stirred at 4 degrees Celcius for 30 minutes and centrifuged for 30 minutes at 20,000xg.
11. The supernatant (nuclear extract) was aliquotted for use.
12. The S100 fraction (resulting supernatant) was mixed with 0.11 volume of high-salt buffer (20 mM HEPES-KOH [pH 8], 1.2 M KCl, 1.5 mM MgCl2, 0.2 mM EDTA, 20% [vol/vol] glycerol, 1 mM DTT, 0.2 mM PMSF) and centrifuged at 100,000xg for 60 minutes at 4 degrees Celcius.
13. This supernatant was dialyzed for 2 hours at 4 degrees Celcius.
14. The sample was centrifuged for 30 minutes at 20,000xg and the supernatant (S100) was aliquotted for use.
Immunoprecipitation
Antibody characterization:
1. HeLa NE and S100 were diluted with 1 volume of RIPA buffer [150 mM NaCl, 1% NP-40, 0.5% DOC, 0.1% SDS, 50 mM Tris [pH 8]).
2. Cleared by spinning at 100,000 g for 20 minutes at 4 degrees Celcius.
3. 1 ml of supernatant (~10 mg total protein) was mixed with 20 ug of primary antibody (NB 100-388) and rotated overnight at 4 degrees Celcius.
4. Supernatant was mixed with 0.05 ml of protein A-sepharose beads (50% slurry) and rotated for 2 hours at 4 degrees Celcius.
5. Immunoprecipitates were washed 3 times with the 10% RIPA in PBS.
6. The washed beads were boiled with 0.04 ml of Laemmli buffer and subjected to SDS-PAGE (4-20% Tris-glycine gel).
Complex purification:
1. NE and S100 were cleared by spinning at 20,000 g for 30 minutes at 4 degrees Celcius.
2. 1.5 ml of supernatant (~15 mg total protein) was mixed with 20 ug of primary antibody (NB 100-388) and rotated for 4 hours at 4 degrees Celcius.
3. Sample and antibody mixture were centrifuged at 15,000 g for 20 minutes at 4 degrees Celcius.
4. Supernatant was mixed with 0.05 ml of protein A-sepharose beads (50% slurry) and rotated for 1 hour at 4 degrees Celcius.
5. Immunoprecipitates were washed 3 times with the NETN buffer (20 mM Tris-HCl [pH 8], 100 mM NaCl, 1 mM EDTA, 0.5% NP-40).
6. The washed beads were boiled with 0.04 ml of Laemmli buffer and subjected to SDS-PAGE (4-20% Tris-glycine gel).
*If an insufficient amount of protein is purified for identification from 15 mg of extract, carry out the same procedure using 50-100 mg of extract to increase the amount of purified protein yield.
Nuclear Extract and Cytoplasmic Fraction Preparation
1. Nuclear extracts (NE) and cytoplasmic fractions (S100) were prepared by Dignam's method (Dignam, Lebovitz, and Roeder, Nucleic Acids Res. 11: 1475-1489. 1983).
2. 100 liters of HeLa cell culture were harvested and washed 3 times with cold PBS.
3. The packed-cell volume (PCV) was measured, and the cell pellet was gently resuspended with 5 PCVs of hypotonic buffer (10 mM HEPES-KOH [pH 8], 10 mM KCl, 1.5 mM MgCl2, 1 mM DTT, 0.2 mM PMSF).
4. Cells were incubated on ice for 10 minutes and then pelleted by centrifugation at 1,800xg for 10 minutes.
5. Hypotonic buffer was added to 2 PCVs, and cells were resuspended and then homogenized with 15 strokes using a pestle B in a Dounce glass homogenizer until the cells were more than 90% lysed, as determined by a light microscope.
6. The lysate was centrifuged at 20,000xg for 30 minutes at 4 degrees Celcius.
7. The supernatant was saved for S100 fraction, and the pellet was saved to measure the packed nuclear volume (PNV).
8. 0.4 ml of extraction buffer (20 mM HEPES-KOH [pH 8], 0.6 M KCl, 1.5 mM MgCl2, 0.2 mM EDTA, 25% [vol/vol] glycerol, 1 mM DTT, 0.2 mM PMSF) per ml of PNV was added.
9. Cell nuclei were homogenized with 10 strokes of pestle A in the homogenizer.
10. Suspension was stirred at 4 degrees Celcius for 30 minutes and centrifuged for 30 minutes at 20,000xg.
11. The supernatant (nuclear extract) was aliquotted for use.
12. The S100 fraction (resulting supernatant) was mixed with 0.11 volume of high-salt buffer (20 mM HEPES-KOH [pH 8], 1.2 M KCl, 1.5 mM MgCl2, 0.2 mM EDTA, 20% [vol/vol] glycerol, 1 mM DTT, 0.2 mM PMSF) and centrifuged at 100,000xg for 60 minutes at 4 degrees Celcius.
13. This supernatant was dialyzed for 2 hours at 4 degrees Celcius.
14. The sample was centrifuged for 30 minutes at 20,000xg and the supernatant (S100) was aliquotted for use.
Immunoprecipitation
Antibody characterization:
1. HeLa NE and S100 were diluted with 1 volume of RIPA buffer [150 mM NaCl, 1% NP-40, 0.5% DOC, 0.1% SDS, 50 mM Tris [pH 8]).
2. Cleared by spinning at 100,000 g for 20 minutes at 4 degrees Celcius.
3. 1 ml of supernatant (~10 mg total protein) was mixed with 20 ug of primary antibody (NB 100-388) and rotated overnight at 4 degrees Celcius.
4. Supernatant was mixed with 0.05 ml of protein A-sepharose beads (50% slurry) and rotated for 2 hours at 4 degrees Celcius.
5. Immunoprecipitates were washed 3 times with the 10% RIPA in PBS.
6. The washed beads were boiled with 0.04 ml of Laemmli buffer and subjected to SDS-PAGE (4-20% Tris-glycine gel).
Complex purification:
1. NE and S100 were cleared by spinning at 20,000 g for 30 minutes at 4 degrees Celcius.
2. 1.5 ml of supernatant (~15 mg total protein) was mixed with 20 ug of primary antibody (NB 100-388) and rotated for 4 hours at 4 degrees Celcius.
3. Sample and antibody mixture were centrifuged at 15,000 g for 20 minutes at 4 degrees Celcius.
4. Supernatant was mixed with 0.05 ml of protein A-sepharose beads (50% slurry) and rotated for 1 hour at 4 degrees Celcius.
5. Immunoprecipitates were washed 3 times with the NETN buffer (20 mM Tris-HCl [pH 8], 100 mM NaCl, 1 mM EDTA, 0.5% NP-40).
6. The washed beads were boiled with 0.04 ml of Laemmli buffer and subjected to SDS-PAGE (4-20% Tris-glycine gel).
*If an insufficient amount of protein is purified for identification from 15 mg of extract, carry out the same procedure using 50-100 mg of extract to increase the amount of purified protein yield.
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunoprecipitation Protocol
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...

![Immunoprecipitation: NCOA2 Antibody [NB100-388] Immunoprecipitation: NCOA2 Antibody [NB100-388]](https://resources.rndsystems.com/images/products/NCOA2-Antibody-Immunoprecipitation-NB100-388-img0003.jpg)