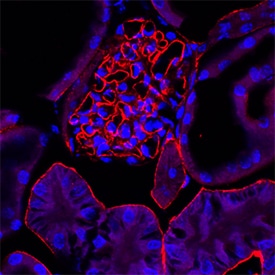
Agrin, a heparan sulfate proteoglycan, is a component of the synaptic basal lamina which promotes acetylcholine receptor (AChR) clustering on cultured myotubes and in vivo. This AChR clustering activity has been shown to be mediated via a receptor complex that includes a receptor-like tyrosine kinase specific to the skeletal muscle termed muscle-specific kinase (MuSK), and an as of yet unidentified myotube-specific accessory component.
Agrin contains a number of distinct domains. The N-terminal half of the molecule, which is responsible for the tight interaction with the extracellular matrix, has nine follistatin-like repeats that share homology to Kazal-type protease inhibitor domains. The C-terminal half, which by itself is sufficient for the AChR clustering activity, has four EGF-like repeats and three laminin globular G domains. Agrin exists in several isoforms which are generated by alternative splicing at multiple splicing sites in the C-terminal half. Some of these isoforms are expressed specifically in the nervous system while other isoforms are expressed in both neural and nonneural tissues. Dramatic differences in AChR clustering activities have been observed between the different isoforms. The highest activity is found to be associated with isoforms found exclusively in neural tissues that contain the four amino acids K-S-R-K at position # 1643‑1646 and the eight amino acids E-L-T-N-E-I-P-A after Ser1779. Rat and chick Agrin share approximately 60% amino acid sequence identity. R&D Systems recombinant C-terminal half fragment of Agrin is a soluble secreted protein which has been found to have AChR clustering activity on chick myotubes.