FGF-8 is a member of the fibroblast growth factor family that was originally discovered as a growth factor essential for the androgen-dependent growth of mouse mammary carcinoma cells (1-4). Alternate splicing of mouse FGF-8 mRNA generates eight secreted isoforms, designated a-h. Only FGF-8a, b, e and f exist in humans (4). FGF-8 contains a 22 amino acid (aa) signal sequence, an N-terminal domain that varies according to the isoform (20 aa for FGF-8a, which is the shortest), a 125 aa FGF domain and a 37 aa proline-rich C-terminal sequence. The FGF domain of FGF-8 shares the most aa identity with FGF17 (75%) and FGF-18 (67%), and the three form an FGF subfamily (2). Human FGF-8a shares 100% aa identity with mouse, rat and bovine FGF-8a, and 99%, 83%, 83% and 78% aa identity with canine, Xenopus, chicken and zebrafish FGF-8a, respectively. FGF-8 is widely expressed during embryogenesis, and mediates epithelial-mesenchymal transitions. It plays an organizing and inducing role during gastrulation, and regulates patterning of the midbrain/hindbrain, eye, ear, limbs and heart in the embryo (2, 5-8). The isoforms may play different roles in development. For example, FGF-8a expands the midbrain in transgenic mice, while FGF-8b transforms midbrain into cerebellum (5). FGF-8 activates the ‘c’ splice forms of fibroblast growth factor receptors FGF R2, FGF R3, and FGF R4, with differential activity among the FGF-8 isoforms (2, 9). FGF-8b shows the strongest receptor affinity and oncogenic transforming capacity, although FGF-8a and e are also transforming and have been found in human prostate, breast or ovarian tumors (1, 5, 10 - 13). FGF-8 shows limited expression in the normal adult, but low levels are found in the reproductive and genitourinary tract, peripheral leukocytes and bone marrow hematopoietic cells (3, 10, 14).
Best Seller
Recombinant Human FGF-8a Protein, CF
R&D Systems | Catalog # 4745-F8

Loading...
Key Product Details
- R&D Systems E. coli-derived Recombinant Human FGF-8a Protein (4745-F8)
- Quality control testing to verify active proteins with lot specific assays by in-house scientists
- All R&D Systems proteins are covered with a 100% guarantee
Source
E. coli
Accession Number
Applications
Bioactivity
Loading...
Product Specifications
Source
E. coli-derived human FGF-8 protein
Gln23-Arg204, with an N-terminal Met
Gln23-Arg204, with an N-terminal Met
Purity
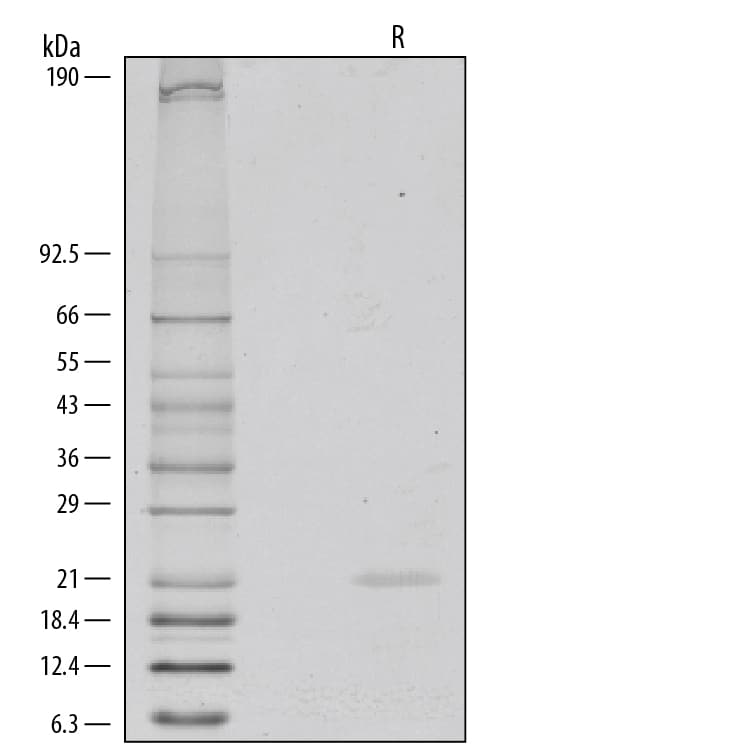
>95%, by SDS-PAGE visualized with Silver Staining and quantitative densitometry by Coomassie® Blue Staining.
Endotoxin Level
<0.01 EU per 1 μg of the protein by the LAL method.
N-terminal Sequence Analysis
Met
Predicted Molecular Mass
21 kDa
Activity
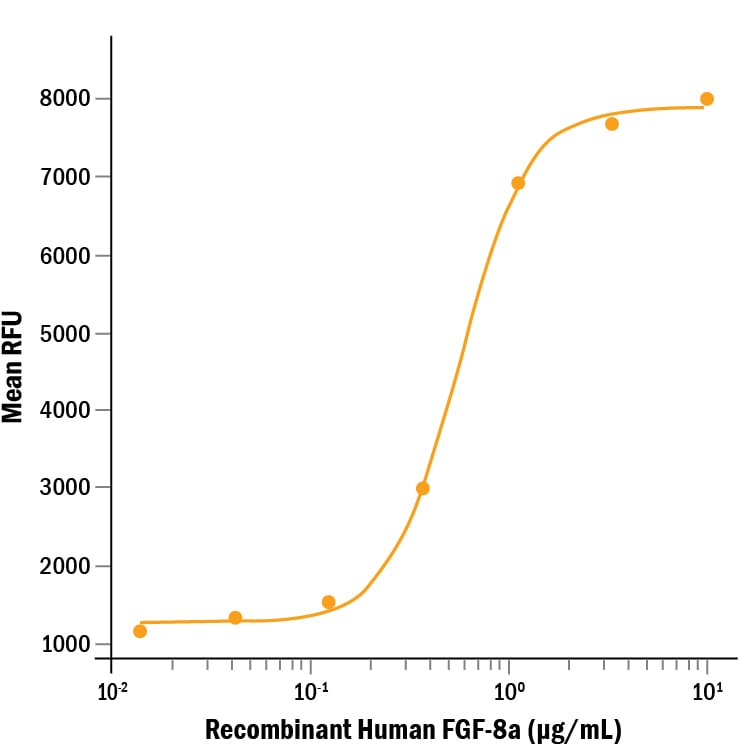
Measured in a cell proliferation assay using NR6R‑3T3 mouse fibroblast cells. Raines, E.W. et al. (1985) Methods Enzymol. 109:749.
The ED50 for this effect is 0.25-1.5 μg/mL in the presence of 10 µg/mL heparin.
The ED50 for this effect is 0.25-1.5 μg/mL in the presence of 10 µg/mL heparin.
Scientific Data Images for Recombinant Human FGF-8a Protein, CF
Recombinant Human FGF-8a Protein Bioactivity
Recombinant Human FGF-8a (Catalog # 4745-F8) stimulates cell proliferation of the NR6R‑3T3 mouse fibroblast cell line. The ED50 for this effect is 0.25-1.5 μg/mL in the presence of 10 μg/mL heparin.Recombinant Human FGF-8a Protein SDS-PAGE
1 μg/lane of Recombinant Human FGF-8a was resolved with SDS-PAGE under reducing (R) conditions and visualized by silver staining, showing a single band at 22 kDa.Formulation, Preparation, and Storage
4745-F8
| Formulation | Lyophilized from a 0.2 μm filtered solution in MOPS, Na2SO4, and EDTA. |
| Reconstitution | Reconstitute at 500 μg/mL in sterile PBS.
Loading...
|
| Shipping | The product is shipped with polar packs. Upon receipt, store it immediately at the temperature recommended below. |
| Stability & Storage |
|
Calculators
Background: FGF-8
References
- Mattila, M.M. and P.L. Harkonen (2007) Cytokine Growth Factor Rev. 18:257.
- Reuss, B. and O. von Bohlen und Halbach (2003) Cell Tissue Res. 313:139.
- Payson, R.A. et al. (1996) Oncogene 13:47.
- Gemel, J. et al. (1996) Genomics 35:253.
- Olsen, S.K. et al. (2006) Genes Dev. 20:185.
- Crossley, P.H. et al. (1996) Cell, 84:127.
- Heikinheimo, M. et al. (1994) Mech. Dev. 48:129.
- Sun, X. et al. (1999) Genes Dev. 13:1834.
- Blunt, A.G. et al. (1997) J. Biol. Chem. 272:3733.
- Ghosh, A.K. et al. (1996) Cell Growth Differ. 7:1425.
- Mattila, M.M. et al. (2001) Oncogene 20:2791.
- Valve, E. et al. (2000) Int. J. Cancer 88:718.
- Valve, E.M. et al. (2001) Lab. Invest. 81:815.
- Nezu, M. et al. (2005) Biochem. Biophys. Res. Commun. 335:843.
Long Name
Fibroblast Growth Factor 8
Alternate Names
AIGF, FGF8, HBGF-8
Gene Symbol
FGF8
UniProt
Additional FGF-8 Products
Product Documents for Recombinant Human FGF-8a Protein, CF
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Recombinant Human FGF-8a Protein, CF
For research use only
Related Research Areas
Citations for Recombinant Human FGF-8a Protein, CF
Customer Reviews for Recombinant Human FGF-8a Protein, CF
There are currently no reviews for this product. Be the first to review Recombinant Human FGF-8a Protein, CF and earn rewards!
Have you used Recombinant Human FGF-8a Protein, CF?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review