Recombinant Human GDF-15 (E. coli-expressed) Protein, CF
R&D Systems | Catalog # 9279-GD

Loading...
Key Product Details
- R&D Systems E. coli-derived Recombinant Human GDF-15 (E. coli-expressed) Protein (9279-GD)
- Quality control testing to verify active proteins with lot specific assays by in-house scientists
- All R&D Systems proteins are covered with a 100% guarantee
Source
E. coli
Accession Number
Structure / Form
Disulfide-linked homodimer
Applications
Bioactivity
Loading...
Product Specifications
Source
E. coli-derived human GDF-15 protein
Ala197-Ile308
Ala197-Ile308
Purity
>95%, by SDS-PAGE visualized with Silver Staining and quantitative densitometry by Coomassie® Blue Staining.
Endotoxin Level
<0.10 EU per 1 μg of the protein by the LAL method.
N-terminal Sequence Analysis
Ala197
Predicted Molecular Mass
12 kDa
SDS-PAGE
12 kDa, reducing conditions
Activity
Measured by its binding ability in a functional ELISA.
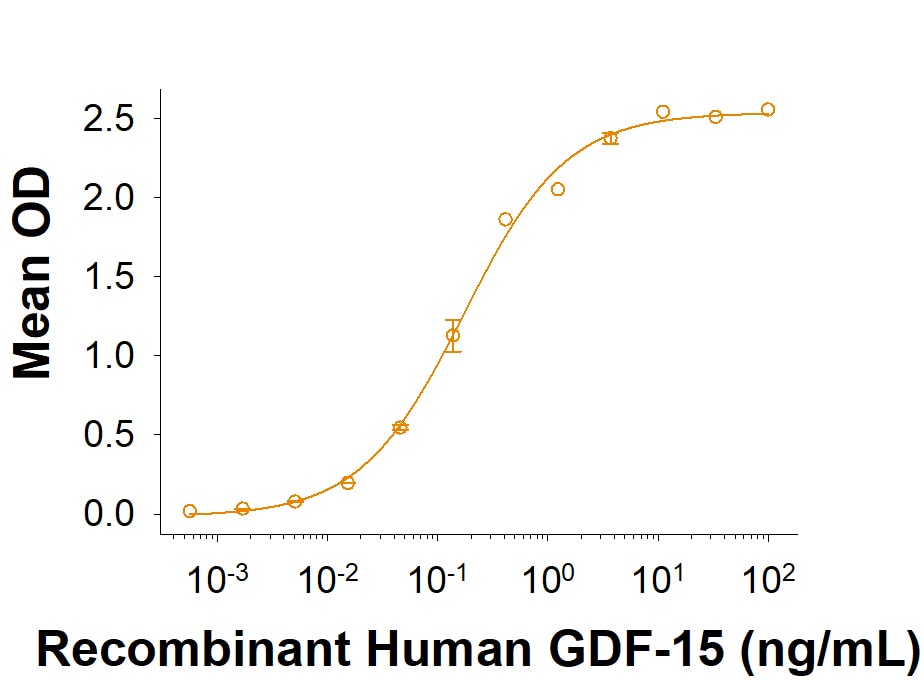
Recombinant Human GDF-15 (Catalog # 9279-GD) binds to Recombinant Human GFR alpha -like Fc Chimera (Catalog # 9697-GR) with an ED50 of 0.0500-0.500 ng/mL.
Recombinant Human GDF-15 (Catalog # 9279-GD) binds to Recombinant Human GFR alpha -like Fc Chimera (Catalog # 9697-GR) with an ED50 of 0.0500-0.500 ng/mL.
Reviewed Applications
Read 2 reviews rated 4.5 using 9279-GD in the following applications:
Scientific Data Images for Recombinant Human GDF-15 (E. coli-expressed) Protein, CF
Recombinant Human GDF‑15 Protein Binding Activity.
In a functional ELISA, Recombinant Human GDF-15 (Catalog # 9279-GD) binds to Recombinant Human GFR alpha -like Fc Chimera (9697-GR) with an ED50 of 0.0500-0.500 ng/mL.Recombinant Human GDF-15 (E. coli-expressed) Protein Bioactivity
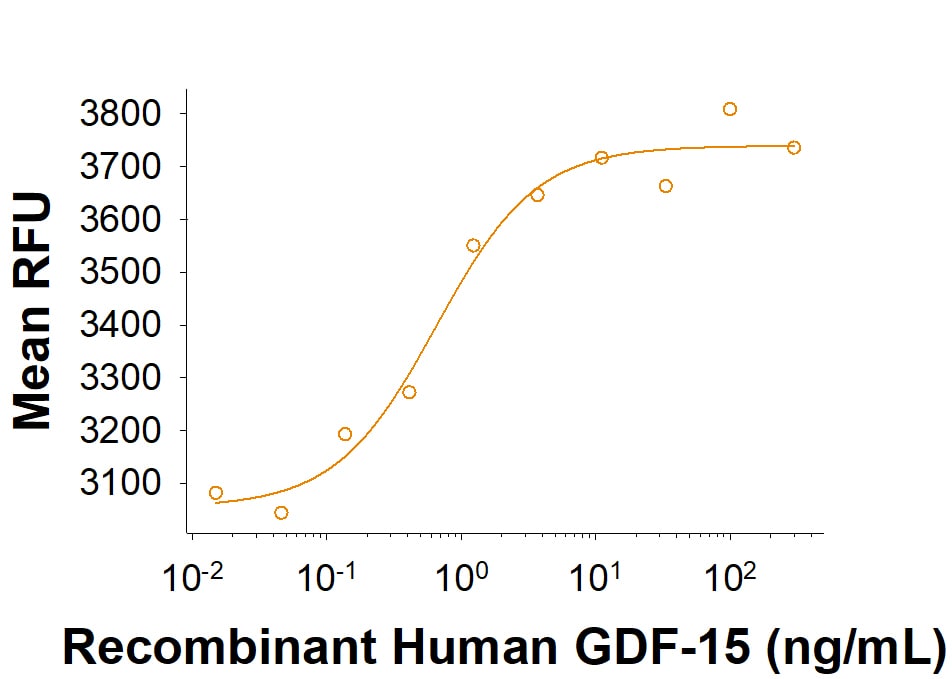
Recombinant Human GDF-15 (Catalog # 9270-GD) activates SRE-SEAP reporter in HEK293 human embryonic kidney cells transfected with human c-Ret and human GFRAL.Formulation, Preparation, and Storage
9279-GD
| Formulation | Lyophilized from a 0.2 μm filtered solution in HCl. |
| Reconstitution | Reconstitute at 250 μg/mL in 4 mM HCl.
Loading...
|
| Shipping | The product is shipped at ambient temperature. Upon receipt, store it immediately at the temperature recommended below. |
| Stability & Storage | Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
|
Calculators
Background: GDF-15
References
- Bootcov, M.R. et al. (1997) Proc. Natl. Acad. Sci. USA 94:11514.
- Unsicker, K. et al. (2013) Cytokine Growth Factor Rev. 24:373.
- Bottner, M. et al. (1999) Gene 237:105.
- Fairlie, W.D. et al. (2001) J. Biol. Chem. 276:16911.
- Paralkar, V.M. et al. (1998) J. Biol. Chem. 273:13760.
- Artz, A. et al. (2016) Blood 128:529.
- Johnen, H. et al. (2007) Nat. Med. 13:1333.
- Strelau, J. et al. (2000) J. Neurosci. 20:8597.
- Strelau, J. et al. (2009) J. Neurosci. 29:13640.
- Whitson, R.J. et al. (2013) J. Cell. Biochem. 114:1424.
- Rossaint, J. et al. (2013) J. Thromb. Haemost. 11:335.
- Song, H. et al. (2012) Mol. Biol. Rep. 39:4017.
- Preusch, M.R. et al. (2013) Eur. J. Med. Res. 18:19.
- Kempf, T. et al. (2011) Nat. Med. 17:581.
- Xu, X.-Y. et al. (2014) J. Biol. Chem. 289:10084.
Long Name
Growth Differentiation Factor 15
Alternate Names
GDF15, MIC-1, NAG-1, PDF, PLAB, PTGF-beta
Gene Symbol
GDF15
UniProt
Additional GDF-15 Products
Product Documents for Recombinant Human GDF-15 (E. coli-expressed) Protein, CF
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Recombinant Human GDF-15 (E. coli-expressed) Protein, CF
For research use only
Related Research Areas
Citations for Recombinant Human GDF-15 (E. coli-expressed) Protein, CF
Customer Reviews for Recombinant Human GDF-15 (E. coli-expressed) Protein, CF (2)
4.5 out of 5
2 Customer Ratings
Have you used Recombinant Human GDF-15 (E. coli-expressed) Protein, CF?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
Showing
1
-
2 of
2 reviews
Showing All
Filter By:
-
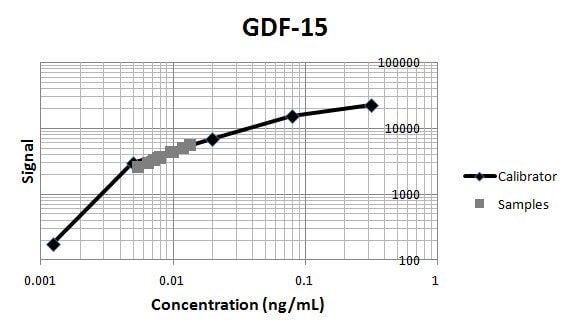
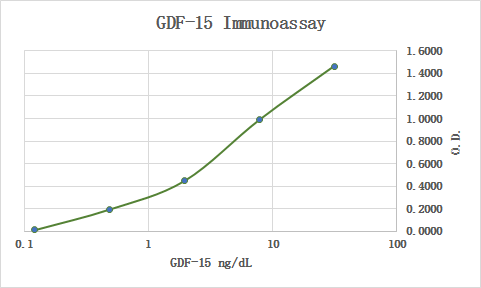
Application: Immunoassay StandardVerified Customer | Posted 11/08/2019
-
Application: Immunoassay StandardVerified Customer | Posted 06/11/2019Used RnD Systems antibodies MAB957 and BAF940 in an GDF-15 immunoassay.
There are no reviews that match your criteria.
Loading...