Ubiquitin is a 76 amino acid (aa) protein that is ubiquitously expressed in all eukaryotic organisms. Ubiquitin is highly conserved with 96% aa sequence identity shared between human and yeast Ubiquitin, and 100% aa sequence identity shared between human and mouse Ubiquitin (1). In mammals, four Ubiquitin genes encode for two Ubiquitin-ribosomal fusion proteins and two poly-Ubiquitin proteins. Cleavage of the Ubiquitin precursors by deubiquitinating enzymes gives rise to identical Ubiquitin monomers each with a predicted molecular weight of 8.6 kDa. Conjugation of Ubiquitin to target proteins involves the formation of an isopeptide bond between the C-terminal glycine residue of Ubiquitin and a lysine residue in the target protein. This process of conjugation, referred to as ubiquitination or ubiquitylation, is a multi-step process that requires three enzymes: a Ubiquitin-activating (E1) enzyme, a Ubiquitin-conjugating (E2) enzyme, and a Ubiquitin ligase (E3). Ubiquitination is classically recognized as a mechanism to target proteins for degradation and as a result, Ubiquitin was originally named ATP-dependent Proteolysis Factor 1 (APF-1) (2,3). In addition to protein degradation, ubiquitination has been shown to mediate a variety of biological processes such as signal transduction, endocytosis, and post-endocytic sorting (4-7).
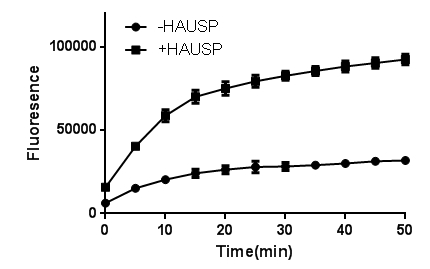
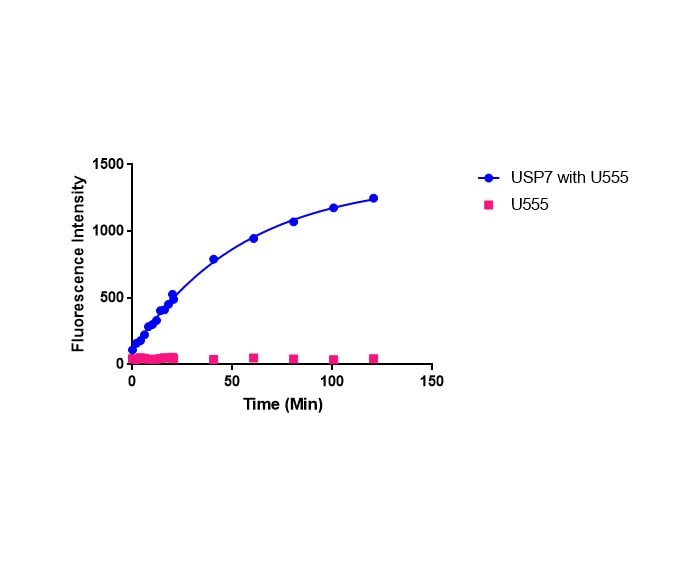
This fluorogenic substrate is based on the C-terminus derivatization of Ubiquitin with Rhodamine 110 (R110) (8). Similar to Ubiquitin-AMC, this is an exquisitely sensitive deubiquitinating enzyme substrate and is useful for studying Ubiquitin C-terminal hydrolytic activity when detection sensitivity or continuous monitoring is essential.