Recombinant Mouse FAP Protein, CF
R&D Systems | Catalog # 8647-SE

Key Product Details
- R&D Systems NS0-derived Recombinant Mouse FAP Protein (8647-SE)
- Quality control testing to verify active proteins with lot specific assays by in-house scientists
- All R&D Systems proteins are covered with a 100% guarantee
Source
Accession Number
Applications
Product Specifications
Source
Leu26-Asp761
Purity
Endotoxin Level
N-terminal Sequence Analysis
Predicted Molecular Mass
SDS-PAGE
Activity
The specific activity is >2000 pmol/min/μg, as measured under the described conditions.
Scientific Data Images for Recombinant Mouse FAP Protein, CF
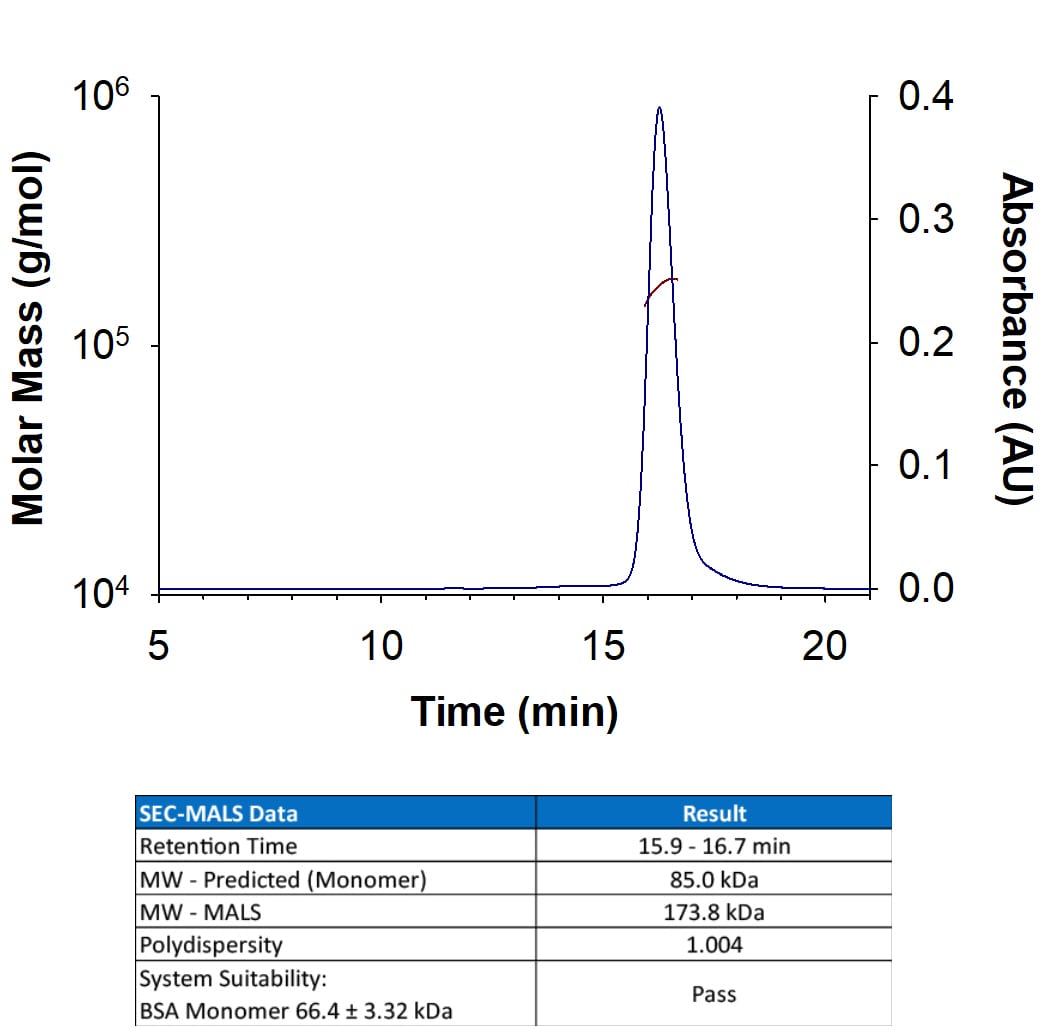
Recombinant Mouse Fibroblast Activation Protein alpha /FAP Protein SEC-MALS.
Recombinant Mouse FAP (Catalog # 8647-SE) has a molecular weight (MW) of 173.8 kDa as analyzed by SEC-MALS, suggesting that this protein is a homodimer. MW may differ from predicted MW due to post-translational modifications (PTMs) present (i.e. Glycosylation).Recombinant Mouse FAP Protein Enzyme Activity
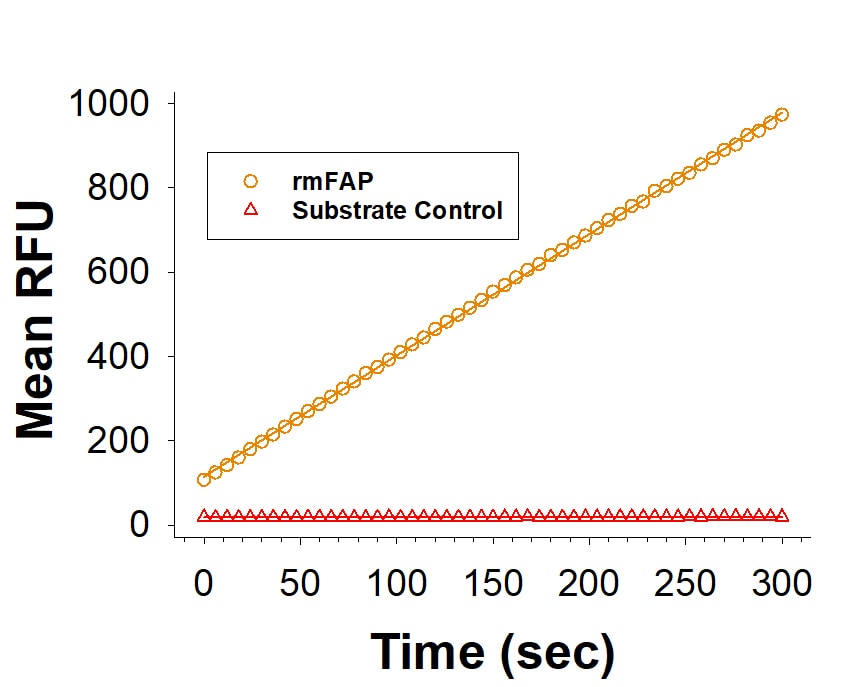
Recombinant Mouse FAP (Catalog # 8647-SE) is measured by its ability to convert the substrate benzyloxycarbonyl-Gly-Pro-7-amido-4-methylcoumarin (Z-GP-AMC) to Z-Gly-Pro and 7-amino-4-methylcoumarin (AMC).Formulation, Preparation, and Storage
8647-SE
| Formulation | Supplied as a 0.2 μm filtered solution in Tris, NaCl and Glycerol. |
| Shipping | The product is shipped with polar packs. Upon receipt, store it immediately at the temperature recommended below. |
| Stability & Storage | Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
|
Background: Fibroblast Activation Protein alpha/FAP
References
- Zi, F. et al. (2015) Mol. Med. Rep. 11:3203.
- Pineiro-Sanchez, M.L. et al. (1997) J. Biol. Chem. 272:7595.
- Niedermeyer, J. et al. (1997) Int. J. Cancer 71:383.
- Scanlan, M.J. et al. (1994) Proc. Natl. Acad. Sci. USA 91:5657.
- Park, J.E. et al. (1999) J. Biol. Chem. 274:36505.
- Rettig, W.J. et al. (1988) Proc. Natl. Acad. Sci. USA 85:3110.
- Bauer, S. et al. (2006) Arthritis Res. 8:R171.
- Aertgeerts, K. et al. (2005) J. Biol. Chem. 280:19441.
- Keane, F.M. et al. (2011) FEBS J. 278:1316.
- Ghersi, G. et al. (2006) Cancer Res. 66:4652.
- Ghersi, G. et al. (2002) J. Biol. Chem. 277:29231.
- Cheng, J.D. et al. (2005) Mol. Cancer Ther. 4:351.
- Cheng, J.D. et al. (2002) Cancer Res. 62:4767.
- Kraman, M. et al. (2010) Science 330:827.
Alternate Names
Gene Symbol
UniProt
Additional Fibroblast Activation Protein alpha/FAP Products
- All Products for Fibroblast Activation Protein alpha/FAP
- Fibroblast Activation Protein alpha/FAP cDNA Clones
- Fibroblast Activation Protein alpha/FAP ELISA Kits
- Fibroblast Activation Protein alpha/FAP Lysates
- Fibroblast Activation Protein alpha/FAP Primary Antibodies
- Fibroblast Activation Protein alpha/FAP Proteins and Enzymes
- Fibroblast Activation Protein alpha/FAP Simple Plex
Product Documents for Recombinant Mouse FAP Protein, CF
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Recombinant Mouse FAP Protein, CF
For research use only
Related Research Areas
Citations for Recombinant Mouse FAP Protein, CF
Customer Reviews for Recombinant Mouse FAP Protein, CF
There are currently no reviews for this product. Be the first to review Recombinant Mouse FAP Protein, CF and earn rewards!
Have you used Recombinant Mouse FAP Protein, CF?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
View specific protocols for Recombinant Mouse FAP Protein, CF (8647-SE):
- Assay Buffer: 50 mM Tris, 1 M NaCl, 1 mg/mL BSA, pH 7.5
- Recombinant Mouse Fibroblast Activation Protein alpha /FAP (rmFAP) (Catalog # 8647-SE)
- Substrate: Z-Gly-Pro-AMC (Bachem, Catalog # I-1145), 10 mM stock in DMSO
- F16 Black Maxisorp Plate (Nunc, Catalog # 475515)
- Fluorescent Plate Reader (Model: SpectraMax Gemini EM by Molecular Devices) or equivalent
- Dilute rmFAP to 0.2 µg/mL in Assay Buffer.
- Dilute Substrate to 100 µM in Assay Buffer.
- Load 50 µL of 0.2 µg/mL of rmFAP into a plate, and start the reaction by adding 50 µL of 100 µM Substrate. Include a Substrate Blank containing 50 µL of Assay Buffer and 50 µL of Substrate.
- Read at excitation and emission wavelengths of 380 nm and 460 nm (top read), respectively, in kinetic mode for 5 minutes.
- Calculate specific activity:
Specific Activity (pmol/min/µg) = | Adjusted Vmax* (RFU/min) x Conversion Factor** (pmol/RFU) |
| amount of enzyme (µg) |
*Adjusted for Substrate Blank
**Derived using calibration standard 7-Amino, 4-Methyl Coumarin (Sigma, Catalog # A9891).
- rmFAP: 0.010 µg
- Substrate: 50 µM
FAQs for Recombinant Mouse FAP Protein, CF
-
Q: Does this enzyme have a tag?
A: No, this enzyme does not have a tag. Please refer to the Source section on the product-specific page or product datasheet for sequence information.