TLR3 Antibody (40C1285.6) - Azide and BSA Free
Novus Biologicals | Catalog # NBP2-33263

Key Product Details
Species Reactivity
Applications
Label
Antibody Source
Format
Product Specifications
Immunogen
Clonality
Host
Isotype
Scientific Data Images for TLR3 Antibody (40C1285.6) - Azide and BSA Free
Western Blot: TLR3 Antibody (40C1285.6)Azide Free [NBP2-33263]
Western Blot: TLR3 Antibody (40C1285.6) - Azide Free [NBP2-33263] - HEK293T cells were transfected with TLR3, TLR3-9-3 and TLR3-3-9. Western blot was performed using anti-TLR3 or anti-HA antibodies. Anti-Beta-actin antibodies were used as a loading control. The representative data from three experiments are shown. Image collected and cropped by CiteAb from the following publication (//doi.org/10.1371/journal.pone.0092391) licensed under a CC-BY license. Image from the standard format of this antibody.Immunohistochemistry: TLR3 Antibody (40C1285.6) - Azide Free [NBP2-33263]
Immunohistochemistry: TLR3 Antibody (40C1285.6) - Azide Free [NBP2-33263] - Tissue section of mouse intestine using TLR3 antibody (clone 40C1285.6) at 1:500 dilution with HRP-DAB detection and hematoxylin counterstaining. The representative image shows a punctate staining of the ER and endosomes in a subset of cells in Peyer's paFlow Cytometry: TLR3 Antibody (40C1285.6) - Azide Free [NBP2-33263]
Flow Cytometry: TLR3 Antibody (40C1285.6) - Azide Free [NBP2-33263] - Expression of TLR3 in Fravel, L363, OPM1, RPMI8226, XG1, and NCI-H929 as determined by flow cytometry. HCMLs were stained using an intracellular staining protocol with TLR3 antibodies followed by relevant secondary fluorescent-conjugated antibodies. Filled histograms (purple) represent the isotype controls and the open histograms (red) indicate TLR3. Data are representative for analysis of >=2 independent experiments. Image collected and cropped by CiteAb from the following publication (//doi.org/10.1371/journal.pone.0060671) licensed under a CC-BY license. Image from the standard format of this antibody.Western Blot: TLR3 Antibody (40C1285.6)Azide Free [NBP2-33263]
Western Blot: TLR3 Antibody (40C1285.6) - Azide Free [NBP2-33263] - All the cell lines displayed a strong expression of TLR3 protein. Cell lysates were electrophoresed and blotted to PVDF membrane, which was probed with TLR3-specific antibody. To confirm the immunoreactivity of the antibody, different positive controls were included. Beta-actin was served as loading control and was used to normalize expression levels between cells. Data are representative for analysis of >=2 independent experiments. Image collected and cropped by CiteAb from the following publication (//doi.org/10.1371/journal.pone.0060671) licensed under a CC-BY license. Image from the standard format of this antibody.Western Blot: TLR3 Antibody (40C1285.6)Azide Free [NBP2-33263]
Western Blot: TLR3 Antibody (40C1285.6) - Azide Free [NBP2-33263] - Analysis of TLR3 in lysates from A) human intestine and F) ovary using TLR3 antibody at 3 ug/ml. Goat anti-mouse HRP conjugate was used as secondary. Image from the standard format of this antibody.Immunohistochemistry: TLR3 Antibody (40C1285.6) - Azide Free [NBP2-33263]
Immunohistochemistry: TLR3 Antibody (40C1285.6) - Azide Free [NBP2-33263] - Analysis of TLR3 in (A) human gut lumen (longitudinal section, transverse region) using this antibody at 10 ug/ml (Data courtesy Dr. Elizabeth Furrie, University of Dundee) and mouse spleen tissue using isotype control (B) and this antibody (C) at 5 mg/mlImmunohistochemistry: TLR3 Antibody (40C1285.6) - Azide Free [NBP2-33263]
Immunohistochemistry: TLR3 Antibody (40C1285.6) - Azide Free [NBP2-33263] - Mouse colon using TLR3 antibody (clone 40C1285.6) at 1:500 dilution with HRP-DAB detection and hematoxylin counterstaining. Intense signal was found in subset of cells at the bases of the crypts. Image from the standard format of this antibody.Flow Cytometry: TLR3 Antibody (40C1285.6) - Azide Free [NBP2-33263]
Flow Cytometry: TLR3 Antibody (40C1285.6) - Azide Free [NBP2-33263] - Expression of TLR3 protein on epithelial cells. HNEC (A), Detroit-562 (B) and FaDu (C) were stained intracellularly with FITC-Abs against TLR3 (open histograms) or appropriate isotype control (shaded histograms) and analyzed by flow cytometry. Representative pictures from one out of three independent experiments are shown. Image collected and cropped by CiteAb from the following publication (//dx.plos.org/10.1371/journal.pone.0098239), licensed under a CC-BY license. Image from the standard format of this antibody.Applications for TLR3 Antibody (40C1285.6) - Azide and BSA Free
Immunocytochemistry/ Immunofluorescence
Immunohistochemistry
Immunohistochemistry-Paraffin
Western Blot
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Formulation
Format
Preservative
Concentration
Shipping
Stability & Storage
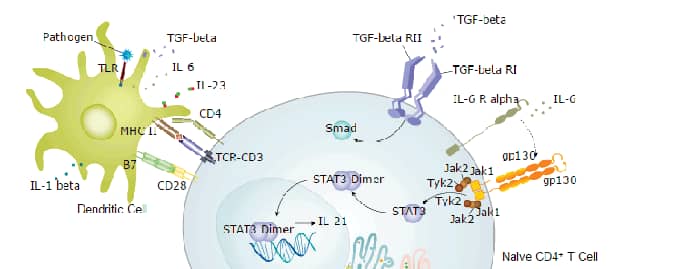
Background: TLR3
Given the role of TLR3 in immune response, its expression or dysfunction has been associated with a number of pathologies from chronic inflammation to autoimmune disorders and cancer (1-5,7). TLR3 is expressed in many cancer types, often related to viral infection, such as cervical cancer, hepatocellular carcinoma (HCC), melanoma, breast cancer, and prostate cancer (1,5). TLR3 signaling has a dual role in cancer, either contributing to pro- or anti-tumor properties depending on the type of cancer (1,5). Therapeutic targeting the TLR3 signaling pathway is under investigation. TLR3 inhibitors or antagonists are being studied for the treatment autoimmune and inflammatory disorders such as of sepsis and atherosclerosis (2,8). TLR3 agonists, either alone or in combination with immune checkpoint inhibitors or therapeutic agents, are being studied as immunotherapeutic treatments of many cancers such as colorectal cancer, prostate cancer, and melanoma (7).
References
1. Zheng X, Li S, Yang H. Roles of Toll-Like Receptor 3 in Human Tumors. Front Immunol. 2021;12:667454. https://doi.org/10.3389/fimmu.2021.667454
2. Zhuang C, Chen R, Zheng Z, Lu J, Hong C. Toll-Like Receptor 3 in Cardiovascular Diseases. Heart Lung Circ. 2022;S1443-9506(22)00080-4. https://doi.org/10.1016/j.hlc.2022.02.012
3. Bianchi F, Pretto S, Tagliabue E, Balsari A, Sfondrini L. Exploiting poly(I:C) to induce cancer cell apoptosis. Cancer Biol Ther. 2017;18(10):747-756. https://doi.org/10.1080/15384047.2017.1373220
4. Matsumoto M, Seya T. TLR3: interferon induction by double-stranded RNA including poly(I:C). Adv Drug Deliv Rev. 2008;60(7):805-812. https://doi.org/10.1016/j.addr.2007.11.005
5. Muresan XM, Bouchal J, Culig Z, Soucek K. Toll-Like Receptor 3 in Solid Cancer and Therapy Resistance. Cancers (Basel). 2020;12(11):3227. https://doi.org/10.3390/cancers12113227
6. Uniprot (O15455)
7. Le Naour J, Galluzzi L, Zitvogel L, Kroemer G, Vacchelli E. Trial watch: TLR3 agonists in cancer therapy. Oncoimmunology. 2020;9(1):1771143. https://doi.org/10.1080/2162402X.2020.1771143
8. Gao W, Xiong Y, Li Q, Yang H. Inhibition of Toll-Like Receptor Signaling as a Promising Therapy for Inflammatory Diseases: A Journey from Molecular to Nano Therapeutics. Front Physiol. 2017;8:508. https://doi.org/10.3389/fphys.2017.00508
Long Name
Alternate Names
Gene Symbol
Additional TLR3 Products
Product Documents for TLR3 Antibody (40C1285.6) - Azide and BSA Free
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Product Specific Notices for TLR3 Antibody (40C1285.6) - Azide and BSA Free
This product is for research use only and is not approved for use in humans or in clinical diagnosis. Primary Antibodies are guaranteed for 1 year from date of receipt.
Customer Reviews for TLR3 Antibody (40C1285.6) - Azide and BSA Free
There are currently no reviews for this product. Be the first to review TLR3 Antibody (40C1285.6) - Azide and BSA Free and earn rewards!
Have you used TLR3 Antibody (40C1285.6) - Azide and BSA Free?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Liperfluo
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars

![Immunohistochemistry: TLR3 Antibody (40C1285.6) - Azide Free [NBP2-33263] Immunohistochemistry: TLR3 Antibody (40C1285.6) - Azide Free [NBP2-33263]](https://resources.rndsystems.com/images/products/TLR3-Antibody-40C1285-6-Azide-Free-Immunohistochemistry-NBP2-33263-img0006.jpg)
![Flow Cytometry: TLR3 Antibody (40C1285.6) - Azide Free [NBP2-33263] Flow Cytometry: TLR3 Antibody (40C1285.6) - Azide Free [NBP2-33263]](https://resources.rndsystems.com/images/products/TLR3-Antibody-40C1285-6-Azide-Free-Flow-Cytometry-NBP2-33263-img0007.jpg)
![Western Blot: TLR3 Antibody (40C1285.6)Azide Free [NBP2-33263] Western Blot: TLR3 Antibody (40C1285.6)Azide Free [NBP2-33263]](https://resources.rndsystems.com/images/products/TLR3-Antibody-40C1285-6-Azide-Free-Western-Blot-NBP2-33263-img0001.jpg)
![Western Blot: TLR3 Antibody (40C1285.6)Azide Free [NBP2-33263] Western Blot: TLR3 Antibody (40C1285.6)Azide Free [NBP2-33263]](https://resources.rndsystems.com/images/products/TLR3-Antibody-40C1285-6-Azide-Free-Western-Blot-NBP2-33263-img0003.jpg)
![Western Blot: TLR3 Antibody (40C1285.6)Azide Free [NBP2-33263] Western Blot: TLR3 Antibody (40C1285.6)Azide Free [NBP2-33263]](https://resources.rndsystems.com/images/products/TLR3-Antibody-40C1285-6-Azide-Free-Western-Blot-NBP2-33263-img0002.jpg)
![Immunohistochemistry: TLR3 Antibody (40C1285.6) - Azide Free [NBP2-33263] Immunohistochemistry: TLR3 Antibody (40C1285.6) - Azide Free [NBP2-33263]](https://resources.rndsystems.com/images/products/TLR3-Antibody-40C1285-6-Azide-Free-Immunohistochemistry-NBP2-33263-img0004.jpg)
![Immunohistochemistry: TLR3 Antibody (40C1285.6) - Azide Free [NBP2-33263] Immunohistochemistry: TLR3 Antibody (40C1285.6) - Azide Free [NBP2-33263]](https://resources.rndsystems.com/images/products/TLR3-Antibody-40C1285-6-Azide-Free-Immunohistochemistry-NBP2-33263-img0005.jpg)
![Flow Cytometry: TLR3 Antibody (40C1285.6) - Azide Free [NBP2-33263] Flow Cytometry: TLR3 Antibody (40C1285.6) - Azide Free [NBP2-33263]](https://resources.rndsystems.com/images/products/TLR3-Antibody-40C1285-6-Azide-Free-Flow-Cytometry-NBP2-33263-img0008.jpg)