320 results for "SCF/c-kit Ligand" in Products
SCF/c-kit Ligand Products
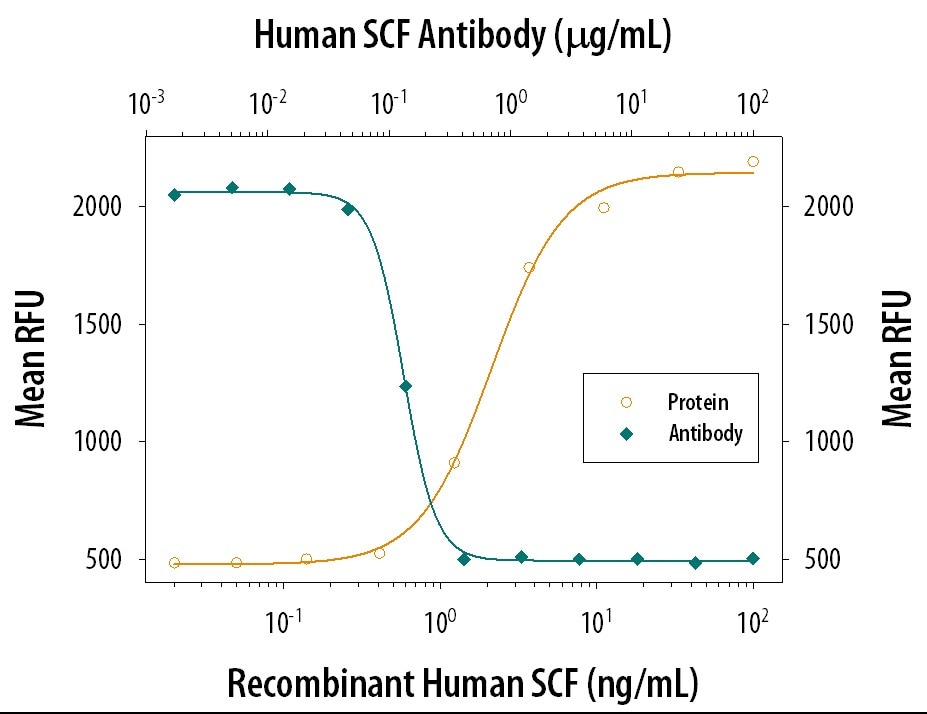
SCF (Stem cell factor), also known as c-kit ligand (KL), mast cell growth factor (MGF), and steel factor (SLF) promotes the survival, differentiation, and mobilization of multiple cell types including myeloid, erythroid, megakaryocytic, lymphoid, germ cell, and melanocyte progenitors. SCF is a primary growth and activation factor for mast cells and eosinophils. SCF is expressed as a transmembrane, but a soluble form can be released by proteolytic shedding. Noncovalent dimers of ...
| Reactivity: | Human |
| Applications: |
Build your own Luminex Assay with our Luminex Assay Customization Tool
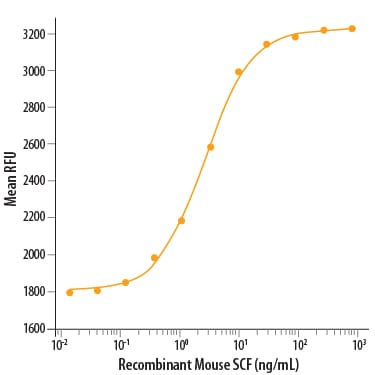
| Reactivity: | Mouse |
| Source: | E. coli |
| Accession #: | Q78ED8 |
| Applications: | Bioactivity |
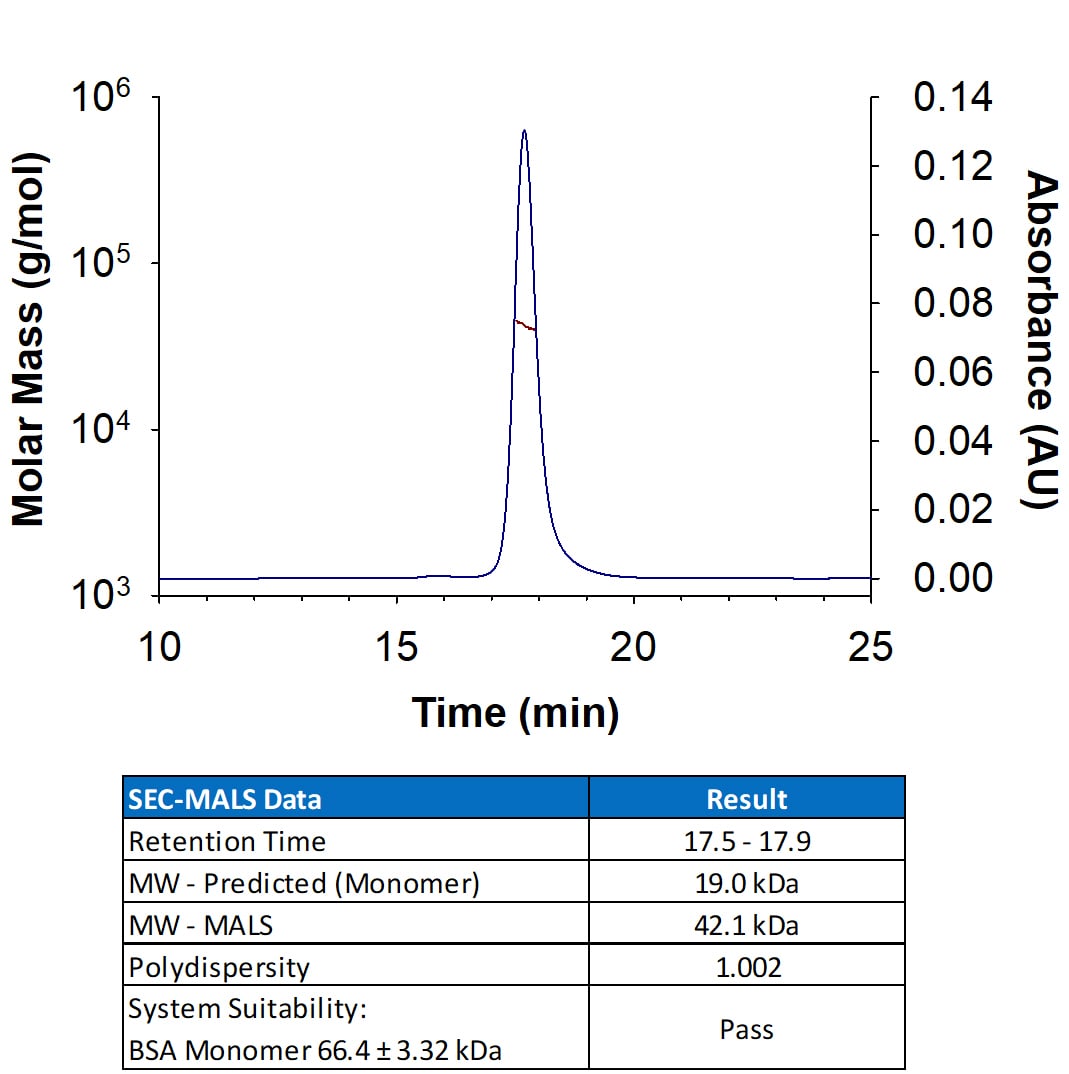
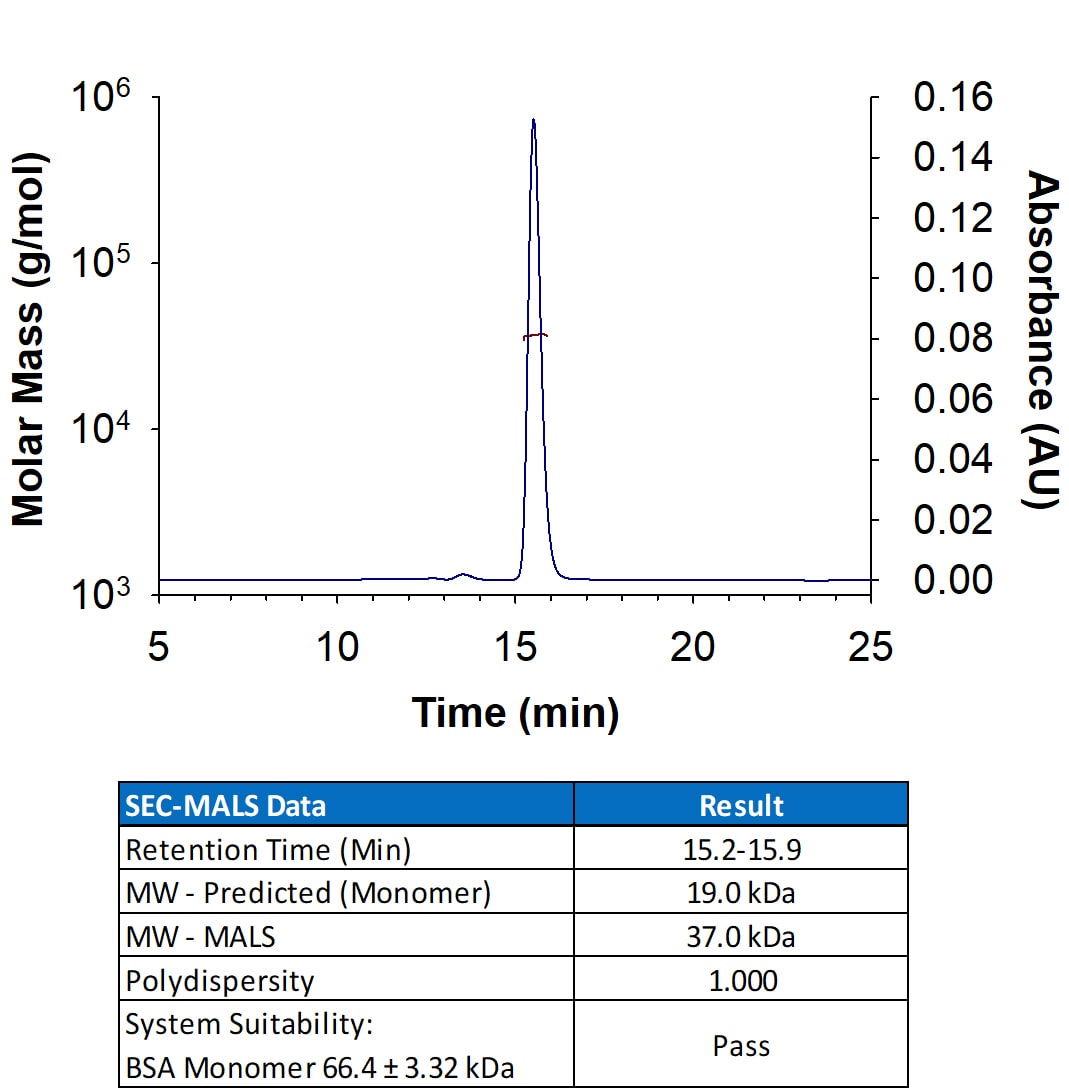
Analyzed by SEC-MALS
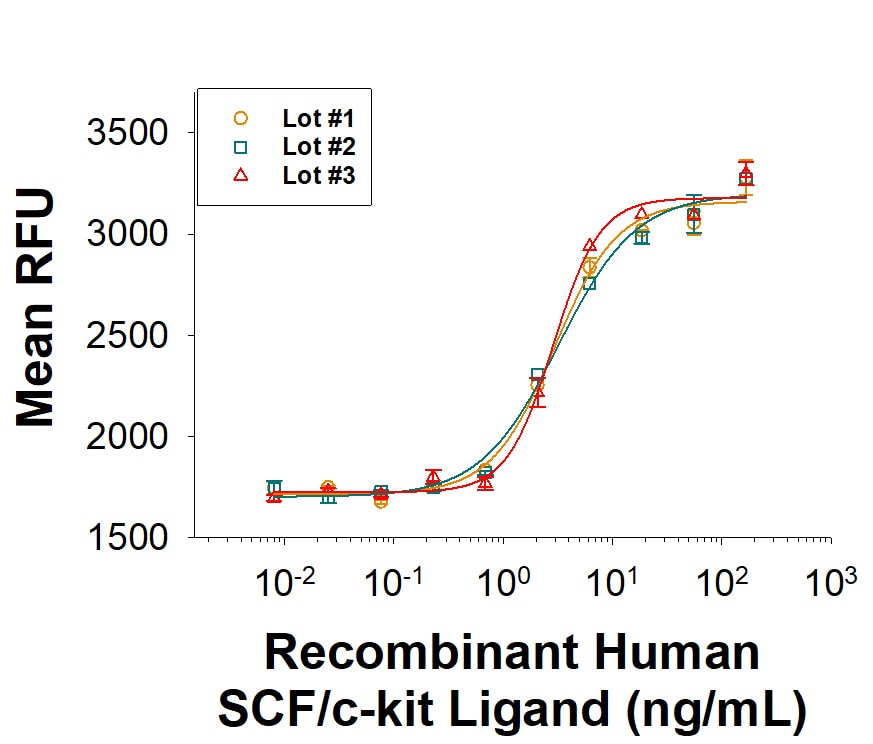
| Reactivity: | Human |
| Source: | E. coli |
| Accession #: | P21583.1 |
| Applications: | Bioactivity |
| Reactivity: | Human |
| Sensitivity: | 9 pg/mL |
| Assay Range: | 31.2-2000 pg/mL (Cell Culture Supernates, Serum, EDTA Plasma, Heparin Plasma, Citrate Plasma) |
| Applications: | ELISA |
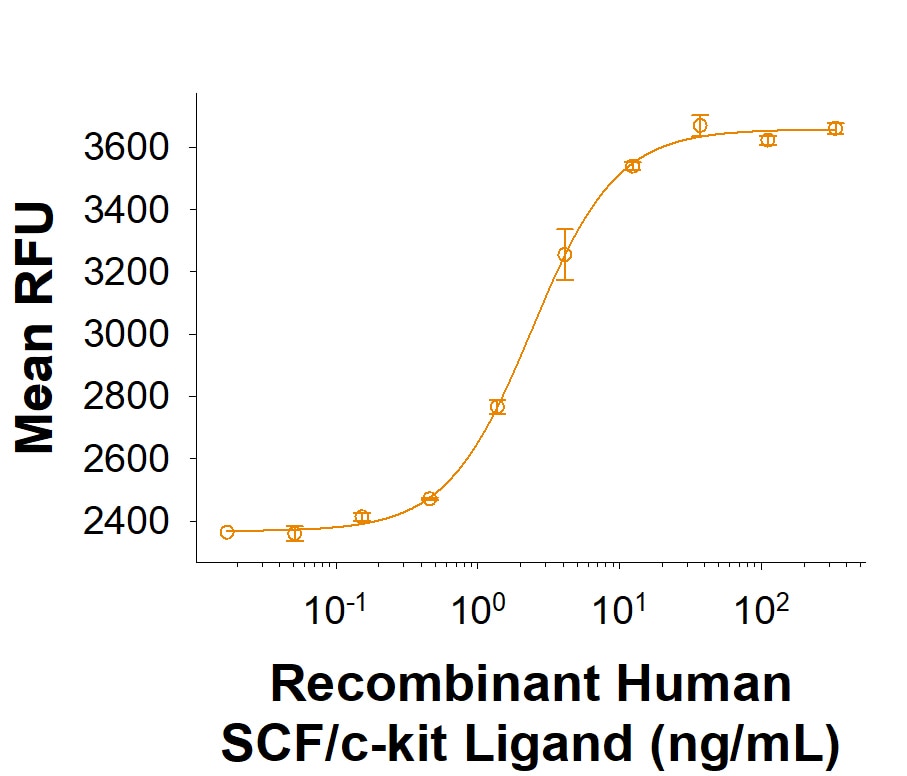
| Reactivity: | Human |
| Source: | E. coli |
| Accession #: | P21583.1 |
| Applications: | Bioactivity |
| Reactivity: | Mouse |
| Sensitivity: | 5 pg/mL |
| Assay Range: | 31.2-2000 pg/mL (Cell Culture Supernates, Serum, EDTA Plasma) |
| Applications: | ELISA |
HEK293-expressed
| Reactivity: | Human |
| Source: | HEK293 |
| Accession #: | P21583.1 |
| Applications: | Bioactivity |
Intended for preclinical researchers who may transition to GMP SCF for their clinical work, Analyzed by SEC-MALS
| Reactivity: | Human |
| Source: | E. coli |
| Accession #: | P21583.1 |
| Applications: | Bioactivity |
| Reactivity: | Human |
| Details: | Goat IgG Polyclonal |
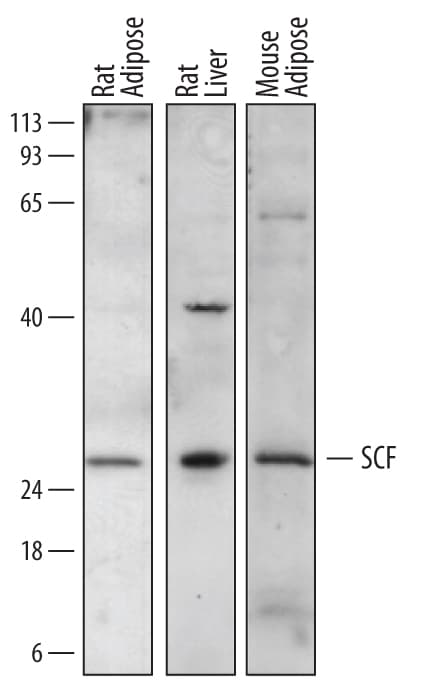
| Applications: | WB, B/N, ICC/IF |
| Reactivity: | Mouse |
| Details: | Goat IgG Polyclonal |
| Applications: | WB, ELISA(Cap), B/N |
Simple Plex Human SCF/c-kit Ligand assay kit for use on Ella instrument. Contains cartridge, sample diluent SD13, and wash buffer.
| Reactivity: | Human |
| Applications: | Simple Plex |
| Reactivity: | Human |
| Details: | Mouse IgG1 Monoclonal Clone #13302 |
| Applications: | WB, ELISA(Cap) |
| Reactivity: | Human, Mouse, Rat |
| Details: | Rabbit IgG Polyclonal |
| Applications: | IHC, WB, ELISA, ICC/IF |
| Reactivity: | Human |
| Details: | Goat IgG Polyclonal |
| Applications: | WB, B/N |
| Reactivity: | Canine |
| Source: | E. coli |
| Accession #: | Q06220 |
| Applications: | Bioactivity |
| Reactivity: | Mouse |
| Details: | Goat IgG Polyclonal |
| Applications: | WB, B/N |
| Reactivity: | Canine |
| Details: | Goat IgG Polyclonal |
| Applications: | WB, ELISA(Cap), B/N, ICC/IF |
| Reactivity: | Human |
| Assay Range: | 31.2-2000 pg/mL |
| Applications: | ELISA |
| Reactivity: | Mouse |
| Assay Range: | 62.5-4000 pg/mL |
| Applications: | ELISA |
| Reactivity: | Mouse, Rat |
| Details: | Sheep IgG Polyclonal |
| Applications: | WB |
| Reactivity: | Mouse |
| Details: | Rat IgG2A Monoclonal Clone #40215 |
| Applications: | B/N |
| Reactivity: | Mouse |
| Details: | Goat IgG Polyclonal |
| Applications: | WB, ELISA(Det) |
| Reactivity: | Feline |
| Source: | E. coli |
| Accession #: | P79169 |
| Applications: | Bioactivity |
| Reactivity: | Feline |
| Details: | Goat IgG Polyclonal |
| Applications: | WB, ICC/IF |
| Reactivity: | Human |
| Details: | Rabbit IgG Polyclonal |
| Applications: | WB |








![Western Blot: SCF/c-kit Ligand AntibodyBSA Free [NBP1-77030] Western Blot: SCF/c-kit Ligand AntibodyBSA Free [NBP1-77030]](https://resources.rndsystems.com/images/products/SCF-c-kit-Ligand-Antibody-Western-Blot-NBP1-77030-img0012.jpg)







![Western Blot: SCF/c-kit Ligand Antibody [NBP2-27162] Western Blot: SCF/c-kit Ligand Antibody [NBP2-27162]](https://resources.rndsystems.com/images/products/SCF-c-kit-Ligand-Antibody-Western-Blot-NBP2-27162-img0002.jpg)